CHEMICAL EQUILIBRIUM
CHEMICAL EQUILIBRIUM. Dr. Saleha Shamsudin. What is chemical equilibrium?. A dynamic state in which the rates of forward (f) and reverse (r) reaction are identical. Chemical Equilibrium – A Dynamic Equilibrium.

CHEMICAL EQUILIBRIUM
E N D
Presentation Transcript
CHEMICAL EQUILIBRIUM Dr. Saleha Shamsudin
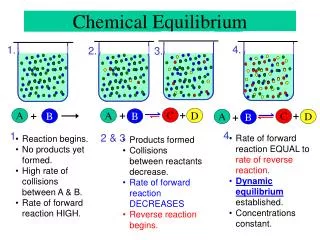
What is chemical equilibrium? • A dynamic state in which the rates of forward (f) and reverse (r) reaction are identical.
Chemical Equilibrium – A Dynamic Equilibrium • Upon addition of reactants and/or products, they react until a constant amount of reactants and products are present = equilibrium. • Equilibrium is dynamic since product is constantly made (forward reaction), but at the same rate it is consumed (reverse reaction).
1. Chemical Reactions 1.1 Introduction • Chemical reactions Involves chemical change. Chemical change: the atoms rearrange themselves in such a way that new substances are formed. Components decrease in quantity are called reactants and those increase in quantity are called products. The chemical reaction is: aA + bB ↔ cC + dD
The rate of reaction = constant x concentration each species raised to the power of the number of molecules participating in the reaction. Forward reaction: kf= rate constant [A] and [B]= molar concentrations of A and B Forward and reverse rates are equal kf[A]a[B]b = kb[C]c[D]d
Molar equilibrium constant, K, kb[C]c[D]d = kf = K [A]a[B]b kb • Rearranging these equation gives molar equilibrium constant (K) : [C]c[D]d = kf = K [A]a[B]b kb When the two rates become equal, the system is in a state of equilibrium. Forward and reverse rates are equal kf[A]a[B]b = kb[C]c[D]d
Type of equilibria • We can write equilibrium constants for many types of chemical processes. The equilibria may represent : • Dissociation (acid/base, solubility) • Formation of products (complexes) • Reaction (redox) • Distribution between 2 phases (water and non-aqueous solvent)
Equilibrium constant of reverse reaction: aA + bB cC + dD cC + dD aA + bB
Equilibrium constants for dissociating and associating species • For weak electrolyte • Slightly soluble substances • Example: 1) AB A + B • Equilibrium constant written as: [A][B] = Keq [AB]
Stepwise dissociation A2B A + AB K1 = [A][AB] [A2B] AB A + B K2 = [A][B] [AB] Overall dissociation: A2B 2A + B Keq = [A]2[B] [A2B]
3) Heterogeneous equilibria • Involving more than one physial phase, example, solids, water and pure liquids. • Concentrations of any solid reactants and products are omitted from the equi. cons. expression. • Molar concentrations of water (or of any liquid reactant or product) is omitted from the equi. cons. expression.
E.g. Write out the equilibrium expression for K using the reaction below: N2(g) + 3H2(g) 2NH3(g) Kc = ?
Calculating Equilibrium Constants Try this example: An aqueous solution of ethanol and acetic acid, each with a concentration of 0.810 M, is heated to 100oC. At equilibrium, the acetic acid concentration is 0.748 M. Calculate K at 100oC for the reaction C2H5OH(aq) + CH3CO2H(aq) CH3CO2C2H3(aq) + H2O(l) ethanol acetic acid ethyl acetate • Write the equilibrium constant expressions in terms of concentrations: • Calculate K at 100oC for the reaction:
The equilibrium constant expressions is: • K = [CH3CO2C2H3] notice that liquid • [C2H5OH][CH3CO2H] water does not • 2. The calculation of K is: appear in equi. • = 0.062 = 0.11 expressions. • (0.748)(0.748)
Calculating an Equilibrium from an Equilibrium Constant Problem: K= 55.64 for H2(g) + I2(g) 2HI(g) Has been determined at 425oC. If 1.00 mol Each of H2 and I2 are placed in 0.5 L flask at 425oC, what are the concentrations of H2, I2 and HI when equilibrium has been achieved?
Solution: • Write the equi. Cons. Expression. • Set up an ICE table • Assume x is equal to the quantity of H2 or I2 • consumed in the reaction
Substitute the equi. Con. Into K expression. 55.64 = (2x)2 = (2x)2 (2.0 – x)(2.0 – x) (2.0 – x)2 55.64 = 7.459 = 2x 2.0 – x x = 1.58 H = I = 2.0 – x = 0.42 M HI = 2x = 3.16 M
Calculating Equilibrium Concentrations • Using initial concentrations, stoichiometry and Kc, equilibrium concentrations of all components can be determined. E.g.2 [H2]o = [I2]o = 24 mM, were mixed and heated to 490oC in a container. Calculate equil. composition. Given that Kc = 46. Solution: set up an equilibrium table and solve for unknown after substitution into equilibrium expression. H2(g) +I2(g)2HI(g)
Equilibrium concentrations of each component are in last row. Substitute into equilibrium expression and solve for x. • It may be necessary to rearrange so that quadratic equation can be used. • . • Rearrange to ax2 + bx + c = 0; determine a, b, c and substitute into quadratic equation: