Understanding Protein Formation: Amino Acids, Peptide Bonds, and Structural Levels
140 likes | 322 Views
This text explores the process of protein synthesis, detailing how amino acids join through condensation reactions to form peptide bonds. It describes the different structures of proteins, from primary to quaternary levels, highlighting the essential roles of hydrogen bonds, ionic bonds, and sulfur bridges in maintaining protein functionality. Additionally, it differentiates between globular and fibrous proteins, discussing examples such as keratin and collagen, and explains the hydrolysis reaction responsible for protein breakdown. Finally, it notes the use of Biuret Reagent for protein identification.

Understanding Protein Formation: Amino Acids, Peptide Bonds, and Structural Levels
E N D
Presentation Transcript
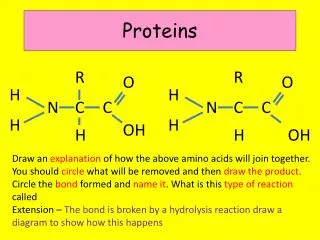
Proteins R R O O H H N C C N C C H H OH H H OH Draw an explanation of how the above amino acids will join together. You should circle what will be removed and then draw the product. Circle the bond formed and name it. What is this type of reaction called Extension – The bond is broken by a hydrolysis reaction draw a diagram to show how this happens
Proteins • Proteins are the most complex and diverse group of bioligical compounds. They have an astonishing range of different functions: • structure e.g. collagen (bone, cartilage, tendon), keratin (hair), actin (muscle) • Enzymes e.g. amylase, catalase, pepsin (>10000) • Transport e.g. haemoglobin (oxygen), transferrin (iron) • Pumps e.g. sodium-potassium pumps in cell membranes • Hormones e.g. insulin, glucagon, adrenalin • Antibodies • Blood clotting • And many more
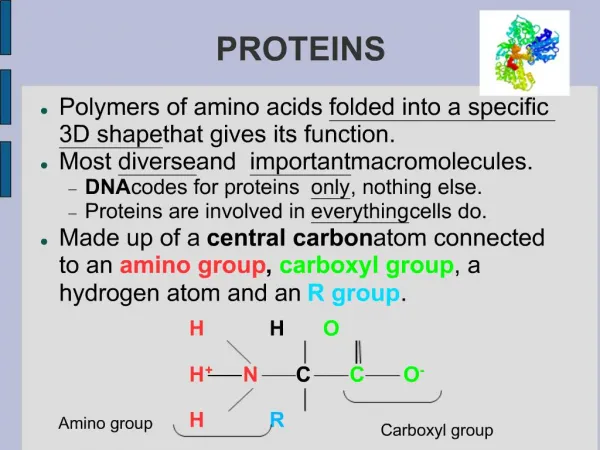
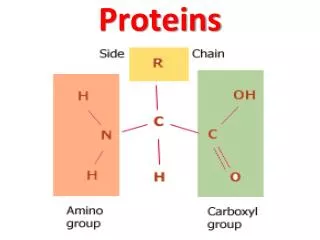
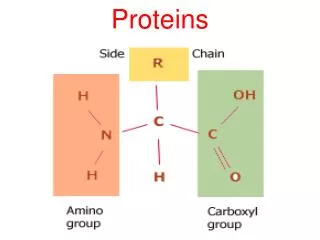
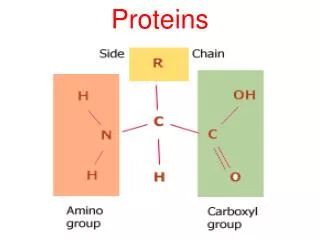
Proteins Proteins are made of amino acids which have a central carbon atom with three different chemical groups attached: R-group (variant) Carboxylic acid group Amino group Alpha carbon Amino acids are so called because they have both amino groups (-NH2) and acidic groups (-COOH). Amino acids are made of the five elements C H O N S There are 20 different R-groups and so 20 different amino acids. This means that there are many, many different proteins with differing numbers and combinations of amino acids
Proteins- making and breaking Joining amino acids involves, again, a condensation reaction. The bond formed is called a peptide bond Two amino acids form a dipeptide, many amino acids form a polypeptide. In a polypeptide, one end is still the amino group and the other end the acidic group. The same type of reaction, hydrolysis, is again involved in breaking down (or hydrolysing) proteins. This can be achieved in the presence of protease enzyme or by boiling with dilute acid.
Protein structure • Polypeptides are just a string of amino acids, but they fold up to form the complex structures of working proteins. To help understand protein structure it is broken down into four levels – but be aware that these are not real sequential stages in protein formation • PRIMARY STRUCTURE • SECONDARY STRUCTURE • TERTIARY STRUCTURE • QUARTERNARY STRUCTURE
Protein: primary structure • This is just the sequence of amino acids in the polypeptide chain, so is not really a structure at all This can also be shown using the three letter abbreviations for each amino acid: Gly – Pro – His – Leu – Tyr – Ser – Trp – Asp - Lys
Protein: secondary structure This is the folding that then occurs, being held together by hydrogen bonds between the amino and carboxyl groups. The two main types of secondary structure are the alpha helix and the beta pleat. In the alpha helix the polypeptide chain is wound round to form a helix that is held together by many hydrogen bonds. In the beta pleat, the polypeptide chain zig-zags back and forward, once again held together by hydrogen bonds
Protein: tertiary structure This is the three dimensional structure formed by the folding up of the whole chain, with every proteins properties and functions being related to this. E.g. the unique shape of an enzymes active site is due to its tertiary structure. Three kinds of bond hold this structure together: Hydrogen bonds,which are relatively weak Ionic bonds between the R-groups, which are quite strong Sulphur bridges between the sulphur containing amino acids, which are strong
Protein: quarternary structure This structure is found only in those proteins that contain more than one polypeptide chain, and simply means how the different chains are arranged together e.g. haemoglobin
Globular or Fibrous? The final 3-D shape of a protein can be described as globular or fibrous GLOBULAR: most proteins, soluble, have biochemical roles e.g. enzymes, receptors, hormones FIBROUS: look like “ropes”, are insoluble and have structural functions e.g. Collagen, keratin
Keratin • Key material making up the outer layer of the human skin • Main structural component of hair and nails
Collagen • Found exclusively in animals • Main component of connective tissue • 25-35% protein in the body
Proteins Proteins can be identified with blue Biuret Reagent (copper sulphate and sodium hydroxide). Blue Biuret reagent turns lilac in the presence of protein