Translation
Translation. Function of 5 ′ CAP. Protect mRNA from degradation-RNAses cannot cleave triphosphate linkages Enhance the translatability of mRNAs-cap needed for binding of cap binding protein which is needed for attachment to ribosome Enhance the transport of the mRNA from nucleus to cytoplasm

Translation
E N D
Presentation Transcript
Function of 5′ CAP • Protect mRNA from degradation-RNAses cannot cleave triphosphate linkages • Enhance the translatability of mRNAs-cap needed for binding of cap binding protein which is needed for attachment to ribosome • Enhance the transport of the mRNA from nucleus to cytoplasm • Enhance the splicing of the mRNAs
Function of Poly (A) tail • Protection of mRNA • Translatability of mRNA- binding of poly (A)-binding protein I to the poly(A) tail region boosts the efficiency with which DNA is translated This protein in turn bind to a translation initiation factor which binds to the cap binding protein attached to the cap, effectively linking the 5’ end of the molecule to its 3’ end. This looped mRNA is more stable and readily translated. • Efficient transport from the nucleus to cytoplasm
Polyadenylation • Here, a multi-protein complex cleaves the 3'-most part of a newly produced RNA and polyadenylates the end produced by this cleavage. • The cleavage is catalysed by the enzyme Cleavage and polyadenylation specificity factor (CPSF) and occurs 10–30 nucleotides downstream of its binding site. • This site is often the sequence AAUAAA on the RNA, but variants of it exist that bind more weakly to CPSF. • Two other proteins add specificity to the binding to an RNA: CstF and CFI. CstF binds to a GU-rich region further downstream of CPSF's site.CFI recognises a third site on the RNA (a set of UGUAA sequences in mammals) and can recruit CPSF even if the AAUAAA sequence is missing. • The polyadenylation signal – the sequence motif recognised by the RNA cleavage complex – varies between groups of eukaryotes.
The RNA is cleaved right after transcription • When the RNA is cleaved, polyadenylation starts, catalysed by polyadenylate polymerase. • Polyadenylate polymerase builds the poly(A) tail by adding adenosine monophosphate units from adenosine triphosphate to the RNA, cleaving off pyrophosphate. • Another protein, PAB2, binds to the new, short poly(A) tail and increases the affinity of polyadenylate polymerase for the RNA. • When the poly(A) tail is approximately 250 nucleotides long the enzyme can no longer bind to CPSF and polyadenylation stops, thus determining the length of the poly(A) tail
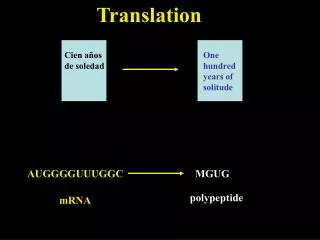
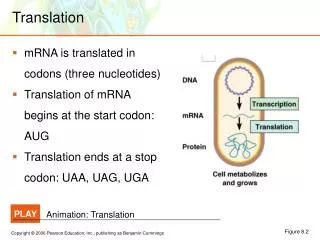
Translation ensures that: • polypeptide bonds are formed between adjacent amino acid • that the amino acids are linked in the correct sequence specified by the codons in mRNA. • mRNAs are read in the 5’→3’ direction. • Proteins are made in the amino to carboxyl direction, hence the amino terminal amino acid is added first.
The genetic code is a set of three –base code words, called codons, in mRNA that instruct the ribosome to incorporate specific amino acids into polypeptides. • Each base is part of only one codon. • There are 64 codons in all. • Three are stop signals and the rest code for amino acids.
Since there are only 20 amino acids then the code is degenerate. This is made possible by: • Isoaccepting tRNAs that bind the same amino acid although they have different anticodons. • The third base in a codon is allowed to move slightly from its normal position to form a non- Watson-Crick pair with the anticodon. • This allows the same aminoacyl-tRNA to pair with more than one codon. • This is called wobble.
Is the Genetic code Universal? • The genetic code is not strictly universal (same codons encoding the same information in all species). • Termination codons in the standard genetic code can code for aa like tryptophan and glutamine in some species.
