Multiple Reactions
Multiple Reactions. 授課教師:林佳璋. Definitions. There are four basic type of multiple reactions: series, parallel, complex, and independent. These types of multiple reactions can occur by themselves, in pair, or all together. When there is a combination of parallel and series reactions, they

Multiple Reactions
E N D
Presentation Transcript
Multiple Reactions 授課教師:林佳璋
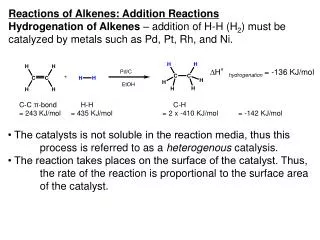
Definitions There are four basic type of multiple reactions: series, parallel, complex, and independent. These types of multiple reactions can occur by themselves, in pair, or all together. When there is a combination of parallel and series reactions, they are often referred to as a complex reactions. Parallel reactions (competing reaction) k1 B A k2 C The reactant is consumed by two different reaction pathways to form different products. Series reactions (consecutive reaction) k1 The reactant forms an intermediate product, where reacts further to form another product. k2 A B C
Complex reactions formation of butadiene from ethanol Involving a combination of both series and parallel reactions Independent reactions cracking of crude oil to form gasoline Occurring at the same time but neither the products nor reactants react with themselves or one another.
Desired and Undesired Reactions Of particular interest are reactants that are consumed in the formation of a desired product, D, and the formation of an undesired product, U, in a competing or side reaction. For a parallel reaction sequence For a series reaction sequence We want to minimize the formation of U and maximize the formation of D because the greater the amount of undesired product formed, the greater the cost of separating the undesired product U from the desired product D (Figure 6-1). As the cost of a reactor system increases in a attempt to minimize U, the cost of separating species U from D decreases.
Selectivity tells us how one product is favored over another when we have multiple reactions. We can quantify the formation of D with respect to U by defining the selectivity and yield of the system. The instantaneous selectivity of D with respective to U is the ratio of the rate formation of D to the rate of formation of U. Overall selectivity for a batch reactor
Example 6-1 Develop a relationship between SD/U and for a CSTR. Solution Consider the instantaneous selectivity for the two parallel reactions just discussed: Overall selectivity For a CSTR the overall and instantaneous selectivities are equal. Carrying out a similar analysis of the series reaction
instantaneous yield overall yield ~Instantaneous yield and overall yield are identical for a CSTR. ~From an economic standpoint, the overall selectivities and yields are important in determining profits. However, the rate-based selectivities give insights in choosing reactors and reaction schemes that will help maximize the profit. ~There often is a conflict between selectivity and conversion (yield) because you want to a lot of your desired product (D) and at the same time minimize the undesired product (U). However, in many instances, the greater conversion you achieve, not only do you make more D, but you also form more U. for a batch system for a flow system
Parallel reactions rate of disappearance of A is instantaneous selectivity is Case 1:1>2 To make this ratio as large as possible, we want to carry out the reaction in a manner that will keep the concentration of reactant A as high as possible during the reaction. A batch or plug flow reactor should be used in this case. CSTR should not be chosen under these circumstances. If the reaction is carried out in the gas phase, we should run it without inerts and at high pressures to keep CA high. If the reaction is in the liquid phase, the use of diluents should be kept to a minimum.
Case 2:2>1 For this ratio rD/rU to be high, the concentration of A should be as low as possible This low concentration may be accomplished by diluting the feed with inerts and running the reactor at low concentrations of A. A CSTR should be used because the concentrations of reactants are maintained at a low level. A recycle reactor in which the product stream acts as a diluent could be used to maintain the entering concentrations of A at a low value.
Because the activation energies of the two reactions in case 1 and 2 are not given, it cannot be determined whether the reaction should be run at high or low temperatures. The sensitivity of the instantaneous selectivity to temperature can be determined from the ratio of the specific reaction rates, A is the frequency factor E is the activation energy Case 4:EU>ED Case 3:ED>EU SD/U SD/U T T The reaction system should be carried out at a low temperature to maximize SD/U. The reaction system should be operated at the highest possible temperature to maximize SD/U.
