LOCAL ANAESTHETIC AGENT
980 likes | 1.34k Views
LOCAL ANAESTHETIC AGENT. By Dr Nur Hafizah binti Ibrahim Moderator: Dr Najwa binti Mansor. OUTLINE OF PRESENTATION. Definition History Commercial Preparations Structure-activity Relationships Mechanism of Action Minimum Concentration Pharmacokinetics Toxicity . DEFINITION.

LOCAL ANAESTHETIC AGENT
E N D
Presentation Transcript
LOCAL ANAESTHETIC AGENT By DrNurHafizahbinti Ibrahim Moderator: DrNajwabintiMansor
OUTLINE OF PRESENTATION • Definition • History • Commercial Preparations • Structure-activity Relationships • Mechanism of Action • Minimum Concentration • Pharmacokinetics • Toxicity
DEFINITION • A drug that produce reversible conduction blockade of impulses along central and peripheral nerve pathways after regional anesthesia, producing autonomic nervous system blockade, sensory anesthesia and skeletal muscle paralysis in the area innervated. • Interruption of transmission of autonomic, somatic sensory and somatic motor impulses. • Removal of the local anesthetic is followed by spontaneous and complete return of nerve conduction, with no evidence of structural damage to nerve fibers as a result of the drug’s effects.
HISTORY • Cocaine • 1st local anesthetic introduced by Kollar in 1884, for use in opthalmology. • It is a naturally occurring compound extracted from leaves of Erythroxylon coca, a plant growing in the Andes Mountains. • Halsted recognized the ability of injected cocaine to interrupt nerve impulse conduction, leading to the introduction of peripheral nerve block anesthesia and spinal anesthesia. • Another unique feature of this drug is the ability to produced localized vasoconstriction.
1st synthetic local anesthetic was the ester derivative procaine, introduced by Einhorn in 1905. • Lidocaine was synthesized as an amide local anesthetic by Lofgren in 1943. • It produces more rapid, intense and longer-lasting effect that procaine. • It is also effective topically and a highly efficacious cardiac antidysarhythmic drug.
COMMERCIAL PREPARATIONS • Poorly soluble in water – marketed most often as water-soluble hydrochloride salt. • These HCl salts are acidic (pH 6) – contributing to the stability of LA. • An acidic pH also important if epinephrine is present in LA solution, because this cathecolamine is unstable at alkaline pH. • Sodium bisulphite (strongly acidic), may be added to LA-ephephrine solutions to prevent oxidative decomposition of epinephrine.
STRUCTURE-ACTIVITY RELATIONSHIPS • Consist of a lipophilic and a hydrophilic portion separated by connection hydrocarbon chain. • Lipophilic group: • usually unsaturated aromatic ring such as paraaminobenzoic acid. • Essential for anesthetic activity • Hydrophilic group: • Usually a tertiary amine (diethylamine). • A hydrocarbon chain (consist of ester or amide) links both portion to produce delicate balance between lipid solubility and water solubility
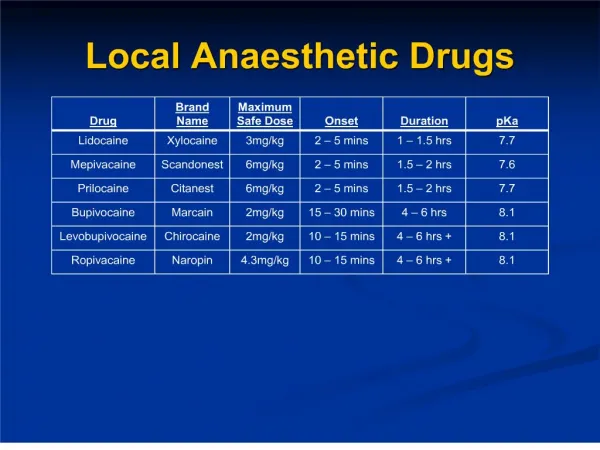
Classifications of LA ESTER AMIDE Lidocaine Prilocaine Bupivacaine Articaine Mepivacaine ***If the local anesthetic has two ‘i’s in its name, it’s an amide • Procaine • Tetracaine • Amethocaine • Cocaine
Physicochemical properties • The chemical structure and physicochemical characteristics of LA affect their clinical properties. • Modification of the chemical structure (lengthening of the hydrocarbon chain within critical length or increasing the number of carbon atoms in the aromatic ring or tertiary amine) may alter lipid solubility, potency, rate of metabolism & duration of action • In particular, these are modified by: • Lipid solubility • Protein binding • Dissociation constant (pKa value)
Lipid solubility • Lipid solubility of diff anesthetics governs their ability to penetrate perineuronal tissues and neural membrane, and reaches their site of action in neuroplasm • More lipid soluble penetrates membrane more easily, less molecule requires for nerve conduction blockade i.e. more potent • E.g. Bupivacaine, levobupivacaine and ropivacaine are app 3-4x as potent as lidocaine or prilocaine due to differences in their lipid-solubility
