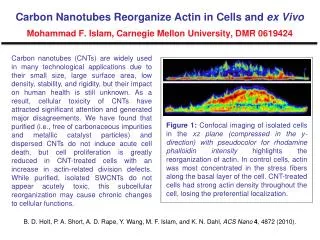
Carbon calculations!!!
Carbon calculations!!!. What’s your usage/day in grams CO 2 ?. Me: . You're pretty low impact - nice work! Water 19,200 GAL Energy 115 MBTU Carbon Dioxide 22,000 LBS Wastewater 19,000 GAL Runoff 121,300 GAL Trash 330 LBS Money 2,890 $. (This has an extra methyl group on the

Carbon calculations!!!
E N D
Presentation Transcript
Carbon calculations!!! What’s your usage/day in grams CO2?
Me: You're pretty low impact - nice work! Water 19,200 GAL Energy 115 MBTU Carbon Dioxide 22,000 LBS Wastewater 19,000 GAL Runoff 121,300 GAL Trash 330 LBS Money 2,890 $
(This has an extra methyl group on the end so it’s C9H13O3N)
Combustion Train for the Determination of the Chemical Composition of Organic Compounds. m 2 m 2 CnHm + (n+ ) O2 = n CO(g) + H2O(g) Fig. 3.4
Ascorbic Acid ( Vitamin C ) - I Contains C , H , and O • Upon combustion in excess oxygen, a 6.49 mg sample yielded 9.74 mg CO2 and 2.64 mg H2O • Calculate its Empirical formula! • C: 9.74 x10-3g CO2 x(12.01 g C/44.01 g CO2) = 2.65 x 10-3 g C • H: 2.64 x10-3g H2O x (2.016 g H2/18.02 gH2O) = 2.92 x 10-4 g H • Mass Oxygen = 6.49 mg - 2.65 mg - 0.30 mg = 3.54 mg O
Vitamin C Combustion - II • C = 2.65 x 10-3 g C / ( 12.01 g C / mol C ) = = 2.21 x 10-4 mol C • H = 0.295 x 10-3 g H / ( 1.008 g H / mol H ) = = 2.92 x 10-4 mol H • O = 3.54 x 10-3 g O / ( 16.00 g O / mol O ) = = 2.21 x 10-4 mol O • Divide each by 2.21 x 10-4 • C = 1.00 Multiply each by 3 = 3.00 = 3.0 • H = 1.32 = 3.96 = 4.0 • O = 1.00= 3.00 = 3.0 C3H4O3
Determining a Chemical Formula from Combustion Analysis - I Problem: Erthrose (M = 120 g/mol) is an important chemical compound as a starting material in chemical synthesis, and contains Carbon Hydrogen, and Oxygen. Combustion analysis of a 700.0 mg sample yielded 1.027 g CO2 and 0.4194 g H2O. Plan: We find the masses of Hydrogen and Carbon using the mass fractions of H in H2O, and C in CO2. The mass of Carbon and Hydrogen are subtracted from the sample mass to get the mass of Oxygen. We then calculate moles, and construct the empirical formula, and from the given molar mass we can calculate the molecular formula.
Determining a Chemical Formula from Combustion Analysis - II Calculating the mass fractions of the elements: Mass fraction of C in CO2 = = = = 0.2729 g C / 1 g CO2 Mass fraction of H in H2O = = = = 0.1119 g H / 1 g H2O Calculating masses of C and H Mass of Element = mass of compound x mass fraction of element mol C xM of C mass of 1 mol CO2 1 mol C x 12.01 g C/ 1 mol C 44.01 g CO2 mol H xM of H mass of 1 mol H2O 2 mol H x 1.008 g H / 1 mol H 18.02 g H2O
Determining a Chemical Formula from Combustion Analysis - III 0.2729 g C 1 g CO2 Mass (g) of C = 1.027 g CO2 x = 0.2803 g C Mass (g) of H = 0.4194 g H2O x = 0.04693 g H Calculating the mass of O: Mass (g) of O = Sample mass -( mass of C + mass of H ) = 0.700 g - 0.2803 g C - 0.04693 g H = 0.37277 g O Calculating moles of each element: C = 0.2803 g C / 12.01 g C/ mol C = 0.02334 mol C H = 0.04693 g H / 1.008 g H / mol H = 0.04656 mol H O = 0.37277 g O / 16.00 g O / mol O = 0.02330 mol O C0.02334H0.04656O0.02330 = CH2O formula weight = 30 g / formula 120 g /mol / 30 g / formula = 4 formula units / cpd = C4H8O4 0.1119 g H 1 g H2O
Some Compounds with Empirical Formula CH2O (Composition by Mass 40.0% C, 6.71% H, 53.3%O) Molecular M Formula (g/mol) Name Use or Function CH2O 30.03 Formaldehyde Disinfectant; Biological preservative C2H4O2 60.05 Acetic acid Acetate polymers; vinegar ( 5% solution) C3H6O3 90.08 Lactic acid Causes milk to sour; forms in muscle during exercise C4H8O4 120.10 Erythrose Forms during sugar metabolism C5H10O5 150.13 Ribose Component of many nucleic acids and vitamin B2 C6H12O6 180.16 Glucose Major nutrient for energy in cells
Two Compounds with Molecular Formula C2H6O Property Ethanol Dimethyl Ether M (g/mol) 46.07 46.07 Color Colorless Colorless Melting point - 117oC - 138.5oC Boiling point 78.5oC - 25oC Density (at 20oC) 0.789 g/mL 0.00195 g/mL Use Intoxicant in In refrigeration alcoholic beverages H H H H H C C O H H C O C H H H H H Table 3.4
Molecular Formula Atoms Molecules Avogadro’s Number 6.022 x 1023 Moles Moles
Chemical Equations Qualitative Information: Reactants Products States of Matter: (s) solid (l) liquid (g) gaseous (aq) aqueous 2 H2 (g) + O2 (g) 2 H2O (g)
Chemical Equation Calculation - I Atoms (Molecules) Avogadro’s Number 6.02 x 1023 Molecules Reactants Products
Chemical Equation Calculation - II Mass Atoms (Molecules) Molecular Weight Avogadro’s Number g/mol 6.02 x 1023 Molecules Reactants Products Moles
Stoichiometry The calculation of the quantities of reactants and products involved in a chemical reaction Interpreting a Chemical Equation The coefficients of the balanced chemical equation may be interpreted in terms of either (1) numbers of molecules (or ions or formula units) or (2) numbers of moles, depending on your needs.
