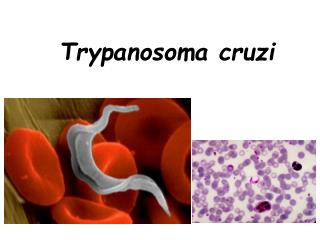
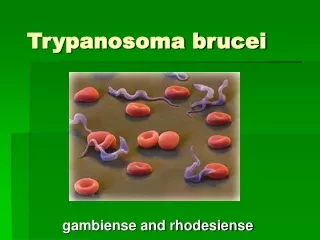
Trypanosoma brucei
Trypanosoma brucei. Dr.T.V.Rao MD. Phylum Sub-Phylum Class Order Genus. Mastigophora Sarcomastigophora Zoomastigophora Kinetoplastidae Trypanosoma. Taxonomy. ? species of mammals, birds, reptiles and amphibians. Trypanosoma brucei.

Trypanosoma brucei
E N D
Presentation Transcript
Trypanosoma brucei Dr.T.V.Rao MD Dr.T.V.Rao MD
Phylum Sub-Phylum Class Order Genus Mastigophora Sarcomastigophora Zoomastigophora Kinetoplastidae Trypanosoma Taxonomy ? species of mammals, birds, reptiles and amphibians
Trypanosoma brucei • Parasitic eukaryotes that diverged 200-500 million years ago. • Pathogens of the African Sleeping Sickness. • Transfer from the gut of the Tsetse fly to the bloodstream of humans and cattle. • Unique biology: - Kinetoplast - Antigenic variation - trans-splicing S.C. IAEA From Mark Field’s lab website Dr.T.V.Rao MD
Trypanosomiasis • Trypanosomes were first described in frogs 1855 • Griffith Evans identifies T. evansi as agent of surra (a horse and camel disease) in 1880 • David Bruce identifies T. brucei as cause of Nagana and demonstrates transmission by Tse-tse flies David Bruce, 1855-1931 Dr.T.V.Rao MD
Sleeping Sickness – as per who • African Trypanosomiasis, also known as "sleeping sickness," is caused by microscopic parasites of the species Trypanosoma brucei. It is transmitted by the tsetse fly (Glossina species), which is found only in rural Africa. historically, it has been a serious public health problem in some regions of sub-Saharan Africa. Currently, about 10,000 new cases each year are reported to the World Health organization; however, it is believed that many cases go undiagnosed and unreported. Dr.T.V.Rao MD
Trypanosoma brucei • Trypanosoma brucei is a protozoan with flagella (protist) species that causes African trypanosomiasis (or sleeping sickness) in humans and nagana in animals in Africa. There are 3 sub-species of T. brucei: T. b. brucei, T. b. gambiense and T. b. rhodesiense. Dr.T.V.Rao MD
Parasite - Trypanosoma brucei ssp Trypanosoma brucei rhodesiense Trypanosoma brucei gambiense Vector - Tse Tse fly Glossina mortisans (Eastern Africa) Glossina palpalis (Western Africa) African Sleeping Sickness Dr.T.V.Rao MD
An obligate and complex parasite • These obligate parasites have two hosts - an insect vector and mammalian host. Due to the large difference between these hosts the trypanosome undergoes complex changes during its life cycle to facilitate its survival in the insect gut and the mammalian bloodstream. It also features a unique and notable variable surface glycoprotein (VSG) coat in order to avoid the host's immune system. There is an urgent need for the development of new drug therapies as current treatments can prove fatal to the patient as well as the trypanosomes. Dr.T.V.Rao MD
African Trypanosomiasis Sleeping Sickness, vector is the tsetse fly Classical example of an emerging infection, 1890-1930 Leading public health problem in Africa during that time, colonialism brought it to new areas Nearly eliminated by 1960 using population screening, case treatment, chemoprophylaxis Re-emerging infection in central Africa
Sleeping sickness - Background • Human African Trypanosomiasis (HAT) occurs in 36 sub-Saharan Africa countries • Dependent upon Tsetse fly (Glossina spp) vectors. • Highest exposure in rural populations dependent on agriculture, fishing, animal husbandry or hunting. Cattle is the key mode of human-animal transmission. • Occurs in two forms, a chronic form, that accounts for 95% of reported cases of sleeping sickness, and an acute form. Each form has different vectors with different modes of transmission. • After continued control efforts, the number of cases reported in 2009 has dropped below 10 000 for first time in 50 years. This trend has been maintained in 2010 with 7139 new cases reported. Dr.T.V.Rao MD
Trypanosomiasis • The insect vector for T. brucei is the tsetse fly. The parasite lives in the midgut of the fly (procyclic form), whereupon it migrates to the salivary glands for injection to the mammalian host on biting. The parasite lives within the bloodstream (bloodstream form) where it can reinfect the fly vector after biting. Later during a T. brucei infection the parasite may migrate to other areas of the host. A T. brucei infection may be transferred human to human via bodily fluid exchange, primarily blood transfusion.. Dr.T.V.Rao MD
Distribution Dr.T.V.Rao MD
Epidemiology of sleeping sickness • .T. b. gambiense is predominant in the western and central regions of Africa, whereas T. b. rhodesiense is restricted to the eastern third of the continent . 6,000 to 10,000 human cases are documented annually. 35 million people and 25 million cattle are at risk. Regional epidemics of the disease are cause of major health and economic disasters. Dr.T.V.Rao MD