Protein binding: • Tissue protein binding primarily affect the duration of action of LA • plasma protein binding> longer duration of action • Plasma protein binding acts as depot • Tissue receptor binding – removed at a slower rate • Examples: • Procaine is not extensively bound to tissue protein, has short duration of action • Bupivacaine, levobupivacaine & ropivacaine are extensively bound to plasma and tissue protein --- prolonged effect
Dissociation constant (pKa value) • pKa is equal to pH at which the concentration of ionized base and non-ionized base are equal. • It can be used to calculate the proportion of 2 forms that are present in solution at diff pH values pH = pKa + log 10 (BH+) / (B) • For weak base, (unionised ) / (ionised) = 10 (pH- pKa)
Is the most important factor affecting rapidity of onset of action. • pKa value governs the proportions of LA that is present in non-ionized form at physiological pH values and therefore available to diffuse across tissue barrier to its site of action. • LA with a pKa near physiological pH will have a greater degree of unionized molecules → More LA diffused across membrane → rapid onset of action
MECHANISM OF ACTION • Modulated-receptor theory • Frequency-dependent blockade • Guarded-receptor theory • Membrane Expansion theory • Surface-charge theory
Modulated-receptor Theory • Bindings of LA is stereospecific and dependent on the conformation state of the Na channel. • 3 functional states: • Activated, open • Inactivated/refractory, closed • Resting, closed • LA has high affinity for the open and inactivated closed state and low affinity for the rested closed state.
LA prevents transmission of nerve impulses (conduction blockade) by inhibiting passage of Na ions through ion-selective Na channels in nerve membrane. • Na channel itself is a specific receptor for LA molecules. • So, occlusion of open Na channel by LA molecules contributes little to overall inhibition of Na permeability. • Failure of Na ion channel permeability to increase will slows the rate of depolarization such that threshold potential is not reached and thus an action potential is not propagated
Frequency-dependent blockade • Defines a situation where the more frequent the channel are activated, the greater the degree of block produced. • During action potential, LA exhibit higher affinity to receptor at Na channel. • But when action potential is over, affinity is less and some unbind. • If another action potential (frequency) arrives before all drugs unbound, and additional increment of blocking will occur.
Guarded-receptor Theory • The affinity towards receptor is constantly high in contrast to first theory which depend on conformational state of Na channel. • However the access of LA to the receptor is limited by channel.
Membrane expansion theory • Lipophilic LA incorporated into lipid bilayer causing a volume expansion & distortion to the conformation of axonal membrane and hence the Na channel resulting in its inactivation. • This is mediated by unionized forms.
Surface charge theory • The protonated form of LA neutralized the negative charge of membrane of the nerve which resulting in hyperpolarizing the nerve.
MINIMUM CONCENTRATION • Minimum concentration (Cm) of LA necessary to produce conduction blockade of nerve impulses. • It is analogous to the MAC for inhaled anesthetic. • Influenced by: • nerve diameter (↑D ↑Cm), • Myelinated nerve ↑Cm • Frequency of nerve stimulation (↑F ↓Cm) • Tissue pH (↑pH ↓Cm)
Differential Conduction Blockade: • Is illustrated by selective blockade of preganglionic sympathetic nervous system B fibers using low concentration of LA. • Slightly higher concentration of LA interrupt conduction in small C fibers and small-and-medium sized A fibers, with loss of sensation for pain and temperature. • However touch, proprioception and motor function still intact. • Sometimes misinterpreted as failure of LA in an anxious patient.
PHARMACOKINETICS • Absorption • Distribution • Metabolism • Excretion
Absorption • Influenced by : • Site of administration • Dosage • Vasoconstrictor effect • Pharmacologic characteristic of the drugs • Patient’s factor age, cardiovascular status and hepatic function.
