CH 19: Aldehydes and Ketones
770 likes | 1.08k Views
CH 19: Aldehydes and Ketones. Renee Y. Becker Valencia Community College CHM 2211. Some Generalizations About Carbonyl Compounds. The most important functional group in organic chemistry. Some Generalizations About Carbonyl Compounds.

CH 19: Aldehydes and Ketones
E N D
Presentation Transcript
CH 19: Aldehydes and Ketones Renee Y. Becker Valencia Community College CHM 2211
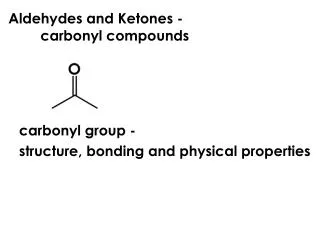
Some Generalizations About Carbonyl Compounds • The most important functional group in organic chemistry.
Some Generalizations About Carbonyl Compounds • carbonyl compounds are planar about the double bond with bond angles 120 due to the sp2 hybridized carbon. • Many types of carbonyl compounds have significant dipole moments. • The polarity of the C-O bond plays a significant role in the reactivity of carbonyl compounds.
Aldehydes and Ketones • Due to the polarity of the carbonyl C-O bond, aldehydes and ketones have higher BPs than alkanes with similar molecular weights. • The lack of H-bonding hydrogens, results in lower BPs than similar alcohols.
Naming Aldehydes • Aldehydes are named by replacing the terminal-e of the corresponding alkane name with –al • The parent chain must contain the CHO group • The CHO carbon is numbered as C1 • If the CHO group is attached to a ring, use the suffix carbaldehyde.
Example 2: Draw • 3-Methylbutanal • 3-Methyl-3-butenal • cis-3-tert-Butylcyclohexanecarbaldehyde
Naming Ketones • Replace the terminal -e of the alkane name with –one • Parent chain is the longest one that contains the ketone group • Numbering begins at the end nearer the carbonyl carbon
Naming Ketones • Ketones with Common Names
Ketones and Aldehydes as Substituents • The R–C=O as a substituent is an acyl group is used with the suffix -yl from the root of the carboxylic acid • CH3CO: acetyl; CHO: formyl; C6H5CO: benzoyl
Ketones and Aldehydes as Substituents • The prefix oxo- is used if other functional groups are present and the doubly bonded oxygen is labeled as a substituent on a parent chain
Example 3: Name 1. 3. 4. 2.
Example 4: Draw • 4-Chloro-2-pentanone • P-bromoacetophenone • 3-ethyl-4-methyl-2-hexanone
Preparation of Aldehydes • Oxidize primary alcohols using pyridinium chlorochromate
Preparation of Aldehydes • Oxidation of alkenes with a vinylic hydrogen
Preparation of Aldehydes • The partial reduction of certain carboxylic acid derivatives. (esters)
Example 5 How would you prepare pentanal from the following: 1. 1-Pentanol • 1-Hexene
Preparing Ketones • Oxidation of secondary alcohols
Preparing Ketones • Oxidation of alkenes if one unsaturated carbon is disubstituted
Preparing Ketones • Friedel-Crafts acylation of aromatic compounds with an acid chloride. Occurs only once!
Preparing Ketones • Hydrations of terminal alkynes • Methyl ketone synthesis • Hg2+ catalyst
Example 6 How would you carry out the following reactions? More than 1 step might be necessary. 1. 3-Hexyne 3-Hexanone 2. Benzene m-Bromoacetophenone 3. Bromobenzene Acetophenone
Reactions of Aldehydes and Ketones • Oxidation reactions • Nucleophilic addition reactions • Conjugate nucleophilic addition reactions
Oxidation of Aldehydes • Jones’ Reagent (preferred) • Preferred over other oxidation reagents due to Room temp. reaction with high yields • Run under acidic conditions (con) • Will react with C=C and any acid sensitive functionality
Oxidation of Aldehydes • Tollen’s reagent • For use with C=C double bonds
Oxidation of Ketones • Ketones are resistant toward oxidation due to the missing hydrogen on the carbonyl carbon • Treatment of ketones with hot KMnO4 will cleave the C-C bond adjacent to the carbonyl group:
Nucleophilic Addition Reactions of Aldehydes and Ketones • Nu- approaches 45° to the plane of C=O and adds to C • A tetrahedral alkoxide ion intermediate is produced
Nucleophiles • Nucleophiles can be negatively charged ( : Nu) or neutral ( : Nu) at the reaction site • The overall charge on the nucleophilic species is not considered
Relative Reactivity of Aldehydes and Ketones • Aldehydes are generally more reactive than ketones in nucleophilic addition reactions • The transition state for addition is less crowded and lower in energy for an aldehyde (a) than for a ketone (b)
Electrophilicity of Aldehydes and Ketones • Aldehyde C=O is more polarized than ketone C=O • As in carbocations, more alkyl groups stabilize + character • Ketone has more alkyl groups, stabilizing the C=O carbon inductively
Reactivity of Aromatic Aldehydes • Aromatic aldehydes are less reactive in nucleophilic addition than straight chain aldehydes • Due to electron-donating resonance effect of aromatic ring • Makes carbonyl group less electrophilic
Nucleophilic Addition of H2O: Hydration • Aldehydes and ketones react with water to yield 1,1-diols (geminal (gem) diols) • Hyrdation is reversible: a gem diol can eliminate water
Relative Energies • Equilibrium generally favors the carbonyl compound over hydrate for steric reasons • Acetone in water is 99.9% ketone form • Exception: simple aldehydes • In water, formaldehyde consists is 99.9% hydrate
Acid & Base-Catalyzed Addition of Water • Addition of water is catalyzed by both acid and base • The base-catalyzed hydration nucleophile is the hydroxide ion, which is a much stronger nucleophile than water • Acid-Catalyzed Addition of Water • Protonation of C=O makes it more electrophilic
Addition of H-Y to C=O • Reaction of C=O with H-Y, where Y is electronegative, gives an addition product (“adduct”) • Formation is readily reversible
Nucleophilic Addition of HCN: Cyanohydrin Formation • Aldehydes and unhindered ketones react with HCN to yield cyanohydrins, RCH(OH)CN
Mechanism of Formation of Cyanohydrins • Addition of HCN is reversible and base-catalyzed, generating nucleophilic cyanide ion, CN • Addition of CN to C=O yields a tetrahedral intermediate, which is then protonated • Equilibrium favors adduct
Uses of Cyanohydrins • Nitriles can be reduced with LiAlH4 to yield primary amines:
Uses of Cyanohydrins • Nitriles can be hydrolyzed with hot aqueous acid to yield carboxylic acids:
Nucleophilic Addition of Grignard Reagents and Hydride Reagents: Alcohol Formation • Treatment of aldehydes or ketones with Grignard reagents yields an alcohol • Nucleophilic addition of the equivalent of a carbon anion, or carbanion. A carbon–magnesium bond is strongly polarized, so a Grignard reagent reacts for all practical purposes as R :MgX +.
Mechanism of Addition of Grignard Reagents • Complexation of C=O by Mg2+, Nucleophilic addition of R :,protonation by dilute acid yields the neutral alcohol • Grignard additions are irreversible because a carbanion is not a leaving group