three different sub-species of T. brucei, • T. brucei gambiense - Causes slow onset chronic trypanosomiasis in humans. Most common in central and western Africa, where humans are thought to be the primary reservoir. • T. brucei rhodesiense - Causes fast onset acute trypanosomiasis in humans. Most common in southern and eastern Africa, where game animals and livestock are thought to be the primary reservoir. • T. brucei brucei - Causes animal African trypanosomiasis, along with several other species of trypanosoma. T. b. brucei is not human infective due to its susceptibility to lysis by human apolipoprotein L1. However, as it shares many features with T. b. gambiense and T. b. rhodesiense (such as antigenic variation) it is used as a model for human infections in laboratory and animal studies. Dr.T.V.Rao MD
Virulence Reservoir Zoonotic Vector Distribution Less More Human/animal Human/animal Less More G.palpalis G.mortisans Western Africa Eastern Africa African Sleeping Sickness T .b.gambiense T.b.rhodesiense
Antigenic variation • 6-10% of the total genome of African trypanosomes is coding for VSGs (more than 1000 genes) • Only one is expressed at a given time the other 999 genes are shut down and completely silent (allelic exclusion) • At a low frequency a switch to a different gene occurs, if the host develops antibodies against the previous VSG the new clone is strongly selected • What is the advantage of expressing a single VSG? • How is expression controlled? • What mechanisms can you think of by which a cell could control gene expression and protein abundance? Dr.T.V.Rao MD
Antigenic variation • mRNA derived from only a single VSG gene can be detected at one time • VSG expression is controlled at the level of transcription initiation • Regulation of promoter activity is used to control gene expression in many organisms Dr.T.V.Rao MD
Antigenic variation • If it is not the promoter or the processing maybe it is the exact location in the genome that predisposes a specific VSG for expression • Where are active and inactive genes in the genome? • How could a location based system switch? Dr.T.V.Rao MD
Antigenic variation • All VSGs are 65 kDA glycoproteins, and are present on the surface as dimers • The outer domain is highly variable and the only conservation detected is the position of cysteines • Other (non-variant) proteins like transferrin receptor or hexose transporter are hidden in the this surface coat
Human Tse fly Trypomastigote Trypomastigote Stumpy Metacyclic Intermediate Epimastigote Slender Trypomastigote African TrypanosomiasisThe Life Cycle
Tse tse flies are vectors • Parasites are taken up with the blood meal (stumpy forms are cell cycle arrested and ‘ready to go’ for the next host • Transformation into procyclic trypomastigotes in the midgut • Migration into the ectoperitrophic space where parasites replicate • Passage into salivary glands, differentiation into epimastigotes which attach to the epithelium and massively replicate • Transformation into infectious metacyclic trypomastigotes • Again these are cell cycle arrested ‘sleepers’ Dr.T.V.Rao MD
For our purposes we will consider only two life cycle stages trypomastigotes in vertebrate host and epimastigote in Glossina which will be transmitted anterior station or salivarian transmission to the vertebrate host. Trypanosoma brucei life cycle Dr.T.V.Rao MD
Life cycle of T. brucei Dr.T.V.Rao MD
Cycle of events • 1.Uninfected tsetse fly (Glossina) bites an infected vertebrate host and ingests trypomastigote circulating in the bloodstream. • 2. Trypomastigotes multiply by longitudinal binary fission in fly gut. Dr.T.V.Rao MD
Cycle of events • 3 Trypomastigotes migrate to the salivary glands and transform into epimastigotes and multiply for several generation. Dr.T.V.Rao MD
Cycle of events • 4Epimastigotes transform back into Metacyclic Trypomastigotes (short stumpy forms) in the salivary glands. These form the infective stage. Dr.T.V.Rao MD
Cycle of events • 5 Tsetse fly bites a human or ruminant host and inoculates metacyclic trypomastigotes into bloodstream. Dr.T.V.Rao MD
Cycle of events • 6Trypomastigotes live and multiply in the blood and lymph. In some cases, trypomastigotes migrate to the central nervous system. Dr.T.V.Rao MD
African Trypanosomiasis Course of Infection • There are four phases. • The first two phases of Trypanosomiasis only show up in people of non-African decent (Europeans). Dr.T.V.Rao MD
African Trypanosomiasis Course of Infection • Phase II: Trypomastigotes enter circulation. • Fever • Headache • Skin rash • Duration is variable Dr.T.V.Rao MD
Phase III: Trypomastigotes collect in lymph nodes and channels. Cells not invaded but there is proliferation of endothelial cells Infiltration of leukocytes Enlargement of lymph nodes African Trypanosomiasis Course of Infection Dr.T.V.Rao MD
