Chemical Equilibrium
Chemical Equilibrium. The Equilibrium State 01. Chemical Equilibrium: A state achieved when the rates of the forward and reverse reactions are equal and the concentrations of the reactants and products remain constant.

Chemical Equilibrium
E N D
Presentation Transcript
The Equilibrium State 01 • Chemical Equilibrium: A state achieved when the rates of the forward and reverse reactions are equal and the concentrations of the reactants and products remain constant. 1. What is the relationship between the concentrations of reactants and products in an equilibrium mixture? 2. Determine equilibrium concentrations from initial concentrations. 3. How to alter the composition of an equilibrium mixture (concentration, pressure, temp, catalyst).
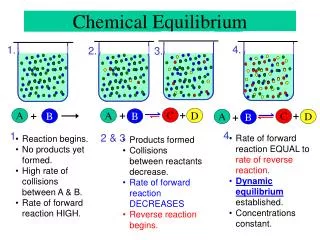
The Equilibrium State 02 • Thus far, we have assumed complete conversion from reactants to products. • Many reactions do not go to completion • Concentrations do not reach constant values because the reaction stops, but because the rates of the forward and reverse reactions become equal.
The Equilibrium State 03 Reversible reactions (arrows in both directions) “reactants” “products”
The Equilibrium State 04 • Graphs of reactant and product concentrations change with time as shown below.
The Equilibrium State 05 Rate of the forward reaction (N2O4 2NO2) decreases as concentration of N2O4 decreases, while the rate of the reverse reaction (N2O4 2NO2) increases as the concentration of NO2increases
Equilibrium Constant Kc 06 What is the relationship between the concentrations of reactants and products in an equilibrium mixture? aA + bB cC + dD Equilibrium Constant: Kc = [C]c[D]d products • [A]a[B]b reactants Kc is independent of concentration changes, but dependent on the temperature.
Equilibrium Constant Kc 07 Kc values are reported without units Thermodynamic state: [ ] = 1M N2O4 2NO2 Equilibrium Constant: Kc = [NO2]2 = (0.0125M/1M)2 • [N2O4] (0.0337M/1M) = 4.64 x 10-3 at 25˚C
Equilibrium Constant Kc’ 08 cC + dD aA + bB Equilibrium Constant: Kc’ = [A]a[B]b products • [C]c[D]d reactants Kc = 1/ Kc’ * important to specify the form of the balanced equation
Example 09 What is the equilibrium equation? (a) N2(g) + 3H2(g) 2NH3(g) (b) 2NH3(g) N2(g) + 3H2(g)
Equilibrium Constant Kp 10 • Equilibrium equations for gas-phase reactions (partial pressures) aA(g) + bB(g) cC(g) + dD(g) Equilibrium Constant: Kp = (PC)c(PD)d products • (PA)a(PB)b reactants
Equilibrium Constant Kp 11 Convert between Kc and Kp using PV = nRT PAV = nART PA = nART = [A]RT V Kp = Kc (RT) ∆n ∆n = moles gas products – moles of gas reactants
2 2 [ ] P NO 2 NO = 2 K = K p c [ ] N O P 2 4 N O 2 4 Equilibrium Constant 12 • Homogeneous Equilibrium: When all reacting species are in the same phase, all reactants and products are included in the expression. • Amounts of components are given as molarity or partial pressure of a gas.
Example 13 • The following pictures represent mixtures of A molecules (red) and B molecules (blue), which interconvert according to the equation A B. If Mixture (1) is at equilibrium, which of the other mixtures is also at equilibrium?
Examples 14 1. Write the Kp and Kc expressions for: • 2 N2O5(g) 4 NO2(g) + O2(g) 2. The equilibrium concentrations for the reaction between CO and Cl2 to form carbonyl chloride (phosgene gas) CO(g) + Cl2(g) COCl2(g) at 74°C are: [CO] = 1.2 x 10–2 M, [Cl2] = 0.054 M, and [COCl2] = 0.14 M. Calculate Kc and Kp.
Example 15 Methane (CH4) reacts with hydrogen sulfide to yield H2 and carbon disulfide, a solvent used in manufacturing. What is the value of Kp at 1000 K if the partial pressures in an equilibrium mixture at 1000 K are 0.20 atm of CH4, 0.25 atm of H2S, 0.52 atm of CS2, and 0.10 atm of H2?
Equilibrium Constant 16 • Heterogeneous Equilibrium:When reacting species are in different phases, solid and liquid phases are excluded from the expression because their concentrations “do not change.” CaCO3(s) CaO(s) + CO2(g) Kc = [CO2] because CaCO3 and CaO are solids.
Write the equilibrium equation for each of the following reactions: (a) CO2(g) + C(s) 2 CO(g) (b) Hg(l) + Hg2+(aq) Hg22+(aq) (c) 2 Fe(s) + 3 H2O(g) Fe2O3(s) + 3 H2(g) (d) 2 H2O(l) 2 H2(g) + O2(g) Examples 18
Using Equilibrium Constants 19 • We can make the following generalizations concerning the composition of equilibrium mixtures: • If Kc > 103, products predominate over reactants. If Kc is very large, the reaction is said to proceed to completion. • If Kc is in the range 10–3 to 103, appreciable concentrations of both reactants and products are present. • If Kc < 10–3, reactants predominate over products. If Kc is very small, the reaction proceeds hardly at all.
Predicting Reaction direction 20 • The reaction quotient (Qc) is obtained by substituting initial concentrations into the equilibrium constant. H2(g) + I2(g) 2HI(g) Qc = [HI]t2 [H2]t[I2]t Qc > Kc System proceeds to form reactants.Qc = Kc System is at equilibrium.Qc < Kc System proceeds to form products.
Example 22 The equilibrium constant (Kc) for the formation of nitrosyl chloride, from nitric oxide and chlorine gas: 2 NO(g) + Cl2(g) 2 NOCl(g) is 6.5 x 104 at 35°C. In an experiment, 2.0 x 10–2 moles of NO, 8.3 x 10–3 moles of Cl2, and 6.8 moles of NOCl are mixed in a 2.0-L flask. In which direction will the system proceed to reach equilibrium?