Two events occur before protein synthesis: • Aminacyl-tRNA synthetases join amino acids to their respective tRNAs: this is done in 2 steps • First activation of the amino acid with AMP. • Secondly tRNAs picks up the activated amino acids. The resulting complexes called aminoacyl-tRNAs are able to bind to the mRNA coding sequences so as to align the amino acid in the correct order to form the polypeptide chain • Second ribosomes dissociate into subunits (happens at the end of each translation event).
Properties of tRNA molecule: • An amino acid is attached to transfer RNA before becoming incorporated into a polypeptide. • tRNAs are responsible for aligning the amino acids in the correct sequence. • Each kind of tRNA binds to a specific aa. • It must have an anticodon, a specific complementary binding sequence for the correct mRNA codon. • It must be recognized by a specific aminoacyl-tRNA synthase that adds the correct aa.
It must have a region for the attachment of the specific aa specified by the anticodon. • It must be recognized by ribosomes. • The tRNAs are polynucleotide chains 70 to 80 nucleotides long, each with several unique bases • Complementary base pairing within each tRNA molecules causes it to be doubled back and folded. • Three or more loops of unpaired nucleotides are formed, one of which contains the anticodon.
The aa binding site is at the 3’ end of the molecule. • The carboxyl group of the aa is bound to the exposed 3’ hydroxyl group of the sugar of the terminal nucleotide. • This leaves the amino group of the aa free to participate in peptide bonds. • The pattern of folding results in constant distance between the anticodon and the aa, allowing for precise positioning of the aa during translation.
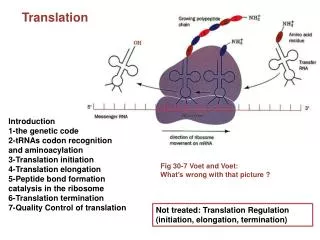
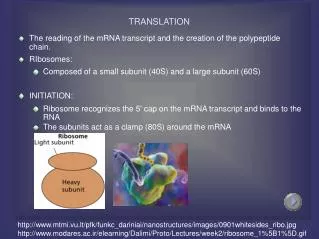
Ribosomes • Made up of 65% ribosomal RNA and 35% ribosomal proteins arranged into small and large subunits. • Ribosomes consist of two subunits that fit together and work as one to translate the mRNA into a polypeptide chain during protein synthesis. • The active part of the ribosome is RNA Eukaryotes Ribosomes: • Eukaryotes have 80S ribosomes, each consisting of a small (40S) and large (60S) subunit. • Their large subunit is composed of a 5S RNA (120 nucleotides), a 28S RNA (4700 nucleotides), a 5.8S subunit (160 nucleotides) and ~49 proteins. • The small subunit has a 1900 nucleotide (18S) RNA and ~33 proteins
The S number given each type of rRNA reflects the rate at which the molecules sediment in the ultracentrifuge. The larger the number, the larger the molecule (but not proportionally). • The 28S, 18S, and 5.8S molecules are produced by the processing of a single primary transcript from a cluster of identical copies of a single gene. The 5S molecules are produced from a different cluster of identical genes.
Prokaryotes Ribosomes: • Prokaryotes have 70S ribosomes, each consisting of a small (30S) and a large (50S) subunit. • Their large subunit is composed of a 5S RNA subunit (consisting of 120 nucleotides), a 23S RNA subunit (2900 nucleotides) and 34 proteins. • The 30S subunit has a 1540 nucleotide RNA subunit (16S) bound to 21 proteins
Translation in Prokaryotes • Translation is divided into three stages: • Initiation • Elongation • Termination.
