The Periodic Table of the Elements
180 likes | 319 Views
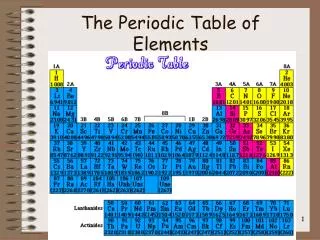
The Periodic Table of the Elements. Pure substances are either compounds or elements. Compounds can be broken down into simpler substances, but elements cannot because they are made of only one kind of atom.

The Periodic Table of the Elements
E N D
Presentation Transcript
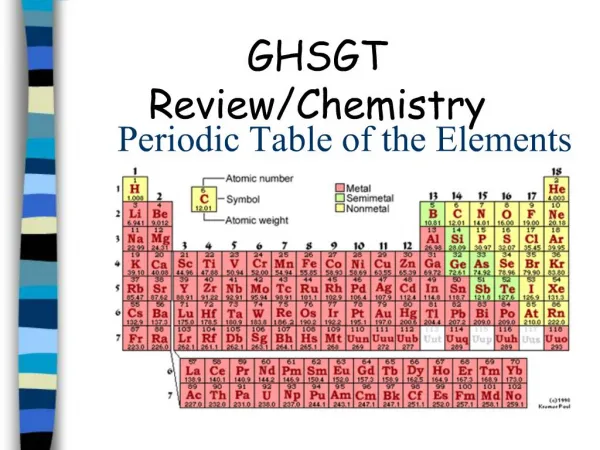
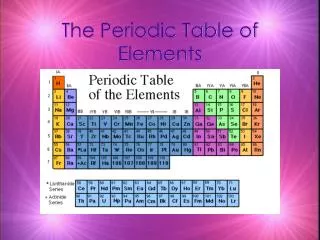
Pure substances are either compounds or elements. Compounds can be broken down into simpler substances, but elements cannot because they are made of only one kind of atom. In the last three centuries, new technologies led scientists to try to identify all of the existing elements and group them into similar categories. Currently, scientists have identified 112 different elements. 92 of them occur naturally, and 20 are man-made. Scientists build super-heavy atoms to test the limits of atomic structure. During the eighteenth and nineteenth centuries, scientists noticed that some elements behaved like other elements. By comparing the behaviors of elements, scientists identified several groups of elements. Other scientists of that time were able to determine the relative atomic masses of different elements. These discoveries led to the development of the Periodic Table of the Elements, which is an important tool of chemists. Classification of Elements
Dmitri Mendeleev is known as the father of the Periodic Table. He was born in Russia in 1834. As were many other scientists of the time, Mendeleev was interested in discovering if a pattern existed relating the known elements. Mendeleev discovered that the physical and chemical properties of the elements occurred in a regular repeating pattern—the Periodic Law. He published his Periodic Table of the Elements in 1869, the first table that successfully summarized the relationships between the elements. In this table, he arranged the known elements in ascending order according to their relative atomic masses, beginning with Hydrogen. Mendeleev also arranged the elements in columns according to their physical and chemical properties. Mendeleev’s table pointed out some errors in accepted atomic weights, and also predicted the existence and properties of elements not yet discovered. Mendeleev’s table did not include any of the noble gases, because none of them had been discovered. The First Periodic Table
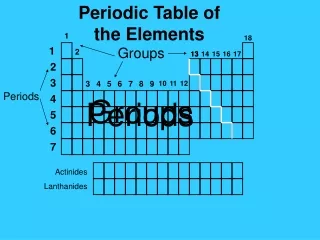
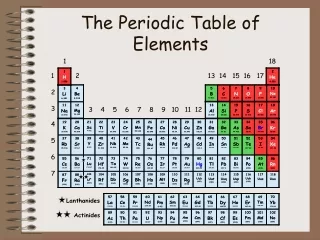
In Mendeleev’s table, there were some elements that appeared to be in the wrong group if arranged strictly by increasing atomic weight. As more elements were discovered after Mendeleev’s death, a few more “misplaced” elements appeared. Some elements, such as were placed in the wrong groups because of multiple valence numbers or errors in the calculation of their atomic weights. In 1914, Henry Moseley discovered the significance of atomic number, the number of protons in the nucleus of an atom. When the Periodic Table was revised, arranging the elements by ascending atomic number, the inconsistencies and errors in the table vanished. The Modern Periodic Table of the Elements has been modified so that the elements are now arranged by atomic number, rather than atomic weight. The Table has seven horizontal rows, called periods, and a number of vertical columns called groups or families. Elements in the same period have the same number of occupied energy levels. Elements in the same group or family have similar properties. Revision of Mendeleev’s Table
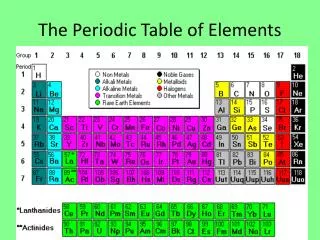
There are 92 naturally occurring and 23 man-made elements listed on the Periodic Table, with space left for future creations. These elements can be divided into three basic types, according to their chemical and physical properties: metals, nonmetals, and metalloids. Most of the elements are metals. Characteristics of metals include: high luster, high malleability, high ductility, good conductivity of heat and electricity, and have high melting and boiling points. Metals tend to loseelectrons in chemical bonding, forming positive ions. Metals are found on the left side of the Periodic Table. Nonmetals are found on the right side of the Periodic Table. Nonmetals are generally dull, brittle, non-malleable, non-ductile, with low melting and boiling points. Many are gases at room temperature. They are non-conductors of heat and electricity. Nonmetals form negative ions because they tend to gain electrons during chemical bonding. Metalloids have some of the properties of metals and some of nonmetals. They are found between metals and nonmetals in the Periodic Table, next to the zigzag line. Metals, Nonmetals, and Metalloids
Allotropes • An allotrope is a different form of the same element – they just have different molecular structures. • Examples: • Diamonds and Graphite – both are forms of carbon molecules. • Silicon – one molecular structure creates a hard, grey substance. Another molecular structure of silicon creates a brown powder.
