Essential Elements in Bioanorganic Chemistry
Explore the essential and toxic trace elements, carbon, hydrogen, oxygen, nitrogen, sulfur, phosphorus, calcium, magnesium, chromium, copper, and more in biological systems. Understand their roles, deficiencies, and toxicities.

Essential Elements in Bioanorganic Chemistry
E N D
Presentation Transcript
Elements Prof. Dr. Richard Průša
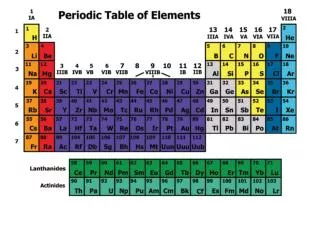
Elements • Bioanorganic chemistry • C, N, P, O, H, S: proteins, nucleic acids, sugars, lipids • Ionts: K, F, Cl, I, Na, Ca, Fe • Essential trace elements: Mg, Cr, Co, Cu, Mo, Se, Zn • Toxic trace elements: As, Ba, Cd, Pb, Li, Hg, Pt
Elements • Trace elements: tissue conc. less than 50 mg/kg • Biologically active elements • Toxic elements, toxicology
Carbon • Nonreactive element • CO, CO-Hb • CO2, HCO3 • HCN
Hydrogen, oxygen • H – highly reactive, H2 • H2O • O, O2, O3 pO2, pCO2 – air, blood • CO, NO • H2O2 • Highly reactive forms of oxygen – anionts and radicals (10 forms): peroxide, superoxide, hydroxyl, peroxyl, perhydroxyl
Nitrogen, sulphur • N2, boiling point -200 C • NH3, NH4, NH2 • N2O, NO, N2O3, NO2, N205 • HNO2 – nitrates, met-Hb • HNO3 – nitrites • H2S, H2SO4 – sulphates, SH
Phosphorus, calcium, magnesium • H3P04, phosphates, bone, buffer, esters • Ca – bone (phosphate, carbonate, fluoride) • Ca – ionized, proteins, citrate, lactate • Oxalate, urate – renal stones • S-Ca 2.15 – 2.50 mmol/l • S-phosphate 0.80 – 1.45 mmol/l • S-Mg 0.65 – 1.1 mmol/l (urine – inh.)
Chromium • Role: metabolism of glucose, chromodulin (intracellular octapeptide), LMWCr factor • Deficiency: impaired glucose tolerance • Toxicity: occupational
Copper • Ceruloplasmin, acerulopl. • Cytochrom c oxidase, lysine 6-oxidase • Ferroxidase I and II, monoamine oxidase • Melanocytes – tyrosinase • Wilson d., Menkes sy., biliary obstr. • S-Cu 11-22 umol/l
Cobalt, fluorine • Component of vitamin B12 • Deficiency – anemia • Prevents tooth decay • Deficiency – increased dental caries
Manganese • Required for glycoprotein and proteoclycan synthesis • Superoxide dismutase • Pyruvate carboxylase • Arginase • Toxicity
Molybdenum • Sulphite oxidase • Xanthine dehydrogenase • Aldehyde oxidase
Selenium • Selenocystein (Se instead of sulphur), 21st aminoacid, UGA, tRNA • Glutathione peroxidases (4x) • Thioredoxin reductase • Selenoprotein P (10 atoms of Se) • Iodothyronine deiodinase (T4 – T3)
Zinc • About 300 zinc metalloenzymes and proteins • Metallothioneins • Carbonic anhydrase, alkaline phosphatase, • RNA and DNA polymerases • Alcohol dehydrogenase, thimidine kinase • Deficiency (AE), toxicity
Iron, Iodine • S-Fe M, F, about 10 – 25 umol/l • Hemoglobin, myoglobin • Cytochroms, Transferrin, Ferritin • Thyroxine, triiodthyronine, free forms • Thyroglobulin x TBG
Other elements • Boron, Nickel, Mercury, Cadmium • Silicon, Beryllium, Aluminium, Baryum • Antimony, Arsenic, Lithium, Vanadium • Lead • Silver, Gold, Platinum • Thallium, Bismuth, Germanium, Titanium
Na, K, Cl • NaCl 0.15 mol/l (0.9 % = 154 mmol/l) • Ionts • Na mmol/l: plasma 136 - 145, ICW 12 • K mmol/l: plasma 3.5 – 5.1, ICW 156 • Cl mmol/l: plasma 98 – 107, ICW 4 • Gastric HCl: 0.1 mol/l
Physiological values • Reference values, mean, 2SD, 5-95 % • Distribution of the data • Biological variability, inter a intra individual • Circadiann rhytms, variation – day, season
Chromium in the blood binds: • A) chromoglobulin • B) chromodulin • C) glucuronide • D) microalbumin
Which of the following proteins incorporates copper into its structure? • A) albumin • B) creatine kinase • C) haptoglobin • D) ceruloplasmin