25.0 g
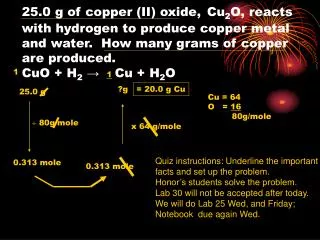
25.0 g of copper (II) oxide, Cu 2 O, reacts with hydrogen to produce copper metal and water. How many grams of copper are produced. CuO + H 2 → Cu + H 2 O. 1. 1. ?g. = 20.0 g Cu. 25.0 g. Cu = 64 O = 16 80g/mole. 80g/mole. x 64 g/mole.

25.0 g
E N D
Presentation Transcript
25.0 g of copper (II) oxide,Cu2O, reacts with hydrogen to produce copper metal and water. How many grams of copper are produced.CuO + H2→ Cu + H2O 1 1 ?g = 20.0 g Cu 25.0 g Cu = 64 O = 16 80g/mole 80g/mole x 64 g/mole Quiz instructions: Underline the important facts and set up the problem. Honor’s students solve the problem. Lab 30 will not be accepted after today. We will do Lab 25 Wed, and Friday; Notebook due again Wed. 0.313 mole 0.313 mole
Ammonia is synthesized from hydrogen and nitrogen according to the following equation: N2 + 3 H2→ 2NH3 If an excess of nitrogen is reacted with 6.82 g of nitrogen, how many grams of ammonia can be produced. N2 + 3 H2→ 2 NH3 2 1 N = 14.0 3H = 3.0 17.0 g/mole ?g = 8.28 g NH3 6.82 g 28.0 g/mole N2 x 17.0g/mole 0.244 mole 0.487 mole
Assume that in the decomposition of potassium chlorate, KClO3, 85 g of O2 form. How many grams of potassium chlorate were used? 2 2 K+Cl- + O2 2 KClO3 3 3 85 g ?g = 520 g KClO3 K =39 Cl = 35 O = 3x16.0 = 48 122g/mole 32 g/mole x 122 g/mole 2.5 mole 4.2 mole
Stoichiometry: Vocabulary positive charges equal negative charges • Balanced Chemical Formula • Balanced Chemical Equation • grams • moles • “known” • “unknown” • gram formula weight (gfw) or gfm • mole ratio All atoms in reactants are present in the same quantity in the products measurement of mass quantity of particles of a substance The “given quantity” The “quantity” you are being asked to find The mass found in one mole of a substance A comparison between the coefficients of the “known” and the “unknown” expressed as a fraction.