African Trypanosomiasis Course of Infection • Phase III: Trypomastigotes collect in lymph nodes and channels. • Fever, headache, and delayed sensation to pain • General weakness • Duration many years with T. b. gambiense; less than 1 year and usually less than 4 mo for T. b. rhodesiense Dr.T.V.Rao MD
Phase III Enlargement of lymph nodes in cervical triangle (on back of neck) Winterbottom’s Sign one of the cardinal signs of African Trypanosomiasis. Dr.T.V.Rao MD
Parasitemia Dr.T.V.Rao MD
African Trypanosomiasis Course of Infection • Phase IV: Invasion of Central Nervous System-African Sleeping Sickness. Dr.T.V.Rao MD
Phase IV: Invasion of Central Nervous System-African Sleeping Sickness. Headaches are severe Emaciation Mental dullness Apathy; disinclination to work Drowsiness and coma Death from asthenia, heart failure, meningitis, severe fall, etc. African Trypanosomiasis Course of Infection Dr.T.V.Rao MD
diagnosis of African Trypanosomiasis • The diagnosis of African Trypanosomiasis is made through laboratory methods, because the clinical features of infection are not sufficiently specific. The diagnosis rests on finding the parasite in body fluid or tissue by microscopy. The parasite load in T. b. rhodesiense infection is substantially higher than the level in T. b. gambiense infection Dr.T.V.Rao MD
diagnosing T. b. gambiense infection • The classic method for diagnosing T. b. gambiense infection is by microscopic examination of lymph node aspirate, usually from a posterior cervical node. It is often difficult to detect T. b. gambiense in blood. Concentration techniques and serial examinations are frequently needed. Serologic testing is available outside the U.S. for T. b. gambiense; however, it normally is used for screening purposes only and the definitive diagnosis rests on microscopic observation of the parasite. Dr.T.V.Rao MD
Importance of CSF Examination • All patients diagnosed with African trypanosomiasis must have their cerebrospinal fluid examined to determine whether there is involvement of the central nervous system, since the choice of treatment drug(s) will depend on the disease stage. The World Health Organization criteria for central nervous system involvement include increased protein in cerebrospinal fluid and a white cell count of more than 5. Trypanosomes can often be observed in cerebrospinal fluid in persons with second stage infection. Dr.T.V.Rao MD
Rapid testing methods in Africa • Detecting trypanosomes in T. b. gambiense infection is more difficult. The card agglutination test for trypanosomiasis/T. b. gambiense is a serologic screening test used for mass population screening in endemic areas of Africa. Dr.T.V.Rao MD
Why is Trypanosomiasis so deadly? • Trypanosomes are highly susceptible to antibodies and complement • They live fully exposed to antibodies in the blood stream • They induce a very strong antibody response • Still they manage to thrive in the same host for a year or longer, until the host dies Dr.T.V.Rao MD
Why is Trypanosomiasisso deadly? • Infection is characterized by periodic waves of Parasitemia • Each wave represents a single antigenically distinct clone or serotype • Antibodies produced in the first week against clone A will not react with clone B • The changing display of different antigens is called antigenic variation • Antigenic variation is an important form of immune evasion Dr.T.V.Rao MD
Management of Trypanosomiasis • Disease management in three steps: • 1) Screening for potential infection. Serological tests and/or checking for swollen cervical glands. • 2) Diagnosis shows whether the parasite is present. • 3) Staging to determine the disease progression. -examination of cerebro-spinal fluid by lumbar puncture
Chemotherapy • Early stage - most recover • Suramin • Melasporol • Pentamidine • Late stage - upto 5% relapse • Only Melasporol • 10% encephalitis - 5% fatal Dr.T.V.Rao MD
Treatments for Trypanosomiasis First stage treatments: Pentamidine: discovered in 1941, used against T.b. gambiense. Despite a few undesirable effects, it is well tolerated by patients. Only drug easily available in the US. Suramin: discovered in 1921, used against T.b. rhodesiense. Effects in the urinary tract and allergic reactions. Second stage treatments: Melarsoprol: discovered in 1949, used against both forms. Arsenic derivative with many side effects. Fatal encephalopathy (3% to 10%). 1997 resistance up to 30%. Eflornithine: was registered in 1990. Only effective against T.b. gambiense. Less toxic alternative to melarsoprol, but the regimen is strict and difficult to apply.
Prevention continues to be difficult • The most effective means of prevention is to avoid contact with tsetse flies. Vector eradication is impractical due to the vast area involved. Immunization has not been effective due to antigenic variation. Dr.T.V.Rao MD
Programme Created by Dr.T.V.Rao MD for Medical and Health Care Workers • Email • doctortvrao@gmail.com Dr.T.V.Rao MD