ATOMS AND THE PERIODIC TABLE chapter three
390 likes | 948 Views
ATOMS AND THE PERIODIC TABLE chapter three. FOCUS ACTIVITY 1. ATOMIC THEORY - history. NUCLEUS. PROTON +. NEUTRON (NEUTRAL). ATOMIC THEORY - STRUCTURE MODEL OF THE ATOM. ELECTRON -. ENERGY LEVELS. ATOMIC STRUCTURE ELECTRON LOCATION. Electrons are found in energy levels of an atom.

ATOMS AND THE PERIODIC TABLE chapter three
E N D
Presentation Transcript
NUCLEUS PROTON + NEUTRON (NEUTRAL) ATOMIC THEORY - STRUCTURE MODEL OF THE ATOM ELECTRON - ENERGY LEVELS
ATOMIC STRUCTURE ELECTRON LOCATION Electrons are found in energy levels of an atom. Electrons occupy the lowest energy level available. Bohr’s Atom and Energy Levels
Energy Levels are like ladder rungs… GAINING ENERGY GOOD BETTER
ORBITALS – where the _____ are located within an energy level. S orbital (Like a sphere) may contain up to 2 electrons first energy level is an s orbital
(Like a dumbbell) may contain up to 2 electrons second energy level may contain an s orbital and up to 3 p orbitals P orbital
_____ electrons are located in the outermost energy level of an atom. They determine the chemical properties of an element.
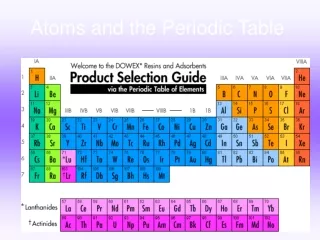
3.2 A TOUR OF THE PERIODIC TABLE • Properties of elements change in a regular pattern that the table helps to describe. • Periods – • Groups(families) - Los Alamos National Laboratory: Periodic Table
Los Alamos National Laboratory's Chemistry Division Presents a Periodic Table of the Elements Traditional Periodic Table
The number of protons in an atom determines an element’s location on the table.Los Alamos National Laboratory: Periodic Table
ATOMIC NUMBER 6 C Carbon 12.001 PROTONS (and ELECTRONS too) SYMBOL NAME PROTONS + NEUTRONS MASS # MASS
PROTONS AND NEUTRONS HAVE EQUAL MASS. ELECTRONS ARE TINY – 1800 EQUAL ONE PROTON. 1 1 1800 1
MASS 35 ATOMIC NUMBER 17 protons _?_ electrons _?_ neutrons _?_ Cl
ELECTRON GAINED = NEGATIVE CHARGE (-) ELECTRON LOST = POSITIVE CHARGE (+) IONS are atoms that have lost or gained an ___.
Electron transfer Na+ Cl
A (+) CHARGED ION IS A ____. A (-) CHARGED ION IS AN ___. EXAMPLES: LITHIUM FLUORIDE TWO KINDS OF IONS:
The atoms of an element always have the same number of protons.BUT…..The atoms of an element may have different numbers of neutrons.This is an ISOTOPE! Two carbon ISOTOPES: CARBON 12 = 6 protons and 6 neutrons CARBON 14 = 6 protons and 8 neutrons
The AVERAGE MASS of an ATOM • Why is the mass number not an even number? • Atoms of the same element exist with different numbers of neutrons. • This makes the mass of different atoms of the same element different. • The average mass is a weighted number so that more common isotopes have a greater affect on the average than rare isotopes. • What is an amu? • It is an “atomic mass unit”. • An amu is equivalent to the mass of 1/12 of a carbon-12 atom.
3.3 FAMILIES OF ELEMENTS: • HAVE THE SAME VALENCE NUMBER. • HAVE SIMILAR CHEMICAL AND PHYSICAL PROPERTIES. • A COLUMN OF ELEMENTS IS A FAMILY.
METALS NONMETALS TWO MAJOR DIVISIONS
VERY REACTIVE ONE VALENCE ELECTRON GROUP ONE:ALKALI METALS
GROUP TWO:ALKALINE EARTH METALS • TWO VALENCE ELECTRONS
GROUP 3-12:TRANSITION METALS • MANY COMMON METALS • NOT AS REACTIVE AS OTHER METALS
GROUP 17:HALOGENS • VERY REACTIVE • FORM SALTS WITH ALKALI METALS • 7 VALANCE ELECTRONS
GROUP 18:NOBLE GASES • INERT / UNREACTIVE • EIGHT VALENCE ELECTRONS
SYNTHETIC ELEMENTS • They are man-made and radioactive. • They include all elements above #92, plus #43 and #61.
B Si Ge As SEMICONDUCTORS (METALOIDS). The elements that are between the metals and nonmetals are known as: SEMICONDUCTORS (METALOIDS) They may exhibit metallic and nonmetallic properties.
3.4 USING MOLES TO COUNT ATOMS • Some counting units: • Reams of paper • Dozens of eggs • Atomic mass units of protons and neutrons • Moles of atoms • A mole is the SI unit that describes the amount of a substance. • Avogadro’s constant is the number of particles in one mole which = 6.022 x1023 • molar mass is the mass in grams of one mol of a substance…it is equal to the average atomic number of an atom.
Chapter 3Studying for the Test • Vocabulary • Parts of an atom • History • Element families • Using the periodic table • Metal vs. nonmetal