1.2 Population inversion
E k. E i. 1.2 Population inversion. Absorption and Emission of radiation. Consider an atom or molecule with two energy levels, E i and E k A direct, radiative transition between these states would be associated with a photon of frequency ν : h ν = Δ E = E k – E i.

1.2 Population inversion
E N D
Presentation Transcript
Ek Ei 1.2 Population inversion Absorption and Emission of radiation Consider an atom or molecule with two energy levels, Ei and Ek A direct, radiative transition between these states would be associated with a photon of frequency ν: hν = ΔE = Ek – Ei Molecule in energy levelEi Consider: What are the possible radiative transitions? What is the probability of a transition taking place? How does the number of photons change with the transition?
Absorption and Emission of radiation Ek hν Ei • Three possible transitions: • “Induced absorption”: • Molecule in Ei absorbs a photon and is excited to Ek One less photon of energy hν Probability of transition is: dPik/dt = Bik ρ(ν) dPik/dtis the probability per second of a molecule absorbing a photon Bik is the Einstein coefficient of induced absorption ρ(ν) is the spectral energy density (the number of photons of frequency ν per unit volume)
Absorption and Emission of radiation Ek hν Ei (b) “Spontaneous emission”: Molecule in Ek decays spontaneously to Ei by emitting a photon in an arbitrary direction One more photon of energy hν (arbitrary phase & direction) Probability of transition is dPki/dt = Aki dPki/dtis the probability per second of the excited molecule emitting a photon Aki is the Einstein coefficient of spontaneous emission (or the spontaneous transition probability) Spontaneous emission is not influenced by the presence of other photons in the medium
Absorption and Emission of radiation Ek hν hν Ei (c) “Induced emission” (or “stimulated emission”): A photon of appropriate frequency induces the transition from Ekto Ei One more photon of energy hν. The new photon has the same frequency, phase, and direction at the original photon Probability of transition is dPki/dt = Bkiρ(ν) dPki/dtis the probability per second of the excited molecule emitting a photon Bki is the Einstein coefficient of induced emission
Absorption and Emission of radiation Relation between Bik and Bki: The Einstein coefficients of induced absorption and emission are directly related through the degeneracy, gx, of each level x: Bik = (gk/gi) Bki In the case where each level has the same degeneracy (gi = gk), the Einstein coefficients of induced absorption and emission are identical – in other words, the probability of induced emission is the same as that of induced absorption How can we make practical use of induced emission?
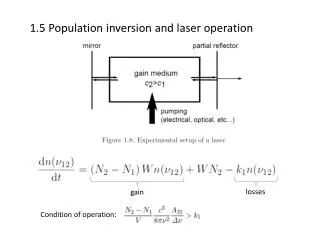
Population inversion Population inversion A system with a population inversion is not in thermal equilibrium; populations of energy levels are not governed by the Boltzmann distribution IF we can alter the population distribution so that more molecules are in higher energy levels rather than lower energy levels, this is called population inversion. With a population inversion, photons passing through the gain medium will be amplified (by induced emission) rather than attenuated (by induced absorption).
Population inversion A resonator or cavity (produced by the two mirrors) is used to achieve selective feedback of some of the cavity modes in the cavity – that is, photons that travel on the axis between the mirrors are preferentially amplified over photons going in different directions:
Threshold condition Threshold condition The probability of stimulated absorption and emission depends on the populations of the upper and lower states. With Ni molecules in level Ei and Nk molecules in level Ek , the intensity after distance z is: I = I0e–α z where I0 is the initial intensity and the absorption coefficient, α, is: Here σ is the absorption cross-section and is related to Bik α = [Ni – (gk/gi) Nk] σ When a population inversion exists (Nk> Ni) the intensity after distance z is greater than the initial intensity (I> I0). However, we also need to consider other photon losses in the cavity
Threshold condition Photons may be lost in the cavity owing to transmission through the mirrors, scattering from surfaces and particles, diffraction losses, and absorption by other materials in the cavity. If all of these losses contribute to a loss coefficient, γ, then the intensity owing to cavity losses after a round trip in the cavity is I = I0e– γ From above, if the cavity has length L, the round trip gain (considering only the population inversion) is then: I = I0e–2αL To compensate for cavity losses, the threshold condition for amplification in the cavity is then: ΔN = [(gk/gi)Nk – Ni] > (γ / 2σL)
Generating a population inversion “Pumping”: delivery of energy to produce a population inversion E4 E3 rapid relaxation E2 rapid relaxation E3 pump E2 pump lasing pump lasing E2 lasing E1 rapid relaxation E1 E1 Two-level system Three-level system Four-level system Notes: A true two-level system cannot produce a population inversion Only E1 is populated at thermal equilibrium (E2 >> E1) A three-level system must be pumped harder than a four-level system – that is, more molecules must be pumped into the excited level to produce lasing Can we sustain a population inversion in a given laser?
Generating a population inversion It is difficult to maintain a population inversion: Lasers that maintain a population inversion indefinitely produce continuous output – termed CW (for continuous wave) lasers Lasers that have a short-lived population inversion produce pulsed output – these are pulsed lasers Pulsed lasers may be of three types: “normal” pulsed lasers, Q-switched lasers, and mode-locked lasers Pumping can be achieved either: 1) optically – e.g., flashlamps (pulsed) or Hg arc lamps (CW operation) 2) electrically – e.g., electric discharge in a gas and in semiconductor lasers
Rate equations for a four-level laser Box 3.1 (Telle)
Spectral characteristics of laser emission The photon emitted between two levels is not perfectly monochromatic. The linewidth is affected inter alia by: Natural lifetime (usually gives narrow linewidth) Molecular motion (Doppler broadening) Collisions (Pressure broadening, solvent effects) This linewidth results in a gain profilefor the laser: Only that part of the gain profile that is above the threshold can lase. The gain profile must be considered together with the cavity modes to determine the laser spectrum