Example 6-2 Reactant A decomposes by three simultaneous reactions to form three products, one that is desired, B, and two that are undesired, X and Y. These gas-phase reactions, along with the appropriate rate laws, are called the Trambouze reactions. The specific reaction rate are given at 300 K and the activation energies for reactions (1), (2), and (3) are E1=10000 kcal/mol, E2=15000 kcal/mol, and E3=20000 kcal/mol. How and under what conditions (e.g., reactor type(s), temperature, concentrations) should the reaction be carried out to maximize the selectivity of B for an entering concentration of A of 0.4M and a volumetric flow rate of 2.0 dm3/s.
maximum Solution The selectivity with respect to B is CA*=? Because the concentration changes down the length of a PFR, we cannot operate at this maximum. As a result, a CSTR would be used and operated at this maximum. The corresponding selectivity at CA* is
The net of formation of A from reactions (1), (2), and (3) is The mole balance on a CSTR for this liquid-phase reaction (v=v0) is
Maximize the selectivity wrt temperature Run at as high a temperature as possible with existing equipment and watch out for other side reactions that might occur at high temperatures. Case 1: If Run at low temperatures but not so low that a significant conversion is not achieved. Case 2: If For the activation energies given above The selectivity for this combination of activation energies is independent of temperature!
What is the conversion of A in the CSTR? If greater than 72% conversion of A is required, then the CSTR operated with a reactor concentration of 0.112 mol/dm3 should be followed by a PFR because the concentration and hence selectivity will decrease continuously from CA* as we move down the PFR to an exit concentration CAf. Hence the system would give the highest selectivity while forming more of the desired product B, beyond what was formed at CA* in a CSTR.
Optimum CSTR followed by a PFR The exit concentrations of X, Y, and B can be found from the CSTR mole balances
If 90% conversion were required then the exit concentration would be CAf=(1-0.9)/(0.4 mol/dm3)=0.04 mol/dm3. The PFR mole balances for this liquid-phase reaction (v=v0) are At τ=0, the values of the entering concentrations to the PFR are the exit concentrations from the CSTR.
maximizing SD/U The two reactions with recycle shown in (i) and (j) can be used for highly exothermic reactions. Here the recycle stream is cooled and returned to the reactor to dilute and cool the inlet stream thereby avoiding hot spots and runaway reactions. The PFR with recycle is used for gas-phase reactions, and the CSTR is used for liquid-phase reactions. The last two reactors, (k) and (I), are used for thermodynamically limited reactions where the equilibrium lies far to the left (reactant side) A+BC+D and one of the products must be removed (e.g., C) for the reaction to continue to completion. The membrane reactor (k) is used for thermodynamically limited gas-phase reactions, while reactive distillation (I) is used for liquid-phase reactions when one of the products has a higher volatility (e.g., C) than the other species in the reactor.
Example 6-3 For the parallel reactions Consider all possible combinations of reaction orders and select the reaction scheme that will maximize SD/U. Solution Case I:1>2, 1>2 To maximize the ratio rD/rU, maintain the concentration of both A and B as high as possible. To do this, use ~A tubular reactor (Figure 6.3 (b)) ~A batch reactor (Figure 6.3 (c)) ~High pressures (if gas phase), and reduce inerts
Case II:1>2, 1<2 To make SD/U as large as possible, we want to make the concentration of A high and the concentration of B low. To achieve this result, use ~A semibatch reactor in which B is fed slowly into a large amount of A (Figure 6.3(d)) ~A membrane reactor or a tubular reactor with side streams of B continually fed to the reactor (Figure 6.3(f)) ~A series of small CSTRs with A fed only to the first reactor and small amounts of B fed to each reactor. In this way B is mostly consumed before the CSTR exit stream flows into the next reactor (Figure 6.3(h))
Case III:1<2, 1<2 To make SD/U as large as possible, the reaction should be carried out at low concentrations of A and B. To achieve this result, use ~A CSTR (Figure 6.3(a)) ~A tubular reactor in which there is a large recycle ratio (Figure 6.3(i)) ~A feed diluted with inerts ~Low pressure (if gas pressure)
Case IV:1<2, 1>2 To make SD/U as large as possible, run the reaction at high concentration of B and low concentration of A. To achieve this result, use ~A semibatch reactor with A slowly fed to a large amount of B (Figure 6.3(e)) ~A membrane reactor or a tubular reactor with side streams of A (Figure 6.3(g)) ~A series of small CSTRs with fresh A fed to each reactor (Figure 6-3(h))
Maximizing the Desired Product in Series Reactions For series of consecutive reactions, the most important variable is time: space-time for a flow reactor and real time for a batch reactor. To illustrate the important of the time factor, we consider the sequence B is the desired product ~If the first reaction is slow and the second reaction is fast, it will be extremely difficult to produce species B. ~If the first reaction (formation of B) is fast and the reaction to form C is slow, a large yield of B can be achieved. ~However, if the reaction is allowed to proceed for a long time in a batch reactor, or if the tubular flow reactor is too long, the desired product B will be converted to the undesired product C.
