Binary Distillation
Binary Distillation. Group 5 Alex Guerrero Andrew Duffy Bernard Hsu Daniyal Qamar Jeff Tyska Ryan Kosak Tomi Damo. Introduction. Binary distillation involves only 2 components It works by using the differences in boiling points for separation Methanol BP: 65 °C Water BP: 100°C

Binary Distillation
E N D
Presentation Transcript
Binary Distillation Group 5 Alex Guerrero Andrew Duffy Bernard Hsu Daniyal Qamar Jeff Tyska Ryan Kosak Tomi Damo ChE 382 Group 5
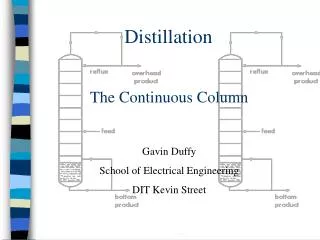
Introduction • Binary distillation involves only 2 components • It works by using the differences in boiling points for separation • Methanol BP: 65°C • Water BP: 100°C • This column uses multiple trays for separation ChE 382 Group 5
Introduction • The column runs under total reflux • Therefore no methanol vapor leaves ChE 382 Group 5
Purpose • The purpose of this experiment is to: • Separate a binary mixture of methanol and water • Learn how to use a refractometer • Create a calibration curve • Use live data from the refractometer to determine the degree of separation throughout the column ChE 382 Group 5
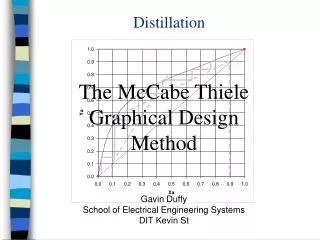
Theory • Works by the difference in component volatilities • Lighter compounds (lower boiling point) will have a higher concentration in the vapor phase • Like wise the heavier compounds will be found in the liquid phase • Relative volatility is the ratio of the volatilities of the lighter component over the volatility of the heavier component ChE 382 Group 5
Theory • Heat is supplied in the reboiler • The condenser at the top uses cold water and runs under total reflux • Total reflux = no operating lines on McCabe-Thiele diagram ChE 382 Group 5
Theory • Murphee tray efficiency • Where: • η = plate efficiency • yn = mole fraction of component in vapor phase at tray n (dimensionless) • yn+1 = mole fraction of component in vapor phase at tray n+1 (dimensionless)yn* = the composition in equilibrium with Xn (dimensionless) ChE 382 Group 5
Apparatus: Column ChE 382 Group 5
Apparatus: Column Details ChE 382 Group 5
Apparatus: Refractometer ChE 382 Group 5
Apparatus: Ice Bath ChE 382 Group 5
Materials ChE 382 Group 5
Procedure Creating the Calibration Curve: • Plug in the Refractometer (23) and ice bath (32). • Obtain ice from the Chemical Engineering office, fill the ice bath about half way with ice, and add water. • Adjust the heating to keep the system at 32°F. • Create samples of methanol and water by mixing solutions of 0-100% (use increments of 10) methanol by volume in small test tubes (cover with para-film until testing). • Place sample under the illumination prism (30), look through the eye piece (24), press the illumination button near the back of the machine then adjust the X to line up with the vertical line using the fine tuning knob (26) and record the value of refraction. • Clean off sample with the provided Kimwipes and repeat for all the samples to create a calibration curve of refraction versus volume percent of methanol. ChE 382 Group 5
Procedure Using the Batch Distillation Column: • Make sure the drain valve for the filling tank (5) is set to the “Fill” setting and the tank is closed. • Measure out 1.6 L of methanol and pour it through the funnel (21) into the filling tank (5) then add 32 L of water to the tank using the filling tank hose just above the funnel to create 0.05% by volume mixture. • Once the tank is full turn the Fill/Drain valve to “Drain” then open the filling tank valve (22) to allow the mixture to fill the round bottom flask (15) of the distillation column. • When the filling tank (5) is empty close the filling valve (22) and call the TA or instructor to turn on the reboiler (16). • Record the temperatures of thermocouples using the digital thermometer (14) every 20 minutes until steady state is met. • Obtain samples of vapor and liquid from each stage using the vapor and liquid release valves (17 and 19). • Use the refractometer (23) to obtain the refraction index of each sample (Use the same procedure as the calibration curve). • Once all the samples are collected and measured turn off the column and allow it to drain. • Unplug and clean the refractometer (23) and ice bath (32). • Dispose of samples accordingly. ChE 382 Group 5
Safety • The reboiler gets hot so avoid contact • The samples taken from the column are very hot • Be careful when using the ladder • Avoid spilling any methanol • Avoid skin contact with methanol • Ensure the area around the column is cleaned of any spilled water • Make sure no methanol vapor leaves the top of the column ChE 382 Group 5
References • “Binary Distillation.”University of Illinois at Chicago - UIC. Web. 13 Sept. 2010. <http://www.uic.edu/depts/chme/UnitOps/entry.html>. • Gilbert, John C., and Stephen F. Martin. Experimental organic chemistry: a miniscale & microscale approach. 4th. BrooksCole Pub Co, 2006. 137. Print. • http://www.separationprocesses.com/Distillation/DT_Chp05.htm • McCabe, Warren L., Julian C. Smith, and Peter Harriott. Unit Operations of Chemical Engineering. New York: McGraw-Hill, 1993. (pp: 578-579) Print. • Stichlmair, Johann. 2000. “Distillation and Rectification.” Encyclopedia of Industrial Chemistry. 7th. 40. New York, NY: Wiley-VCH Verlag GmbH & Co., 2010. (pp: 1-97) Print. • Wankat, Phillip C. Separation Process Engineering. (2nd Edition). Boston, MA: Pearson • Education, Inc., 2007. (pp: 187 – 284) Print ChE 382 Group 5