The Alkali Metals are found in the first group of the Periodic Table. They are a group of elements that are so reactive that they are always found combined with other elements in nature. The reason that these elements are so reactive is that they only have one electron in their outer shell, which they lose when bonding with other elements, to get a full outer shell of electrons. The alkali metals form +1 ions. Alkali Metals are malleable and ductile, good conductors of heat and electricity, but are very soft. The alkali metals include Lithium, Sodium, Potassium, Rubidium, Cesium, and Francium. Alkali metals can explode when in contact with water. Cesium and Francium are the most reactive in this group. Francium is the most reactive of all metals. Although Hydrogen is a nonmetal, it is often listed with this group because it has one electron in its only energy level. Hydrogen is highly reactive but only forms covalent bonds. The Alkali Metals – Group IA H Li Na K Rb Cs Fr
The Alkaline Earth Elements are found in the second group of the Periodic Table. Even though they are not as reactive as the alkali metals, the alkaline earth metals are highly reactive, and are not found in the elemental state in nature. Each of the alkaline earth metals has two electrons in its outer energy level. Alkaline earth metals lose their two valence electrons and form +2 ions when bonding. The alkaline earth elements include Beryllium, Magnesium, Calcium, Strontium, Barium, and Radium. The Alkaline Earth Metals Be Mg Ca Sr Ba Ra
The Boron Family is the third group on the Periodic Table. The members of this family have three electrons in their outer shell. Although Boron is never found uncombined in nature, some of the other members of this family sometimes are. Members of the Boron Family tend to lose three electrons and form +3 ions. Boron is a metalloid, and all other group members are metals. Aluminum is found in this group. It is the most abundant metal on Earth. It is generally found in nature as aluminum oxide. Other elements found in this family are Gallium, Indium, and Thallium. Although Boron is a metalloid, all other elements in this family are metals. The Boron Family B Al Ga In Tl
The Carbon family is the fourth group, and contains one nonmetal, two metalloids, and two metals. All members of the carbon family have four electrons in their outer energy level. Members of this family include Carbon, Silicon, Germanium, Tin, and Lead. Members of this family tend to form covalent bonds. Carbon is the sixth most abundant element in the universe, but not as common on Earth. Carbon exists in several bonding forms such as graphite and diamonds. C-14 is used in radiological dating of some fossils. It can form four covalent bonds with other elements and even other carbon atoms, resulting in an almost infinite number of carbon-containing compounds. Life on earth would be impossible without carbon. Silicon is also found in the Carbon Family. It is the second most abundant element in the Earth’s crust, and is used in solar-electric cells and semi-conductors. Germanium is also used in electronic devices. Tin and lead have a number of uses. The Carbon Family C Si Ge Sn Pb
The Nitrogen Family is the fifth group and contains two nonmetals, two metalloids, and only one metal. They all have five electrons in their outer energy level. They tend to gain three electrons when forming ionic bonds, and form -3 ions. Nitrogen makes up about 4/5 of the Earth’s atmosphere, and is a colorless, odorless gas at room temperature. It is a nonmetal. Nitrogen forms many compounds with oxygen, such as NO2 and N2O. Phosphorus occurs naturally in solid form as red phosphorus or white phosphorus. It is highly reactive and will burst into flame in the presence of air. It is stored under water. Arsenic is a metalloid, and is used in the semiconductor industry and in manufacturing. It is highly poisonous and is used in weed killer and rat poison. Antimony is a hard brittle metalloid used in electronics. It is, like arsenic, highly poisonous. Bismuth is the heaviest naturally occurring element that is not radioactive. It has a variety of uses from Pepto-Bismol to paint pigments, to electrical solder and the heads of fire sprinkler systems. The Nitrogen Family N P As Sb Bi
Members of the Oxygen Family all have six electrons in their outermost energy level. The Oxygen Family is the sixth group on the Periodic Table and contains three nonmetals and two metalloids. There are no metals in the Oxygen Family. Members of the Oxygen Family tend to gain two electrons and form -2 ions, when bonding ionically. Oxygen is the most common element in the Earth’s crust and makes up 1/5 of the Earth’s atmosphere. It is a colorless, odorless gas at room temperature. All elements except the noble gases can form compounds with oxygen. Sulfur is a yellow solid at room temperature. Sulfur is in sulfuric acid and also in acid rain. It is in the gas H2S, which smells like rotten eggs. Selenium is a semiconductor that is sensitive to light, so it is often used in light sensors. Tellurium is a brittle metalloid used in blasting caps. Polonium is radioactive and was discovered by Marie Curie. It has few commercial uses. The Oxygen Family O S Se Te Po
