Re-randomisation in trials
Re-randomisation in trials. Simon Gates 25 April 2007. Contents. Introduction: the problem Inclusion issues Analysis issues Discussion. The problem. Recruitment period of trials can last a long time

Re-randomisation in trials
E N D
Presentation Transcript
Re-randomisation in trials Simon Gates 25 April 2007
Contents • Introduction: the problem • Inclusion issues • Analysis issues • Discussion
The problem • Recruitment period of trials can last a long time • A participant may be randomised and have trial treatment, then re-present with the same problem and be eligible again later. • Randomising again is equivalent to randomising episodes of disease rather than patients.
Examples • Trauma • Pregnancy • Recruitment period of 2 or 3 years is long enough for some women to participate in trial, then get pregnant and become eligible again • Acute asthma • People are eligible when they attend hospital with acute asthma • People will recover within a short period • Some may have attacks several times in a year.
What should we do with these people? • Randomise again or not? • If not, what treatment should they receive? • Should their second episode be included in analyses? • If so, how should they be analysed?
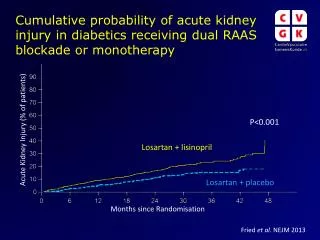
Is this an important question? • Number of participants affected usually small and hence low potential for bias in the results. • Example MAGPIE trial (magnesium sulphate for pre-eclampsia): 15 out of 10141 women randomised twice.
Is this an important question? • Major issue in some fields • Cochrane review “Colony stimulating factors for chemotherapy-induced febrile neutropenia.” • 4/13 trials randomised episodes; contained 25% of episodes and patients in the review. • Re-randomisation stated by methodology consensus statement to be an acceptable design. Immunocompromised Host Society. The design, analysis, and reporting of clinical trials on the empirical antibiotic management of the neutropenic patient. Report of a consensus panel J. Infect Dis 1990, 161(3):397-401.
Problem occurs in many trials. Much time and effort invested by triallists and statisticians in dealing with it (re-inventing the wheel) No agreed best practice. Is this an important question?
Literature • Very little published on this issue • One methodological paper: Hozo I, Djulbegovic B, Clark O, Lyman GH. Use of re-randomized data in meta-analysis. BMC Med Res Methodol. 2005 May 10;5(1):17.
Include or not? • Randomise again and include in analysis as a separate data point • Allocate treatment deterministically (i.e. not by randomisation), and include second participation in analyses • treatment originally allocated • treatment not originally allocated • intervention group • control group
Include or not? • Randomise or allocate deterministically to a treatment option but do not include second participation in analyses • Do not give any of the trial treatments (and do not include second episode in analysis)
Factors affecting decision • Best strategy is likely to vary depending on the nature of the trial • Need to consider what strategy is best under what conditions
Duration of follow-up? • If participants are still being followed up from their first participation, do not want them to receive a different treatment. • Participants would be in both groups simultaneously • Erodes difference between groups • Allocate to original treatment? • Include second episode in analysis or not?
Follow-up • Trial comparing new treatment versus usual care with long-term follow-up. • Likely that a proportion of the new treatment group will receive usual care during follow-up for subsequent episodes (e.g. at hospitals not in the trial). • Triallists may not know about this.
Recovery from first episode • Outcomes of second episodes of disease within a short period may be different. • Treatment received during first episode may affect the outcome of the second.
Patient preferences • Patients who participate once and have a good outcome may be keen to participate again • Is it ethically acceptable to refuse, or to include them and not use the data?
Identifying previous participants • Patients who present for a second time may not be identified as previous participants • May therefore not be possible to allocate them to desired treatment • They may be randomised in belief that it is first presentation
Identifying previous participants • Planning of strategy for re-randomisation must consider practicalities of identifying participants. • Example: patient presents to a different hospital for second episode.
Analysis • Main problem in analysis is that observations from the same individual are not independent • Some individuals may be randomised more than once even if the trial protocol forbids this – so problem is likely to arise in most trials where this is possible
Analytical strategies Ignore • Analyse as if two randomisations were independent • Probably the commonest approach
Analytical strategies Include data from only one randomisation for each patient • Ensures there is no non-independence • Second participation will be ignored • Is it ethical/scientifically acceptable to randomise (or otherwise include) with the intention of ignoring the data?
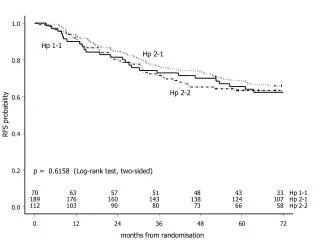
Multiple participations with the same treatment • Equivalent to cluster randomised trials • Standard methods for analysis • Have been used for situations like multiple pregnancies
Multiple participations with different treatments • Analysis not so straightforward • If treatments always different, similar to crossover trial • If a mixture of the same and different, ???