Amino Acids and Protein Structure: Essential Chemistry for Biology Studies
190 likes | 316 Views
Explore the diverse world of amino acids, from their chemical structure to acid-base properties. Learn about protein structure levels and the role of amino acids. Dive into titration calculations and understand the titration of glutamic acid in detail.

Amino Acids and Protein Structure: Essential Chemistry for Biology Studies
E N D
Presentation Transcript
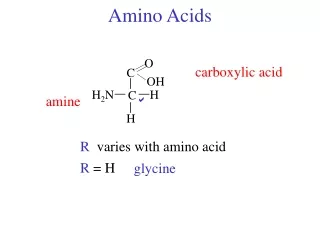
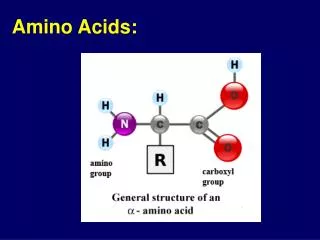
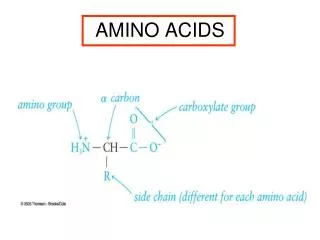
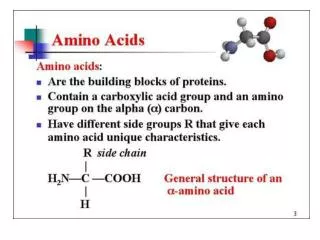
O C OH Amino Acids carboxylic acid H2N H C amine H R R varies with amino acid R = H glycine
-CH-CH3 CH3 -CH2-CH-CH3 CH3 -CH-CH2-CH3 CH3 Nonpolar R-groups -H glycine alanine -CH3 valine proline 2o amine leucine phenylalanine isoleucine methionine -CH2-CH2-S-CH3
-CH2-C-NH2 -CH2-C-NH2 O O -CH-CH3 -CH2 OH Polar R-groups serine asparagine -CH2-OH threonine glutamine tyrosine tryptophan cysteine -CH2-SH
O = -CH2-CH2-C OH O = -CH2-C OH Acidic R-groups glutamic acid aspartic acid
-CH2-CH2-CH2-NH-C-NH2 = NH Basic R-groups lysine -CH2-CH2-CH2-CH2-NH2 arginine histadine
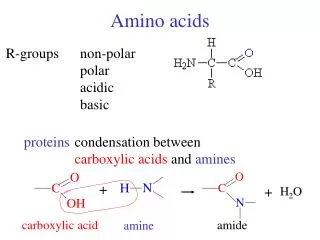
C H H2N R O C OH Amino acids O- H3N+ H R glycine non-chiral all other -amino acids in proteins L-enantiomers zwitterion at neutral pH (pH = 7.0) very high b.p. (> 200oC) very soluble in water
C H C H H3N+ H2N C H H2N R R R O O O C C C O- OH OH acid-base chemistry O- H3N+ low pH neutral pH high pH amine protonated amine and c.a. deprotonated amine and c.a. protonated c.a. deprotonated no net charge negative charge positive charge pH = pHI isoelectric point amino acids diprotic acids 2 pKa
H3N+ C H R O C OH Titration of an amino acid alanine R = CH3 pKa1 = 2.34 pKa2 = 9.69 4.57 x 10-3 Ka1 = 10-2.34 = CH3 Ka1 = [H+][A-] [H+] [A-] [HA] 0.1 M [HA] initial 0 0 0.1 change +x +x -x equil. +x +x 4.57 x 10-3 = x2 0.1-x 0.1 - x • pH = 1.87 [H+] X = 2.14 x 10-2 =
9.0 8.0 7.0 6.0 pH 5.0 4.0 3.0 2.0 1.0 0 equivalents of OH- net charge +1 ×
net charge 9.0 8.0 7.0 6.0 pH 5.0 4.0 3.0 2.0 × 1.0 0 equivalents of OH- +1 +1/2 0.05 M pKa1 = 2.34 0.05 M 0.05 M × pH = pKa + log [A-] [HA] 0.05 M 1/2 pH = pKa = 2.34
pKa2 = 9.69 9.0 8.0 7.0 6.0 pH 5.0 4.0 3.0 × 2.0 × 1.0 0 equivalents of OH- net charge at equivalence point: +1 +1/2 0 pKa1 = 2.34 × isoelectric point pKa1 + pKa2 pH = 2 pH = (2.34 + 9.69)/2 1 1/2 pH = pHI = 6.02
net charge 9.0 8.0 7.0 6.0 pH 5.0 4.0 3.0 2.0 × 1.0 0 equivalents of OH- at 2nd half-way point: +1 +1/2 0 -1/2 -1 × × pKa2 = 9.69 × pH = pKa2 = 9.69 1 1/2 3/2
resonance structure amides dipeptide glycine alanine Ala-Gly +H2O
H H H _ _ _ = = = O O O R R R _ _ _ Proteins “backbone” _ H N1- C1- C1- N2- C2- C2- N3- C3- C3- OH peptide bonds C-terminal residue N-terminal residue biological activity = structure 4 levels protein structure order of the amino acids Primary structure =
H H H _ _ _ _ H N1- C1- C1- N2- C2- C2- N3- C3- C3- OH = = = O O O R R R _ _ _ Secondary structure hydrogen bonding backbone groups H-bond donors H-bond acceptors -helix Two main secondary structures: -sheet
H C N = O C = O Alpha helix Every C=O bonded to N-H 4 residues away forms a helix core is backbone R-groups outside 3.6 amino acids per turn proline no H-bonding breaks helix
Beta sheet Every C=O bonded to N-H far apart in 1o structure on different chains peptide chains extended side-by-side maximal H-bonding for anti-parallel chains small R-groups above and below the sheet if not -helix or -sheet random coil
H3N+ C H O = CH2CH2C R OH O C OH glutamic acid R = - CH2CH2COOH (-COOH) pKa1 = 3.20 pKa2 = 4.25 (R-COOH) pKa3 = 9.67 (-NH3) It will take ___ equivalents to titrate glutamic acid 3 1st group 2nd group 3rd group
O -2 +1 0 -1 C 12 OH H3N+ H C = O CH2CH2C 10 × OH 8 pKa1 = 3.20 × pH pKa2 = 4.25 pHI = 3.7 pKa3 = 9.67 × 4 × × = 3.7 3.2 + 4.25 2 2 = 7.0 4.25 + 9.67 2 1 2 3 equivalents OH-