All member s of the Halogen Group have seven electrons in their outer energy level. They are the seventh group on the Periodic Table. This group contains four nonmetals and one metalloid. There are no metals among the halogens. Because they only need one electron to complete their outer energy level, they are the most reactive of the nonmetals. They gain one electron and form -1 ions when bonding ionically. Fluorine is the most reactive nonmetal and is never found alone in nature. It is a poisonous greenish-yellow gas at room temperature. Fluorine compounds are added to many water systems because they prevent tooth decay. Teflon is a fluorocarbon compound. Chlorine is not as reactive as fluorine, but is also a poisonous greenish-yellow gas. One of its most common compounds is NaCl. Bromine is a reddish-brown liquid at room temperature. It is often used to disinfect hot tubs. Iodine is a solid at room temperature that turns to a purple gas when heated. It is needed in your diet to keep your thyroid gland functioning properly. Astatine is a radioactive metalloid, but not much is known about its properties. The Halogens F Cl Br I At
The noble gases are the eighth group on the Periodic Table, and all of the members of this group are unreactive because their outer electron shell is already full. Helium is the second lightest and second must abundant gas in the universe, but is relatively rare on Earth. Helium has two electrons in its only energy level. No compounds of Helium are known. It is used in balloons and blimps. Neon is the best known of the noble gases because of its use in decorative lighting fixtures. It forms no compounds. Argon is used to fill incandescent light bulbs to keep the filament from burning up. It is also used in welding. Krypton is not a green solid that hurts Superman. It is an unreactive gas used in bright strobe lights and airport runway lights. Xenon is a gas that is also used in strobe lights. It can be forced to react with fluorine, but is ordinarily unreactive. Radon is a glowing yellow radioactive noble gas that is considered to be a health hazard if breathed in large amounts. It is used in cancer treatments. The Noble Gases He Ne Ar Kr Xe Rn
The Transition Metals are the elements found on the Periodic Table between the Alkaline Earth Metals and the Boron Family. While the Transition Metals all have no more than 2 electrons in their outer energy level, their next energy level is incompletely filled. The properties of the Transition Metals depend on the electron configuration of the outer two energy levels. Transition elements form alloys easily. A copper-tin alloy is for mirrors and copper-zinc makes brass. Except for copper, the transition metals are all shiny and white, with high melting points and high densities. Many of the most commonly known metals, such as gold, silver, mercury, iron, copper and nickel are in this group. The Transition Metals Sc Ti V Cr Mn Fe Co Ni Cu Zn Y Zr Nb Mo Tc Ru Rb Pd Ag Cd La Hf Ta W Re Os Ir Pt Au Hg Ac Rf Db Sg Bh Hs Mt Uun Uuu Uub
There are 30 Rare Earth Elements. They are all metals, and they are divided into two sets: The Lanthanide Series begins with element # 57, Lanthanum and ends with element # 71 Lutetium. The Actinide Series begins with element # 89 Actinium and ends with element # 103 Lawrencium. One element of the Lanthanide Series and most of the elements of the Actinide series are man-made. Many are radioactive. All of the Rare Earth elements are found in Group 3 of the Periodic Table in the sixth and seventh periods. The Rare Earth Elements Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Lanthanide Series Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr Actinide Series
Each block on the Periodic Table contains valuable information about the element listed. The name of the element is often (but not always) listed. The element’s chemical symbol is always listed. This is either one or two letters. The first is always capitalized and the second (if there is one) is never capitalized. The Atomic Number tells how many protons are in the nucleus of an atom of that element. The Atomic Mass tells the average mass of one atom of that element in atomic mass units. It can be rounded off to determine the element’s mass number. A Representative Block on the Periodic Table 8 O Oxygen 16.000 Atomic Number ChemicalSymbol Name of Element Atomic Mass