Calculations in Chemistry
90 likes | 339 Views
Calculations in Chemistry. You need to know how to carry out several calculations in Additional and Triple Chemistry

Calculations in Chemistry
E N D
Presentation Transcript
Calculations in Chemistry • You need to know how to carry out several calculations in Additional and Triple Chemistry • This booklet gives you a step by step guide to carrying out these equations and examples of each one. Follow the same rules for the questions in your exam and chemistry calculations are actually quite easy, honest! • These are: • Additional: • Relative formula mass • Mole masses • Percentage of element in a compound • Formula of a compound from its percentage composition • Masses of reactants and products • Percentage yield • Atom economy • Triple: • Titration calculations • Bond energy calculations
Additional Chemistry Calculations Relative atomic and Formula Masses The mass of an atom is too small to deal with in real terms, so we use ‘relative’ masses – Carbon is given a mass of 12, and everything else is compared with it and given a mass, e.g. Oxygen is ‘heavier’, so its relative mass is 16. Relative formula mass can be found by adding up the relative atomic masses of each element in a compound. E.g. Carbon Dioxide (CO2) Carbon has a relative atomic mass of 12 Oxygen has a relative atomic mass of 16 The relative formula mass of Carbon Dioxide is therefore: 12 + (16 x 2) = 44 Moles Because saying ‘relative formula (or atomic!) mass in grams’ is a bit clumsy, we simply say ‘moles’ instead. This means that 1 mole of Carbon Dioxide is 44 grams, or 44g. Simple! The relative atomic mass can be found by looking at the periodic table, It is always the larger of the two numbers. x 2 because it’s “O2”
Percentage of an element in a compound We can use the relative atomic mass (Ar) of elements and the relative formula mass (Mr) of compounds to find out the percentage composition of different elements. E.g. What percentage mass of white Magnesium Oxide is actually Magnesium, and how much is Oxygen? • Work out the mass of MgO • Convert to grams • Work out the percentage 24 + 16 = 40 40g 24 X 100% = 60% is Magnesium, so 40% must be Oxygen! Work out the formula mass of the compound Convert this into grams Work out the percentage by using this equation: 40 Formula of a compound from its percentage composition We can also do this backwards! If we know the percentage composition of a compound we can work out the ratio of atoms. This is known as the Empirical Formula. Sometimes this is the same as the molecular formula, but not always (e.g. water has an empirical and molecular formula of H2O. Hydrogen peroxide's empirical formula is HO, but it’s molecular formula is H2O2. E.g. If 9g of Aluminium react with 35.5g of Chlorine, what is the empirical formula of the compound formed? Aluminium 9 27 Chlorine 35.5 35.5 • Mass of element • Total mass of compound x 100% Divide the mass of each element by its relative atomic mass to find out the number of moles reacted Create a ratio and simplify if necessary Write a formula based on the ratio = 1/3 moles of Aluminium atoms = 1 mole of Chlorine atoms Al : Cl 1/3 : 1 1 : 3 AlCl3
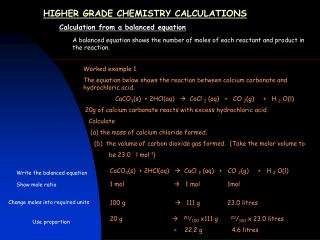
Masses of reactants and products This is an important calculation when we want to know how much of each reactant to react together. For example, sodium hydroxide reacts with chlorine gas to make bleach. If we have too much Chlorine, some will be wasted. Too little and not all of the sodium hydroxide will react. 2NaOH + Cl2 NaOCl + NaCl + H2O How much Chlorine gas should we bubble through 100g of Sodium Hydroxide to make Bleach? 1. NaOH 23 + 16 + 1 = 40g is one mole of NaOH 2. We have 100g in our reaction so… 100 = 2.5 moles 40 3. The chemical equation tells us that we need 2 moles of Sodium Hydroxide (2NaOH) for every mole of Chlorine (Cl2). So we need: 2.5 = 1.25 moles of Chlorine 2 4. 35.5 x 2 = 71g is one mole of Cl2 So we need 1.25 x 71 = 88.75g of Chlorine to react with 100g of Sodium Hydroxide. Bleach Salt Water Sodium Hydroxide Chlorine Work out the mass of one mole of Sodium Hydroxide Calculate how many moles you have in your reaction Work out how many moles of Chlorine you need Convert this into a mass for Chlorine
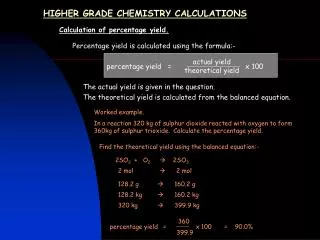
