Understanding Atoms, Molecules, and States of Matter
This overview explores the fundamental concepts of atoms and molecules, their properties, and how they arrange themselves in different states of matter. Atoms, the smallest units that retain all chemical properties, bond to form molecules characterized by unique physical and chemical traits. We delve into the periodic table's shorthand notation, chemical symbols, and molecular formulas. Additionally, the varying states of matter—solid, liquid, and gas—are discussed, along with the behavior of gases and concepts of liquid crystals and metal alloys used in everyday applications.

Understanding Atoms, Molecules, and States of Matter
E N D
Presentation Transcript
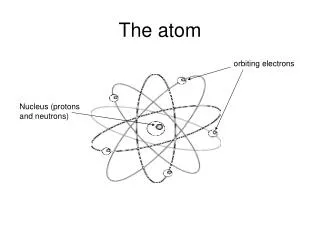
ATOM (Ch 3) • Smallest, most fundamental particle that retains all physical and chemical properties of an element. • Atomic diameter = @1.0E-10 m • Microscopic pictures show a range of ways atoms arrange themselves: from highly ordered crystals to disordered amorphous solids.
MOLECULE (Ch 3) • A molecule is a unit of matter that results when 2 or more atoms are bonded together. • Can be separated into its constituent atoms by chemical means, not physical means. • Has characteristic phys/chem properties. • Most elements exist as single atoms, but others exist as diatomic molecules (O2, H2)
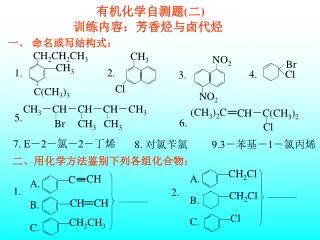
CHEMICAL SYMBOLS MOLECULAR FORMULAS • The Periodic Table lists all known elements in a shorthand notation called chemical symbols (1- or 2- letter symbols). • Compounds and molecules can also be described in a shorthand notation called molecular formulas. These include the chemical symbols for the atoms and numeric subscripts for the number of atoms.
Examples of Chemical Formulas • Atoms: H (hydrogen), He (helium), Ag (silver), Hg (mercury), Cl (chlorine) • All atoms are elements • Molecules: H2 (hydrogen), Cl2 (chlorine), N2 (nitrogen) • Some elements are molecules at room T, P • Molecules: CH4 (methane), COCl2 (phosgene), NaOCl (bleach), CH3CO2H (acetic acid), N2O (laughing gas), NO2 (nitrogen dioxide), C9H8O4 (aspirin)
COLLOIDS • Heterogeneous mixture in which particles (size = 10-6 to 10-9 m) of one substance is dispersed in another. • The two substances in a colloid are not soluble in each other and will separate unless stabilized by emulsifiers. • Table 1.2
STATES OF MATTER • Solid: atoms are closely packed and are relatively stationary; a solid maintains its own shape and volume, and is incompressible. • Gas: atoms are far apart and very mobile; a gas takes the shape and volume of the container, and is compressible. • Liquid: has intermediate properties; a liquid maintains its own volume but takes the shape of the container, and is incompressible.
STATES OF MATTER AND TEMPERATURE The atoms of a solid at low temperature remain fixed due to attractive forces. As the temp. is raised, these attractive forces are overcome by thermal motion and the atoms move away from each other thereby forming the liquid state. If the temp. continues to increase, all attractive forces are disrupted and a gas forms.
BEHAVIOR OF GASES • Consider properties of gases: P, V, T • How does V depend on T at constant P? or V = f(T) stated mathematically. As T , then V ? • V = f(P) at constant T? As P , then V? • Model of Ideal Gas: molecules (O2) are in constant motion, collide elastically, move faster as T .
LIQUID CRYSTALS • Liquid Crystals (0.05% of known cmps) have properties intermediate between solids (highly ordered) and liquids (flow slowly) • Typical chemical structures are long organic molecules that can line up in the presence of an electric voltage or T change • Typical applications are: LCD displays, dyes (cholesterics), advanced materials (Kevlar, Vectra), membranes, T measurement (by changing colors), solvents for chemical analysis
METALS • 75% of elements; all solid at room T and P except mercury (liquid) • Shiny, conductors of electricity and heat (copper wire), malleable (gold leaf), • Mercury: Hg, Z = 80, d = 13.6 g/mL, deadly, UMD Chemistry eliminated nearly all Hg from teaching program • Hypoallergenic jewelry
METAL ALLOYS • Alloy: mixture of metals to realize desired properties: strength, hardness, resistance to other cmps, malleability, weight • Gold: Au, Z = 79, most malleable, very soft, all refined gold = 60 cu ft volume • Pure gold is 24 carat = 24K • There are many alloys of gold with copper (Cu), silver (Ag): 50% gold is 12K, 58% is 14K, 42% is 10K
METAL ALLOYS • Stainless Steel: 74% Fe (iron), 18% Cr (chromium) and 8% Ni (nickel) • Steel: 99% Fe and 1% C (carbon) • Aluminum cans: Al and Mn (manganese) • Dental fillings: Hg and Ag amalgams