GENUS PASTEURELLA
GENUS PASTEURELLA . Order. Family. Genus. Pasteurella Mannheimia. Pasteurellales. Pasteurellaceae. PASTEURELLA.

GENUS PASTEURELLA
E N D
Presentation Transcript
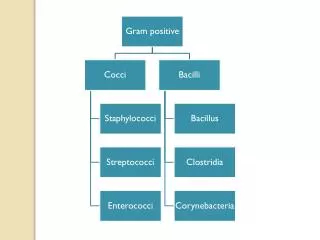
Order Family Genus Pasteurella Mannheimia Pasteurellales Pasteurellaceae
PASTEURELLA Gram- coccobacilli to rod-shaped, bi-polar staining, facultative anaerobe do not form spore glucose fermented (w/o gas) oxidase +, catalase + Habitat mucosal commensals of oropharynx and gastrointestinal tract of healthy mammals, birds and reptile
Classification Polysaccharide capsules A B C D E and F 5 Serogroups 16 somatic types-serology on cell wall LPS type A - fowl cholera, rabbits snuffles - bovine pneumonia type B or E - hemorrhagic septicemia type D - swine atrophic rhinitis type F - recovered primary from turkeys the role is unclear typed by agar gel precipitation indirect hemagglutination of capsular antigen
Transmission Endogenous; * may invade tissue of immunosuppressed animals Exogonous; * direct contact * aerosol Pasteurella multocida
Hemorrhagic septicemia • acute, highly fatal disease of cattle water buffaloes*, American bison caused byP. multocida B:2 (Asia, middle east, some southern European countries), E:2 (Africa) • water buffaloes, most susceptible, sporadically in goats, sheep, swine, deer, camels, and elephants • predisposing - stress, weather change, poor nutrition, overwork, poor body condition
Hemorrhagic septicemia(cont.) • morbidity & mortality 50-100% • clinical sign: high fever, profuse salivation, depression dyspnea, submucosal petichiation subcutaneous edema around the throat dewlap, brisket or perineum, occasional dysentery • death within 24 hr. • gross lesion: generalized petichiation pulmonary edema, enteritis, lymphadenopathy
Bovine and Porcine pneumonia • severe fibrinous bronchopneumonia and pleuropneumonia seen frequently in young weaned animals • opportunistic pathogen of low virulence that invades 2ry to viral or bacterial pneumonias
Fowl Cholera • highly contagious & affects a wide range of domesticated and wild birds in most countries • septicemia withhigh morbidity and mortality (upto 70%), also occur in chronic form • turkey, duck and geese are more susceptible than chicken • mature chickens are more susceptible than birds less than 16 weeks of age
Transmission • contaminated crates, feeders, waterer may introduce FC to new birds • excretion from infected bird directly & indirect • aerosol
acute - septicemia - fever, diarrhea, mucoid oral discharge, ruffled feathers and tachypnea lesion - hemorrhage on serous surface and accumulation of fluid in body cavities - bipolar-staining organism can be detected in blood smear - P. multocida can be isolated from blood, liver and spleen
Atrophic Rhinitis (AR) • upper respiratory disease of piglet 1-8wks of age toxigenic strain of P. multocida type D or A (AR+) cause a severe progressive form of AR • infection with Bordetella bronchiseptica maycause mild, non-progressive turbinate atrophy but predispose to infection with P. multocida (AR+) • other factors include overstocking and poor ventilation
Clinical sign • pig between 3 and 8 wks of age are affected • excessive lacrimation, sneezing and occasionally epitaxis • damage toturbinate bones may predispose to 2ry bacterial infection of the lower respiratory tract • deviation of snout may develop
Rabbits Pasteurellosis “snuffles” Recurring purulent rhinitis • major concern for commercial rabbitries and research facilities • half of conventionally raised rabbit may have upper respiratory tract colonization by P. multocida and many are symptomatic • factors: temperature fluctuation, increased ammonia level, poor sanitation, old age may initiate clinical disease
Clinical sign: • otitis media or interna pneumonia, abscesses, conjunctivitis, mastitis, metritis, septicemia • signs exhibited depend on the site infection include oculonasal discharge, snuffing, dypsnea, vaginal discharge, subcutaneous swelling, and sudden death
Back abscess Normal uterus
PATHOGENESIS • diverse withrespect to host predilection and antigenic variation Fowl cholera : enter tissues through of mucous membrane of pharynx, upper respiratory passages, conjunctivae, rarely through cutaneous wound
attach to the membranes overlining nasal turbinates Atrophic rhinitis dermonecrotic toxin destroy osteoblast osteoclastic lysis turbinate destruction
VIRULENCE FACTOR capsule - hyarulonic acid capsule are more resistant to phagocytosis and intracellular killing by macrophage endotoxin (LPS) -pyrogenic - leukotoxin (bovine) exotoxin adhesin - serogroup A, B, D have type 4 fimbriae associated with adhesin to host epithelial cell surfaces (colonization) Swine strain:hemagglutinin and fimbriae