Percentage Yield Rather than talk about the yield of a chemical reaction in terms of mass (grams, tonnes etc.) we can talk about the percentage yield. This gives us an idea of the amount of product that the reaction really makes, compared to what it could possibly make under perfect conditions. There are many reasons why we don’t make 100% every time, such as: • The reaction may be reversible • Some product could be left behind in the apparatus • The reactants may not be pure • It may be difficult to separate the products if more than one are made. Using this reaction “A + B C”, it was found that in perfect conditions, scientists could make 2.5g of C. However, when they tried it out, they only made 1.5. What is the percentage yield of this reaction? 1.5 2.5 Percentage Atom Economy Sometimes reactions will make more than one product. Working out the atom economy is a way of finding out how much of the reactant ends up in the useful product. Ethanol (C2H5OH) can be converted into ethene (C2H4) to make polyethene. However, the reaction also produces water. C2H5OH C2H4 + H2O Mass of useful product = (12 x 2) + (1 x 2) = 28 Mass of all products = 28 + [(1x4) + 16] = 28 + 20 = 48 28 48 The higher the percentage yield and atom economy, the better the reactions are for the Earth’s resources, as there’s less waste! Amount of product produced Maximum amount of product possible x 100% x 100% = 60% percentage yield Relative formula mass of useful product Relative formula mass of all products x 100% x 100% = 58% atom economy
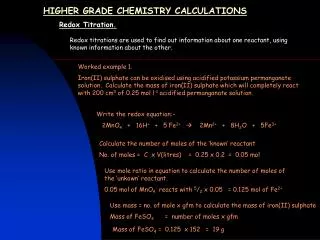
Triple Chemistry Calculations Titration Calculations Titrations are used to find out the volumes of solutions that react exactly. If you know the concentration of one of the solutions, and the volumes that react together are known as well, you can use this to work out the concentration of the other solution. It is important that you know that mol/dm3 is a term for concentration of a solution. It means that X moles of substance are dissolved in 1000cm3 (1dm3) of a solvent. 1mol/dm3 of Sodium Hydroxide means that 40g (the mass of one mole) is dissolved in 1000cm3 of the solvent. 20g dissolved in the same volume would be 0.5mol/dm3. E.g. a student put 25.0cm3 of sodium hydroxide solution with an unknown concentration into a conical flask using a pipette. The sodium hydroxide reacted with exactly 20.0cm3 of 0.50mol/dm3 hydrochloric acid added from a burette. What was the concentration of the sodium hydroxide solution? The equation for this reaction is: NaOH + HCl NaCl + H2O 0.50 moles of Hydrochloric acid are dissolved in 1000cm3 of acid. We used 20cm3 in this reaction. So… 0.50 1000 Calculate the number of moles reacted in the known substance: (Concentration/1000) x volume used X 20 = 0.10 moles of HCl in 20cm3 of acid.
Use the volume of the second unknown solution to work out the concentration in 1dm3 (number of moles/volume of solution) x 1000 Titration Calculations continued… We know from the equation that 1 mole of HCl reacts with 1 mole of NaOH. So, if there is 0.10 moles of HCl in the reaction, there must be 0.10 moles of NaOH as well. Now we can work out the concentration of NaOH. If 0.10 moles of NaOH are in 25cm3 of solution, we can use the equation in the box on the right to work out how many are in 1000cm3, therefore the concentration! 0.10 25 X 1000 = 0.40 moles of NaOH in 1000cm3 of solution = 0.4 mol/dm3 This is the concentration!
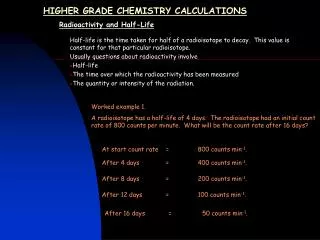
And Finally… Bond Energy Calculations The energy needed to break a bond between two atoms is called the bond energy. It is measured in kJ/mol – kilojoules per mole. We can use this to work out the energy change (ΔH) for chemical reactions. However, we need a list of the bond energies to do this: To calculate bond energy reactions we need to know two things: • How much energy is needed to break the chemical bonds in the reactants • How much energy is released when the new bonds are formed in the products e.g. Hydrogen and Nitrogen are used in the Haber process to make ammonia. What is the overall energy change in this reaction? 3H2 + N2 = 2NH3 You don’t need to remember this! Draw out the structure of each atom so you can work out what bonds are involved Work out the energy needed to break the reactants into atoms. Work out the energy released when the bonds in the products are formed Work out the energy change 2. Energy needed to break one mole of nitrogen, and three moles of hydrogen 945 + (3 x 436) = 2253 kJ/mol 3. Energy released when 3 N-H bonds are formed in one mole of ammonia = 3 x -391 = -1173 Energy released in two moles of ammonia = 2 x -1173 = -2346 kJ The bond energies are: NΞN = 945 kJ/mol H-H = 391 kJ/mol Overall energy change = +2253 – 2346 = -93kJ