Information Contained in a Balanced Equation Viewed in Reactants Products terms of: 2 C2H6 (g) + 7 O2 (g) = 4 CO2 (g) + 6 H2O(g) + Energy Molecules 2 molecules of C2H6 + 7 molecules of O2 = 4 molecules of CO2 + 6 molecules of H2O Amount (mol) 2 mol C2H6 + 7 mol O2 = 4 mol CO2 + 6 mol H2O Mass (amu) 60.14 amu C2H6 + 224.00 amu O2 = 176.04 amu CO2 + 108.10 amu H2O Mass (g) 60.14 g C2H6 + 224.00 g O2 = 176.04 g CO2 + 108.10 g H2O Total Mass (g) 284.14g = 284.14g
1. Convert grams of A to moles of A Using the molar mass of A 2. Convert moles of A to moles of B Using the coefficients of the balanced chemical equation 3. Convert moles of B to grams of B Using the molar mass of B • To find the amount of B (one reactant or product) given the amount of A (another reactant or product):
Propane, C3H8, is normally a gas, but it is sold as a fuel compressed as a liquid in steel cylinders. The gas burns according to the following equation: C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g) How many grams of CO2 are produced when 20.0 g of propane is burned?
Molar masses C3H8: 3(12.01) + 8(1.008) = 44.094 g CO2: 1(12.01) + 2(16.00) = 44.01 g 59.9 g CO2 (3 significant figures)
Limiting Reactant The reactant that is entirely consumed when a reaction goes to completion Once one reactant has been completely consumed, the reaction stops. Any problem giving the starting amount for more than one reactant is a limiting reactant problem.
All amounts produced and reacted are determined by the limiting reactant. How can we determine the limiting reactant? Use each given amount to calculate the amount of product produced. The limiting reactant will produce the lesser or least amount of product.
Magnesium metal is used to prepare zirconium metal, which is used to make the container for nuclear fuel (the nuclear fuel rods): ZrCl4(g) + 2Mg(s) 2MgCl2(s) + Zr(s) How many moles of zirconium metal can be produced from a reaction mixture containing 0.20 mol ZrCl4 and 0.50 mol Mg?
ZrCl4 is the limiting reactant. 0.20 mol Zr will be produced.
Urea, CH4N2O, is used as a nitrogen fertilizer. It is manufactured from ammonia and carbon dioxide at high pressure and high temperature: 2NH3 + CO2(g) CH4N2O + H2O In a laboratory experiment, 10.0 g NH3 and 10.0 g CO2 were added to a reaction vessel. What is the maximum quantity (in grams) of urea that can be obtained? How many grams of the excess reactant are left at the end of the reactions?
Molar masses NH3 1(14.01) + 3(1.008) = 17.02 g CO2 1(12.01) + 2(16.00) = 44.01 g CH4N2O 1(12.01) + 4(1.008) + 2(14.01) + 1(16.00) = 60.06 g CO2 is the limiting reactant. 13.6 g CH4N2O will be produced.
To find the excess NH3, we find how much NH3 reacted: Now subtract the amount reacted from the starting amount: 10.0 at start -7.73 reacted 2.27 g remains 2.3 g NH3 is left unreacted. (1 decimal place)
Theoretical Yield The maximum amount of product that can be obtained by a reaction from given amounts of reactants. This is a calculated amount.
Actual Yield The amount of product that is actually obtained. This is a measured amount. Percentage Yield
2NH3 + CO2(g) CH4N2O + H2O When 10.0 g NH3 and 10.0 g CO2 are added to a reaction vessel, the limiting reactant is CO2. The theoretical yield is 13.6 of urea. When this reaction was carried out, 9.3 g of urea was obtained. What is the percent yield? Theoretical yield = 13.6 g Actual yield = 9.3 g = 68% yield (2 significant figures)