Electrochemistry
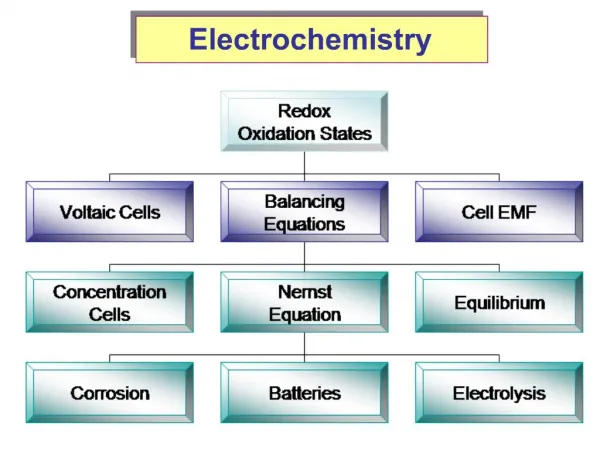
This comprehensive guide explores the fundamentals of electrochemistry, focusing on redox reactions, specifically the balanced equation for the reaction between permanganate (MnO4-) and oxalate (C2O42-) in acidic solution. It covers the determination of oxidation numbers, half-reaction separation, mass balance, and charge balancing using electrons. Additional sections detail the operation of galvanic cells, the significance of standard reduction potentials, and methods to prevent corrosion. The text also highlights electrolysis, battery function, and the principles of electroplating, providing a solid foundation for understanding electrochemical processes.

Electrochemistry
E N D
Presentation Transcript
Redox reactions • Write the balanced equation for the reaction of permanganate and oxalate in acidic solution. • MnO4- + C2O42- Mn2+ + CO2 • 1. Find oxidation numbers
Permanganate & oxalate • Separate into half reactions • Balance for mass, balance O with H2O, balance H with H+, balance charge w/ electrons
Finishing the problem • Balancing the electrons • Combine and cancel
Redox titration • Use the ratio from the balanced redox reaction to determine concentrations. • How will we know when the reaction is complete? • We use an indicator • In our earlier redox reaction, permanganate is a “self indicator”.
Activity series • Some substances are more easily oxidized than others. • Listing these in order of their ability to oxidize gives us an activity series. • A substance will oxidize if its above the item reducing it on the table. • Cu will be oxidized by Ag+ because its higher on the activity series. Ag cannot be oxidized by Cu2+.
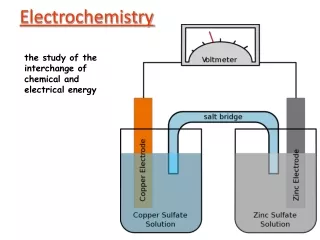
Galvanic Cell • Using a spontaneous redox reaction to generate electricity. • Anode = oxidation (AnOx) • Reduction at the cathode (RedCat) • Electrons flow From Anode To Cathode (FatCat)
Salt Bridge • Allows ions to travel between solutions, maintaining charge balance. • Can be a solution, a paste or a porous membrane
Galvanic cell notation • A shorthand way of writing the structure of a galvanic cell is • Anode | Anode ion || Cathode Ion | Cathode • | denotes a phase barrier (solid metal and aqueous solution) • || denotes the salt bridge • Mg(s) | Mg2+(aq) || Al3+(aq) | Al(s) is the cell notation of an aluminum/magnesium cell
Cell potential • The Standard Reduction Potential table measures the voltage produced when that half reaction is reduced by the standard hydrogen electrode • A negative voltage means that electricity must be applied to cause the reaction (more later) • All concentrations are assumed 1M, pressures are 1 atm at 298K. • How does this compare to the activity series?
Cell potential of a galvanic cell • Ecell = Ered + Eox • When you flip the oxidation half reaction, change the sign of the voltage!!! • What is the cell potential of a copper/zinc cell?
Still more gibbs (and some equilibrium) • ΔG° = -nFE° • n is the number of moles of electrons transferred (from balanced equation) • F is the Faraday constant (~96,500 coulomb/mol) • E° is the standard cell potential
Practice • Calculate ΔG° and K from the following reaction: • 4Ag(s) + O2(g) + 4H+(aq) 4Ag+(aq) + 2H2O(l)
cell potential at nonstandard conditions • Gibbs at Nonstandard Conditions • ΔG = ΔG° + RT lnQ • Substituting –nFE into the equation gives us the Nernst Equation: At 298K, we can rewrite it as
Practical use of nonstandard conditions • Concentration cell • Same anode and cathode • Same electrolyte • E° of the cell is 0. • Due to the huge differences in concentration, Q will not = 1. This gives a small voltage.
concentration cell • What voltage will be generated by the cell on the right?
hydrogen Fuel Cells • Hydrogen is oxidized to form H+ and e-. • The electron passes through a wire and performs work. • H+ travels through the membrane, combines with O2 and the electron to form H2O.
Corrosion • Most metals allow free flow of electrons. • In the presence of H+, Fe will oxidize to Fe2+. • Water acts as both a source of H+ and the electrolyte for Fe2+ ions to flow. • Further oxidation of Fe2+ and combination with oxygen form Fe2O3.
Preventing corrosion • Galvanization • Coating a corrodible metal will prevent corrosion. • Cathodic Protection • Attaching a metal that is easier to oxidize will prevent corrosion. • Ships attach large pieces of magnesium to their hull. The magnesium will oxidize in place of the iron.
Electrolysis • Adding electricity to a nonspontaneous reaction will cause it to occur. • We can generate sodium metal and chlorine gas from a molten NaCl sample.
predicting electrolytic half reactions • Use your reduction potential table. The combination that produces the most positive voltage will occur. • Beware of aqueous solutions • Water can be oxidized to form O2, or reduced to form H2. You must keep these in mind when figuring out what reactions occur.
molten vs aqueous • Molten NaI will form metallic sodium and iodine. Will NaI(aq) do the same? • There are four possible reactions: • Oxidation • 2I- I2 + 2e- E = -0.54V • 2H2O O2 + 4H+ + 4e- E = -1.23V • Reduction • Na+ + e- Na E = -2.71V • 2H2O + 2e- H2 + 2OH- E = -0.83V • Let’s prove it!
electroplating • If we have a metallic anode and an electrolyte solution of that metal, we can coat an object (serving as the cathode) with that metal.
electroplating stoichiometry • We know that: • 1 mol e- = 96,500 coulombs (Faraday’s Constant) • 1 Amp = 1 coulomb/sec • Electroplating questions are simple factor-label!
Example • Calculate the number of grams of aluminum produced in 1.00 hour by the electrolysis of molten AlCl3 at a current of 10.0 A. • Al3+ + 3e- Al