Site of administration: • Intravenous fastest • Mucuos membrane/trachea • Intercostal block • Caudal block • Epidural block • Brachial plexus block • Peripheral nerve block • Subcutaneous infiltration slowest
Dosage • Blood level of LA is related to total dose of drug rather than specific volume or conc of solution • Linear relationship between total dose & peak blood concentration achieved • Patient’s factor • In pregnancy ↑ LA effect d/t ↓ protein binding • In abscess ↓ LA effect d/t more ionized fraction in acidic pH • In hyperkalemic state ↑ LA effect d/t more inactive channel
Vasoconstrictor effect: • Vasoconstriction at site of injection • ↓ rate of absorption, ↓ Systemic toxicity, ↑ duration of action • By addition of adrenaline 5μg/ml (1:200 000) • Higher Dosage offers no additional benefits but increases symphatomimeticactivites • Ropivacaine & Cocaine has intrinsic vasoconstrictor activities • Lignocaine, mepivacaine, bupivacaine, etidocaine exhibit vasodilator effects
all local anesthetics possess some degree of vasoactivity; most producing some level of vasodilation • ester local anesthetics are potent vasodilatingdrugs • Procaine (Novocaine) possesses tremendous vasodilating abilities which are employed to halt arteriospasm (accidental IA injection)
Specific properties of the drugs ↑ Lipid solubility reduced protein binding reduce rate of ↓ pKa absortion Degree of ↓ molecular ionization weight
Distribution • Depends on organ uptake, which determined by: • Tissue perfusion • Tissue/blood partition coefficient • Tissue mass • Lung extraction • Placental transfer
Tissue perfussion: • highly diffuse organ (brain, lungs, liver, kidney & heart) are responsible for initial rapid uptake • Followed by slower redistribution to moderately perfused tissue (muscle & gut) • Tissue/blood partition coefficient • strong plasma protein binding egBupivacaine,tends to retain LA in the blood • Influence by changes in concentration of alpha1 acid glycoprotein (eg; pregnancy, old age, concurrent liver disease) • high lipid solubility; facilitates tissue uptake
Tissue mass: • muscle provides greatest reservoir for LA agents d/t its large mass • Lung extraction: • First pass pulmonary extraction esplidocaine, bupivacaine & prilocaine. • Limit the concentration of drug that reaches systemic circulationfor distribution to the coronary and cerebral circulation.
Placental transfer: • Highly plasma protein binding LA limits diffusion across placenta • Esters undergo rapid hydrolysis hence not available for transfer across placenta • Acidosis in fetus, which may occur during prolonged labour, can result in accumulation of LA molecules in the fetus (ion trapping) • This results in an accumulated concentration of drug in the fetus for two reasons. • once the drug becomes ionized it cannot readily diffuse back across the placenta. This is known as ion trapping. • a concentration gradient of nonionized drug is maintained between the mother and the fetus.
Ion trapping • Fetal blood is slightly more acidic than maternal blood, with a pH about 0.1 unit less than maternal blood pH. • The lower pH of fetal blood facilitates the fetal uptake of drugs that are basic. • Weakly basic drugs, such as local anesthetics and opioids, that cross the placenta in the nonionized state become ionized in the fetal circulation
Metabolism of ESTER • Undergo hydrolysis by cholinesterase enzyme, in the plasma and liver • Rate of hydrolysis varies, and resulting metabolites are pharmacologically inactive • PABA may be associated with allergic reaction • Systemic toxicity is inversely propotional to the rate of hydrolysis • In SAB, in view of lack of cholinesterase enzyme in cerebrospinal fluid, so the termination of action of intrathecal LA depends on absorption into the blood stream.
Prolonged in neonates, liver dysfunction, ↑BUN, parturient and atypical plasma cholinesterase homozygotes activity only ~50% in the newborn and doesn’t reach adult values until about 1 yr
Metabolism of AMIDE • Metabolized by microsomal enzyme located primarily in the liver. amide base aminocarboxilic acid + cyclic aniline derivative ↓ ↓ N-dealkylationhydroxylation • The rate of metabolism are more slower and complex as compared to ester • Systemic toxicity are more likely.
initial reaction N-alkylation subsequently hydrolysis • exception to Prilocaine • hydrolysis take place first forming o-toluidine • further metabolized to 4 and 6-hydroxytoluidine • o-toluidine responsible in metHb in high doses • Benzocaine also can cause metHb • Rx: methylene blue ( Fe3+ Fe2+ )
COCAINE • an alkaloid derived from leaves of Erythroxylon coca • at low dose : • produces a feeling of well-being and euphoria • BRADYCARDIA d/t central vagal stimulation • block uptake of noradrenaline results vasoconstriction and mydriasis
At higher dose: • stimulates vomiting center • central and peripheral sympath stimulation • tachycardia • ↑ capacity muscular work • eventually causes • Convulsion • coma • myocardial depression • medullary depression • respiratory depression • VF and DEATH
cocaine blocks conduction when applied directly to nerve tissue & may therefore be used in surface anesthesia • cocaine was used in the past for ophthalmological procedures, however has now been abandoned • due to sloughing of the corneal epithelium & increased intraocular pressure • the only use today is as a topical local anaesthetic in ENT (5%) • cocaine itself constricts blood vessels and the use of adrenaline is contraindicated as it sensitizes the myocardium