Quantum Effects
Quantum Effects. Quantum dots are unique class of semiconductor because they are so small, ranging from 2-10 nanometers (10-50 atoms) in diameter. At these small sizes materials behave differently, giving quantum dots unprecedented tunability. Relative size of quantum dots.

Quantum Effects
E N D
Presentation Transcript
Quantum dots are unique class of semiconductor because they are so small, ranging from 2-10 nanometers (10-50 atoms) in diameter. At these small sizes materials behave differently, giving quantum dots unprecedented tunability
Relative size of quantum dots http://www.qdots.com
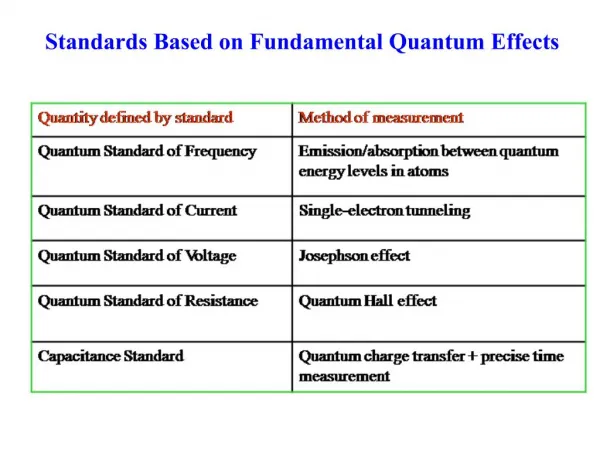
Compound Semiconductors III V
Density of States(how closely packed energy levels are) Quantum confinement
Matter Waves Particle in a Box Analogy de Broglie wavelength
The Schrödinger Equation The Schrödinger equation is an equation for finding a particle’s wave function (x)along the x-axis.
Particle in a box Quantized energy levels are found by solving the Schrödinger equation. Wave function: Allowed Energies:
Quantum Dots - A tunable range of energies Because quantum dots' electron energy levels are discrete rather than continuous, the addition or subtraction of just a few atoms to the quantum dot has the effect of altering the boundaries of the bandgap Changing the length of the box changes the energy levels http://www.kqed.org/quest/television/nanotechnology-takes-off (3:30)
With quantum dots, the size of the bandgap is controlled simply by adjusting the size of the dot http://nanopedia.case.edu/NWPage.php?page=in.jjm8.007
Absorption and Emission J. Lee et al Adv. Materials, 12, 1102 (2000) The figure above charts the absorption and emission with corresponding visible spectrum of light colors based upon nanocrystal (quantum dot) size.
ZnSe CdSe CdTe InAs 460 564 335 388 729 1033 1771 Wavelength (nm) Spectral Codes O. Dabbousi et al, J. Phys. Chem., 101, 9463 (1997). Additionally, the spectral codes of nanocrystals may vary depending on the type of material used. For example, ZnSe emits at the ultraviolet wavelength spectrum; CdSe and CdTe are wavelengths that are visible to the human eye; and InAs is at the infrared spectrum. This figure details the varying spectral codes of the materials which are color coded by semiconductor material listed in the legend.
How Quantum Dots are Made Quantum dots are manufactured in a two step reaction process in a glass flask. Nucleation: This is initiated by heating a solvent to approximately 500 degrees Fahrenheit and injecting precursors such as cadmium and selenium. They chemically decompose and recombine as pure CdSe (cadmium selenide) nanoparticles. Growth: The size of the nanocrystals can be determined based upon varying the length of time of reaction.
How Quantum Dots are Made http://www.youtube.com/watch?v=MLJJkztIWfg http://www.mrsec.wisc.edu/Edetc/nanolab/CdSe/index.html
Self-assembled quantum dots Each dot is about 20 nanometers wide and 8 nanometers in height
Adding Shells to Quantum Dots capping a core quantum dot with a shell (several atomic layers of an inorganic wide band semiconductor) reduces nonradiative recombination and results in brighter emission, provided the shell is of a different semiconductor material with a wider bandgap than the core semiconductor material http://www.youtube.com/watch?v=ohJ0DL2_HGs&feature=related
Quantum Dot Applications LEDs (light emitting diodes); solid state white light, lasers, displays, memory, cell phones, and biological markers. Biological marker applications of quantum dots have been the earliest commercial applications of quantum dots. In these applications, quantum dots are tagged to a variety of nanoscale agents, like DNA, to allow medical researchers to better understand molecular interactions. (The Next Big Thing is Really Small, Jack Uldrich with Deb Newberry, p. 81)
Nanoparticles of cadmium selenide (quantum dots) glow when exposed to ultraviolet light. When injected, functionalized quantum dots can target cancer tumors. The surgeon can see the glowing tumor, and use it as a guide for more accurate tumor removal.
Functionalizing a Quantum Dot The basic parts of a quantum dot include the core, shell, and surface ligand. The shell usually enhances the emission efficiency and stability of the core quantum dot. In functional uses, such as biological applications, a chemical hook is used to target complimentary materials.
Live cell imaging with biodegradable Q dot nanocomposites Antibody-coated QDs within biodegradable polymeric nanospheres. Upon entering the cytosol, the polymer nanospheres undergo hydrolysis and thereby release the QD bioconjugates.
Quantum dots, visible under UV light, have accumulated in tumors of a mouse.
Here, the nucleus is blue, a specific protein within the nucleus is pink, mitochondria look yellow, microtubules are green, and actin filaments are red. QUANTUM DOT CORP., HAYWARD, CA
Quantum Dots in Photovoltaics The quantum dots can be engineered to absorb a specific wavelength of light or to absorb a greater portion of sunlight based on the application.
Quantum Dot Laser 0-D confinement in quantum dots allows for higher efficiencies and brighter lasers because you have better control of photon energies. http://www.youtube.com/watch?v=OaLDF4AJ1hc
Quantum Dot LED http://www.youtube.com/watch?v=SVyC8JW-Q3A&feature=related