Cycles in Ecosystems
410 likes | 675 Views
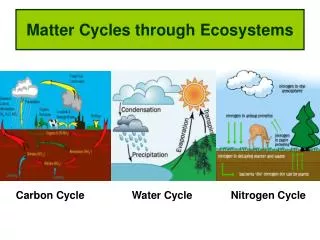
Cycles in Ecosystems. The Water Cycle. Water never leaves the Earth. It is constantly being cycled through the atmosphere, ocean, and land . This process, known as the water cycle , is driven by energy from the sun. The water cycle is crucial to the existence of life on our planet.

Cycles in Ecosystems
E N D
Presentation Transcript
Water never leaves the Earth. It is constantly being cycled through the atmosphere, ocean, and land. • This process, known as the water cycle, is driven by energy from the sun. The water cycle is crucial to the existence of life on our planet.
During part of the water cycle, the sun heats up liquid water and changes it to a gas by the process of evaporation. Water that evaporates from Earth’s oceans, lakes, rivers, and moist soil rises up into the atmosphere.
The process of evaporation from plants is called transpiration. (In other words, it’s like plants sweating.)
As water (in the form of gas) rises higher in the atmosphere, it starts to cool and become a liquid again. This process is called condensation. When a large amount of water vapor condenses, it results in the formation of clouds.
When the water in the clouds gets too heavy, the water falls back to the earth. This is called precipitation.
When rain falls on the land, some of the water is absorbed into the ground forming pockets of water called groundwater. Most groundwater eventually returns to the ocean. Other precipitation runs directly into streams or rivers. Water that collects in rivers, streams, and oceans is called runoff.
Questions: • How does the water cycle (hydrosphere) interact with the other three spheres? Use terms from the cycle • How does human activity impact/interact with the water cycle? • Give at least 3 specific examples. • Is the activity considered a positive or negative impact on the environment?
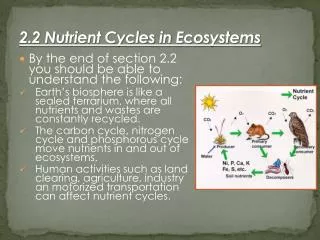
Nutrients • Chemicals that are required for plant and animal growth and other life processes • Continuous flows of nutrients in and out of stores = nutrient cycles
Carbon • The most important element in living organisms, found in: • Carbohydrates – CHO • Proteins – CHON • Lipids (fats) – CHO (long chains) • All steriods are from “fats”, waxes
Carbon Cycle: Chemical Processes The 2 most important chemical processes on earth to sustain life are: • 1. Photosynthesis • Chemical reaction that converts solar energy into chemical energy • Takes in sunlight (energy), CO2 and water – and produces carbs (sugars) and oxygen • Basic Formula: Energy + 6CO2 + 6H2O → C6H12O6 + 6O2
2. Cellular Respiration • Plants and animals release CO2 back into the atmosphere by converting carbs and oxygen into carbon dioxide and water • Basic Formula: C6H12O6 + 6O2 → Energy + 6CO2 + 6H2O Other cycling: • Ocean Mixing • Moves carbon throughout the world’s oceans and pumps more carbon into the oceans, then released back to atmosphere
Carbon • Carbon reservoirs: oceans, forests and fossil fuels release carbon slowly into the environment • Carbon sink: Reservoirs that absorb more carbon than they release. • Examples: Trees, biomass, and oceans (some dissolved CO2 reacts with salt water to form calcium carbonate, which makes shells) • Fossil Fuels: Formed when photosynthetic organisms die and compress to form oil, gas, and coal • They are a reservoir for carbon until they burn, when carbon (CO2) & energy are released. This means less “stored” carbon.
There is cycling of carbon through ecosystems. Photosynthesis and cellular respiration complement each other. Carbon is also cycled through: • Digestion • Volcanic Activity • Erosion • Combustion
Carbon is involved in the Greenhouse effect: Greenhouse Gases • When the two greenhouse gases (carbon dioxide (CO2) and methane (CH4)) trap the heat from the sun in the atmosphere there is a greenhouse effect. • Note: the earth NEEDS these gases to hold in the heat; however, too much may cause a change in climate (global warming)
Questions: Carbon Cycle • 1. Where is carbon stored on Earth? • 2. Where is the largest stores of carbon? • 3. Why is photosynthesis and cellular respiration important to the cycle? • 4. What is the impact of deforestation on the carbon cycle? • 5. What is the importance of decomposition on the carbon cycle? • 6. How has burning fossil fuels changed the natural carbon cycle? • 7. What other human activities have an impact on the carbon cycle?
The Nitrogen Cycle • All living organisms also require the element Nitrogen • DNA • RNA • Proteins
Nitrogen Cycle • Nitrogen Fixation • Process in which Nitrogen is converted into compounds that contain nitrate or ammonium (used by plants) • Nitrogen gas (N2) is not very useful as a molecule, it must be combined or fixed with hydrogen (or other elements) to form ammonia (NH3) so that it can be used by plants for growth. This process is called nitrogen fixation. • Certain bacteria can fix nitrogen • Some nitrogen fixing bacteria are also found in the roots of certain plants called legumes • Lightning can also fix nitrogen in smaller quantities.
Occurs in 3 ways: • Atmosphere • Nitrogen is converted into nitrate and other nitrogen-containing compounds by lightning • Nitrate and other nitrogen –containing compounds enter ecosystems in rain • Soil • Nitrogen is converted into ammonium by bacteria during decomposition (Nitrogen fixing bacteria) • Water bodies • Cyanobacteria in water fix nitrogen into ammonium
Nitrification • Excess ammonia formed from nitrogen fixation dissolves in water to form ammonium ions (NH4+) • Nitrification is the process that changes ammonium (NH4+) to nitrite (NO2-)to nitrates (NO3-) which is taken up by the plant’s roots • Animals obtain their nitrogen by consuming plants or other consumers • Fertilizers add nitrates to soil
Denitrification is the process where ammonia and nitrate is converted back to N2 gas.
Questions: Nitrogen Cycle • 1. Where does nitrogen fixation take place? • 2. How are nitrogen fixation and nitrification related? • 3. How are nitrification and denitrification related? • 5. What human activities contribute excess nitrogen to ecosystems?
The Phosphorus Cycle • Another critical element for life is Phosphorus (P) found as: • PO4 in DNA backbone • P in energy molecules (ATP) • Cell membranes (with lipids - phospholipids) • Shells, bones, teeth of animals
The Phosphorus Cycle • Essential for a variety of life processes in plants and animals • Trapped in phosphate and makes up phosphate rock and sediments of ocean floors
The Phosphorus Cycle • Not found in gaseous form, so it must be cycled through ecosystems • All phosphorus originates from rock in the earth’s crust as phosphate (PO43) • The phosphates dissolve in water and are absorbed by plants then animals then decomposers.
Weathering • Breaking down rock into smaller fragments – releases phosphate into the soil • Chemical Weathering: Chemicals cause phosphate rocks to break down and release phosphate into the soil • Physical/MachanicalWeathering: wind, rain, and freezing release phosphate into soil
Questions: Phosphorus Cycle • 1. How is phosphorus stored in ecosystems? • 2. What is weathering? • 3. How does phosphorus enter ecosystems? • 4. What human activities contribute excess phosphate to ecosystems?