Water
Water. Water is a very unusual compound; it is very common and is found in all three conditional states, solid (as ice), liquid ( as water) and gas (as water vapor). . How Much is There?. Approximately three-fourths of the earth is water. The Importance of Water

Water
E N D
Presentation Transcript
Wateris a very unusual compound; it is very common and is found in all three conditional states, solid (as ice), liquid (as water) and gas (as water vapor).
How Much is There? • Approximately three-fourths of the earth is water.
The Importance of Water - Water is the most abundant substance in living systems, making up 70% or more of the weight of most organisms. - Water is of critical importance in all physiological processes in body tissues.
The Importance of Water • - Water carries substances to and from the cells and makes possible the essential processes of absorption, exchange, secretion., and excretion.
Physical properties of water • Pure water is colorless, odorless, and tasteless. • Water is stable as a liquid at standard temperature and pressure. • Density at 4ºC is: 1.0 g/ml.
4. Molecular weight: 18. • 5. Water can dissolve many different substances in large amounts. For this reason, it is called the universal solvent.
6. Heat of Fusion: 80 cal/g. The quantity of heat energy required to convert one gram of solid to liquid at the melting point. 7. Heat of vaporization: 540 cal/g. The amount of heat energy required to convert of liquid to vapor at the boiling point.
8. Solid/liquid density ration: 0.92 Water freezes to form ice at 0ºC but its maximum density is at 4ºC (ice floats over water).
Chemically stable i.e. not easily decomposed. • Reacts violently with sodium and potassium. • Reacts with metal oxides to form bases e.g. CaO. • Reacts with non-metal oxides to form acids e.g. S2O. Chemical Properties of Water
5. Polar molecule 6. Formation of hydrates. Hydrates: when water solutions are evaporated, the substances separate as crystals. Crystals that contain water in their structure are called hydrates.
Structure of Water Molecule • Water is a covalent compound. • Hydrogen atom in water shares its one electron with the oxygen atom. H O H
Oxygen atom attracts electrons towards it more than the hydrogen atoms. Electrons moved closer to the oxygen will give the oxygen atom a slight negative charge while the hydrogen atoms would have a slight positive charge making the water molecule a Polar Molecule. Structure of Water Molecule
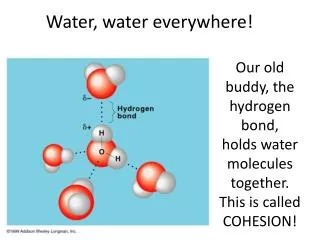
Because of partial charges, water molecules are attracted to each other. Oxygen atom of one water molecule will be attracted to a hydrogen atom of another water molecule. This type of electrostatic attraction is called: Hydrogen Bond. Structure of Water Molecule
Hydrogen Bonds • Extraordinary Properties that are a result of hydrogen bonds. • Cohesive behavior • Resists changes in temperature • High heat of vaporization • Expands when it freezes • Versatile solvent • This type of bond occurs in both inorganic molecules such as water and organic molecules such as DNA.
Cohesion Because of charge difference, water molecules will stick together forming a hydrogen bond. Cohesion is the force of attraction between molecules of the same substance. Cohesion among water molecules plays a key role in the transport of water against gravity in plants
Adhesion Adhesion is the force of attraction between molecules of different substances such as glass and water. Adhesion holds the drops to the glass.
Surface tension • Cohesion actually helps water support • small objects. • Water has a greater surface tension than most other liquids because hydrogen bonds among surface water molecules resist stretching or breaking the surface • Water molecules form a film and this is • called surface tension. • Some animals can stand, walk, or run on • water without breaking the surface.
Moderates Temperatures on Earth Water stabilizes air temperatures by absorbing heat from warmer air and releasing heat to cooler air. Water can absorb or release relatively large amounts of heat with only a slight change in its own temperature.
Solvent for Life • Substances, such as salts, that mix with or dissolve in water are described as hydrophilic (water-loving) • Ionic compounds dissolve in water • Polar molecules (generally) are water soluble • Substances, such as fat , that repel and do not dissolve in water are describes as hydrophobic (water-fearing) • Nonpolarcompounds
Natural water contains many dissolved and suspended materials. • Rainwater contains dissolved gases ''like O2, N2, and CO2'', air pollutants, dust particle, and other particulate matter. • Ground water contains minerals dissolved from soil and some materials. Impurities Present in Water
4. Sea water contains 3.5% dissolved matter (NaCl). 5) Both ground water and sea water contain dissolved and undissolved pollutants. 6) Lake and river water may appear clear but may contain mud or bacteria and other micro- organisms that are harmful to our body. Impurities Present in Water
The purification of water is necessary for removal of impurities and can be done by 4 methods: Distillation Boiling Sedimentation and filtration Aeration Purification of Water
Distillation is a process of converting water to steam and then changing the steam back to water by condensing it. 1. Distillation
Boiling for 15 minutes will kill any bacteria present in the water and makes it safe for drinking although it does not remove dissolved impurities. 2. Boiling
Water is first allowed to stand in large reservoirs where most of the suspended dirt, clay and mud settle down. This process is called sedimentation. • Then, water is filtered to remove the rest of suspended material. After this, water is treated with chlorine to kill the bacteria and make it safe for drinking. 3. Sedimentation and filtration
Water can be purified by exposing it to air for a considerable period of time. • The oxygen in the air dissolves in water and destroys bacteria by the oxidation process. • The oxygen also oxidizes the dissolved organic material in water so that the bacteria have no source of food. 4. Aeration