DIAGNOSIS isolation from affected tissues - ferment of D-mannose differentiates Pasteurella (+) from Mannheimia spp. (-) serologic testfor diagnosis of FC (plate agglutination or agar gel diffusion, precipitation test)have limited value in chronic disease and no value in acute forms of disease
isolation & identification should be followed by tests to confirm toxigenic strain, demonstrates of toxicity for tissue culture cell ELISA for toxin detection PCR for toxin gene
GENUS MANNHEIMIA • established in 1999 to include trehalose-negative members of Pasteurella haemolytica • all strain ferment mannitol but not ferment D-mannose (differentiated from genus Pasteurella)
Hosts and significance Mannheimia spp. Species Host(s) Significance pneumonia pneumonia, septicemia, mastitis M. haemolytica cattle sheep M. granulomatis panniculitis cattle M. glucosida sheep normal respiratory flora M. rumimalis cattle, sheep normal ruminal flora pigs cattle septicemia, enteritis, pneumonia septicemia, pneumonia, mastitis M. varigina
Mannheimia haemolytica 17 serotypes IHA of capsular surface antigen 2 biotypes A,T (ferment L-arabinose or trehalose) T biotype strain (serotype 3, 4, 10 and 15) reclassified as Pasteurella trehalosi serotype A11 - M. glucosida A biotype strain - M. haemolytica
Habitat mucosal commensals of the oropharynx and gastrointestinaltract of healthymammals, birds and reptiles survive poorly outside the host
Mannheima haemolytica • most important pathogen of domestic cattle bovine pneumonic pasteurellosis “ shipping fever ” (transported animals) • serotype A1 – predominant strains (60% pneumonic lung) • multifactorial disease - environment factors - infection with virus & other bacteria
Transmission shipping fever (bovine pneumonia) organism shed from nasal cavity serve as a source of infection of other animals inhalation of bacteria-containing droplets direct nose-to-nose contact ingestion of food contaminated with nasal discharge
PATHOGENESIS impairing host defense – virus, mycoplasma synergy with M. haemolytica proliferation of serotype A1 in nasopharynx reaching lung infect alveolar epithelium pulmonary damage (bacterial product & leukocyte platelet-mediated injury) acute fibrinous pleuropneumonia
VIRULENCE FACTORS Leukotoxin - (1ry virulent) pore-forming cytolysin - impair pulmonary macrophage function LPS - stimulate production of proinflammatory cytokines capsule polysac.- mucosa inhibit phagocytosis resist to complement- mediate lysis neutrophil chemoattractant fimbriae - adherent siderophore - iron acquisition
Hemorrhage and pus in goat
DIAGNOSIS • isolation from clinical specimens Blood agar glucose agar plate + serum colony morphology biochemical test
GENUS RIEMERELLA Rimerella anatipestifer(Pasteurella anatipestifer) Gram- rod grow on enriched media non - motile atmosphere of 5-10% CO2 asaccharolytic dose not grow on MacConkey agar oxidase +, catalase +
important pathogen of ducks infectious serositis new duck disease duck septicemia anatipestifer septicemia
21 serotypes serotype 1, 2, 3, 5 and 15 are most prevalent in severe outbreak of infectious serositis high mortality, decreased growth poor feed conversion, increased condemnation
Disease • acute or chronic septicemia: air sacculitis, fibrinous pericarditis, perihepatitis, caseous salpingitis, meningitis • organism has been recovered from wild birds, and rarely from pigs
mechanism of infection becomes in a flockis unknown, because routes of natural infection have not been definitively established • may be a normalcomponent of the pharyngeal flora of healthy duck • skin puncture, particular feet, are a possible route of infection
disease in turkeys may be transmitted by arthropod vectors • predisposing factors: - adverse environment condition - concomitant bacterial or viral infection
VIRULENCE FACTOR • have not been identified • gene homology to virulence-associated protein of other bacteria • CAMP cohemolysin-maybe an iron-scavenging mechanism
DIAGNOSIS bacterial culture - biochemical testsgelatin arginine hippurate etc bacterin and live vaccine confer protection against homologous serotypes but no cross-protection against heterologous serotype exposure
Ornithobacterium rhinotracheale (ORT) 1990s - 1st characterized with respiratory disease, increased mortality, good retardation in poultry - since been isolated from diseased duck, pigeon, pheasant, quail, many kind of birds *- turkey & chickens throughout the world, phenotypically similar bacteria – Riemerella anapestifer