Electrons and the EM Spectrum
340 likes | 421 Views
Explore the evolution of atomic models from Rutherford to Bohr, diving into electron behavior, emission spectra, and the dual nature of light as wave and particle. Learn about the properties of light, atomic emission spectra, and the electromagnetic spectrum. Discover how excited electrons emit light at different energy levels and colors, and unravel the wave-particle duality of light in quantum theory.

Electrons and the EM Spectrum
E N D
Presentation Transcript
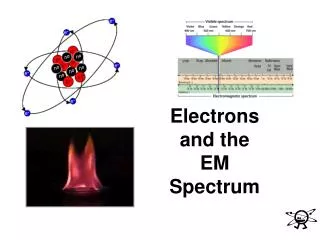
Models of the Atom So far, the model of the atom consists of protons and neutrons making up a nucleus surrounded by electrons. After performing the gold foil experiment, Rutherford hypothesized a model of the atom that looked much like the one below.
What about the electrons?? • Rutherford didn’t know exactly where the electrons were located in the atom, just that they surrounded it, or why chemical bonding occurred. • Bohr figured out that electrons orbit in energy levels around the atom.
The Bohr Model • Niels Bohr (1852-1962) was a student of Rutherford and believed the model needed improvement. • Bohr proposed that an electron is found only in specific circular paths, or orbits, around the nucleus. (write this in before “However, this model….”)
Models of the Atom However, this model could not explain the chemical and physical properties of the elements.
Models of the Atom For example it could not explain: 1. why metals give off certain colors when heated in a flame -or- 2 .why objects heated to high temperatures first glow dull red, then yellow, then white
Light is all about the Electrons! • Light is a form of electromagnetic radiation that travels like a wave through space as PHOTONS. • When electrons get excited, they jump up to higher energy levels and then fall back down • Depending on how high they jump, they will give off a different color of LIGHT
Atomic Emission Spectra excited state ENERGY IN PHOTON OUT GAIN energy LOSE energy ground state
Energy of Electrons • When atoms are heated, bright lines appear called line spectra • An electron absorbs energy to “jump” to a higher energy level. (excited) • When an electron falls to a lower energy level, energy is emitted. (ground level state)
Atomic Emission Spectra • Every element has a UNIQUE emission spectrum. The colors that you see represent the element’s electrons jumping through the energy levels! • LONG JUMPS are represented by HIGH energy colors (violet & blue) • SHORT JUMPS are represented by LOW energy colors (red).
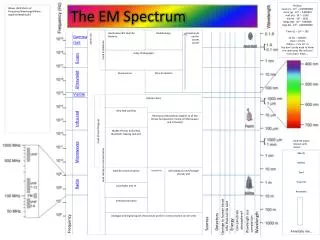
EM Spectrum HIGH ENERGY LOW ENERGY C. Johannesson
R O Y G. B I V red orange yellow green blue indigo violet EM Spectrum HIGH ENERGY LOW ENERGY
Properties of Light • Movement of excited electrons to lower energy levels, and the subsequent release of energy, is seen as light! Before 1900, scientists thought light behaved solely as a wave. This belief changed when it was later discovered that light also has particle-like characteristics. This is called the wave-particle duality of light.
First, Let’s look at the WAVE nature of light Light as a WAVE
crest A trough Wave Properties Lesser frequency greater frequency
Properties of Light • The significant feature of wave motion is its repetitive nature, which can be characterized by the measurable properties of wavelength and frequency.
Waves • Wavelength () - length of one complete wave • Frequency (f) - # of waves that pass a point during a certain time period • hertz (Hz) = 1/s
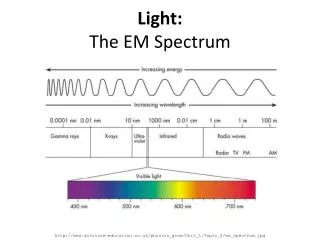
Properties of Light • Electromagnetic radiation is a form of energy that exhibits wavelike behavior (wavelength, frequency, ect) as it travels through space. Together all forms of electromagnetic radiation form the electromagnetic spectrum.
On the electromagnetic spectrum, the lowest energy waves (longest wavelength and lowest frequency) are radio waves. The highest energy waves (shortest wavelength and highest frequency) are gamma rays.
FREQUENCY and WAVELENGTH are INVERSELY proportional. (f ↑ ↓) • ENERGY and FREQUENCY are DIRECTLY proportional. (E↑ f ↑)
Now, let’s look at the PARTICLE nature of light Light as a PARTICLE
Properties of Light • In the early 1900’s, scientists conducted experiments involving interactions of light and matter that could not be explained by the wave theory of light.
Properties of Light • One experiment involved a phenomenon known as the photoelectric effect.The discovery of the photoelectric effect led to the description of light as having both wave and particle properties.
EM Spectrum • Frequency & wavelength are inversely proportional c = f c: speed of light (3.00 108 m/s) : wavelength (m, nm, etc.) f: frequency (Hz)
Example 1 If the frequency of a wave is 500 hz, what is the wavelength? C = λf C = 3.0 x 108 m/s f = 500 hz λ = ? 3.0 x 108 = 500 x λ λ = 600,000 m One sig fig or 6 x 105
Quantum Theory • The energy of a photon is proportional to its frequency. E: energy (J, joules) h: Planck’s constant (6.626 10-34 J·s) f: frequency (Hz) E = hf
Example 2 What is the energy of a wave if the frequency is 300. hz? E = hf f= 300. hz h = 6.626 x 10-34 E = 300. x 6.626 x 10-34 E = 1.99 x 10-31 3 sig figs
Example 3 • If the energy of a wave is 9.00 x 10-19 J, find frequency and wavelength E = hf E= 9.00 x 10-19hz h = 6.626 x 10-34 9.00x 10-19 = 6.626 x 10-34 x f f = 1.36 x 1015hz 3 sig figs
If the energy of a wave is 9.00 x 10-19 J, find frequency and wavelength C = λf If f = 1.36 x 1015 hz C = 3.00 x 108 m/s 3.00 x 108 m/s = λ x 1.36 x 1015 λ = 2.21 x 10 -7m 3 sig figs.