Example 6-4 The oxidation of ethanol to form acetaldehyde is carried out on a catalyst of 4 wt % Cu-2 wt % Cr on Al2O3. Unfortunately, acetaldehyde is also oxidized on this catalyst to form carbon dioxide. The reaction is carried out in a threefold excess of oxygen and in dilute concentration (ca. 0.1% ethanol, 1% O2, and 98.9% N2). Consequently, the volume change with the reaction can be neglected. Determine the concentration of acetaldehyde as a function of space-time, The reactions are irreversible and first order in ethanol and acetaldehyde, respectively. Solution O2 in excess
Mole balance on A: Mole balance on B: integrating factor
Mole balance on C: Optimum yield
The yield has been defined as shown as a function of conversion in Figure E6-4.2 Another technique is often used to follow the progress for two reactions in series. The concentrations of A, B, and C are plotted as a singular point at different space time (e.g., 1’, 2’) on a triangular diagram (see Figure 6.-4). The vertices correspond to pure A, B, and C. For (k1/k2)>>1, a large quantity of B can be obtained. For (k1/k2)<<1, very little B can be obtained.
Algorithm for Solution of Complex Reactions ~In complex reactions systems consisting of combination of parallel and series reactions, the availability of software packing (ODE solvers) makes it much easier to solve problems using moles Nj or molar flow rates Fj rather than conversion. ~For liquid systems, concentration is usually the preferred variable used in the mole balance equations. The resulting coupled differential mole balance equations can be easily solved using an ODE solver. ~For gas systems, the molar flow rates are usually the preferred variable in the mole balance equation.
Mole Balances Table 6-1 gives the forms of the mole balance equation we shall use for complex reactions where rA and rB are the net rates of formation of A and B.
We find the net rate of reaction for each species in terms of the concentration of the reacting species in order to combine them with their respective mole balances.
Net Rates of Reaction Consider the following reactions
Example 6-5 Consider the following set of reactions: Write the rate law for each species in each reaction and then write the net rates of formation of NO, O2, and N2. Solution Net Rates of Reaction
Net rate NO Net rate N2 Net rate O2
Stoichiometry: Concentration for liquid-phase reactions, v=v0 for gas-phase reactions T=T0, P=P0 where fn represents the functional dependence on concentration of the net rate of formation
Multiple Reactions in a PFR/PBR We now insert rate laws written in terms of molar flow rates into the mole balances. After performing this operation for each species, we arrive at a coupled set of first-order ordinary differential equations to be solved for the molar flow rates as a function of reactor volume. In liquid-phase reactions, incorporating and solving for total molar flow rate is not necessary at each step along the solution pathway because there is no volume change with reaction. for gas-phase reactions Combing mole balance, rate laws, and stoichiometry T=T0, P=P0 solved simultaneously with a numerical package or by writing an ODE solver. V=?
Example 6-6 Consider again the reaction in Example 6-5. Write the mole balances on a PFR in terms of molar flow rates for each species. Solution T=T0, P=P0 total molar flow rate of all the gases is
mole balance on NO mole balance on NH3 mole balance on H2O
mole balance on N2 mole balance on O2 mole balance on NO2
Table 6-2 shows the equation for species j and reaction i that are to be combined when we have q reactions and n species.
Example 6-7 The production of m-xylene by the hydrodealkylation of mesitylene over a Houdry Detrol catalyst involves the following reactions: m-xylene can also undergo hydrodealkylation to form toluene: The second reaction is undesirable, because m-xylene sells for a higher price than toluene ($1.32/lb vs. $0.30/lb). Thus we see that there is a significant incentive to maximize the production of m-xylene. The hydrodealkylation of mesitylene is to be carried out isothermally at 1500R and 35 atm in a packed-bed reactor in which the feed is 66.7 mol% hydrogen and 33.3 mol% mesitylene. The volumetric feed rate is 476 ft3/h and the reactor volume (i.e., V=W/b) is 238 ft3.
The rate laws for reactions 1 and 2 are, respectively where the subscripts are: M=mesitylene, X=m-xylene, T=toluene, Me=methane, and H=hydrogen (H2). At 1500R, the specific reaction rates are The bulk density of the catalyst has been included in the specific reaction rate (i.e, k1=k1’b). Plot the concentration of hydrogen, mesitylene, and xylene as a function of space time. Calculate the space time where the production of xylene is a maximum (i.e., opt)
Solution 1.Mole balances: 2.Rate laws and net rates:
3.Stoichiometry: T=T0, P=P0
4.Combining: If we know CM, CH, and CX, then CMe, and CT can be calculated from the reaction stoichiometry. 5.Parameter evaluation:
Multiple Reactions in a CSTR ~If there is no volume change with reaction, we use concentration, Cj, as variables. ~If the reactions are gas phase and there is volume change, we use molar flow rates, Fj, as variables. The total molar flow for n species is
Example 6-8 For the multiple reactions and conditions described in Example 6-7, calculate the conversion of hydrogen and mesitylene along with the exiting concentrations of mesitylene, hydrogen, and xylene in a CSTR. Solution