The initiation codon in prokaryotes is usually AUG. The initiating aminoacyl-tRNA is N-formyl-methionyl-tRNAfmet. • N-formyl-methionine is the first amino acid incorporated into the polypeptide chain. • A 30S initiation complex is formed from a free 30S subunit plus a mRNA and fMet-tRNAfmet. • The 16S rRNA of the 30S initiation complex first base pairs with a sequence called the Shine-Delgarno sequence upstream from the initiation codon.
http://themedicalbiochemistrypage.org/images/shine-delgarno.jpghttp://themedicalbiochemistrypage.org/images/shine-delgarno.jpg
This binding is mediated by IF3 with the help of IF2 and IF1. • The initiation complex then slides along the mRNA until it reaches the initiation codon. • A 30S initiation complex is formed from a free 30S subunit plus a mRNA and fMet-tRNAfmet, GTP, IF1, IF2 and IF3.
GTP is hydrolysed after the 50S subunit joins the 30S complex to form the 70S initiation complex.
Elongation: • Is the addition of other aa to the growing polypeptide chain. • Takes place in three steps: 1. EF-Tu with the help of GTP, binds an aminoacyl-tRNA to the A site by specific base-pairing of its anticodon and the complementary mRNA codon. • The amino group of the aa at the A site is aligned with the carboxyl group of the preceding aa at the P site. 2. Peptidyl transferase forms a peptide bond between the peptide in the P site and the newly arrived aminoacyl- tRNA in the A site.
In this process the aa that is attached to the P site is released from its tRNA and becomes attached to the aminoacyl-tRNA at the A site • Peptidyle transferase activity resides on the 50S subunit (on its 23S rRNA) 3. EF-G, with GTP translocates the growing peptidyl-tRNA, with its mRNA codon to the P site. leaving the A site free for the next aminoacyl-tRNA. • Each translocation moves the mRNA one codon’s length through the ribosome so that the mRNA codon specifying the next aa in the polypeptide chain becomes positioned in the unoccupied A site.
The end of the mRNA which is transcribed first is also the first to be translated. • An average sized protein of about 360 aa can be assembled by a prokaryote in 18 seconds and by a eukaryotic cell in little over a minute.
Elongation in eukaryotes Elongation in eukaryotes is carried out with two elongation factors: eEF-1 and eEF-2: • The first is eEF-1, whose α subunit act as counterparts to EF-Tu. • The second is eEF-2, the counterpart to prokaryotic EF-G
Animation of translation in prokaryotes • http://www.phschool.com/science/biology_place/biocoach/translation/elong1.html • http://www.chromosome.com/DNA_animations/protein.mov
Accuracy of elongation Mediated in two ways: • The protein-synthesizing machinery gets rid of ternary complexes bearing the wrong aminoacyl-tRNA before GTP hydrolysis. • It also eliminates incorrect aminoacyl-tRNA in the proofreading step before its amino acid gets incorporated into the polypeptide.
Termination: • The synthesis of the polypeptide chain is terminated by release factors that recognize the termination or stop codon at the end of the coding sequence. • Prokaryotic translation termination is mediated by three factors: RF1, RF2 and RF3. RF1 recognizes UAA and UGA. RF3 is a GTP-binding protein that facilitates binding of RF1 and RF2 to the ribosome. • The release factors release the newly formed protein, the mRNA, and the last tRNA used, • The ribosome dissociates into its two subunits, which are then reused.
Initiation in Eukaryotes • Eukaryotic 40S ribosomal subunits and initiator tRNA (tRNAimet) locate the appropriate start codon by binding to the 5′-cap of an mRNA and scan downstream until they locate the first AUG. • The initiation factors are also different than those in prokaryotic initiation: • eIF2 is involved in binding Met-tRNAimet to the ribosome. • eIF2 is a GTP-binding protein responsible for bringing the initiator tRNA to the P-site of the pre-initiation complex. • It has specificity for the methionine-charged initiator tRNA, which is distinct from other methionine-charged tRNAs specific for elongation of the polypeptide chain. • Once it has placed the initiator tRNA on the AUG start codon in the P-site, it hydrolyzes GTP into GDP, and dissociates.
eIF2B activates eIF2 by replacing its GDP with GTP. • eIF3 binds to the 40S ribosomal subunit and inhibits its reassociation with 60S subunit: • eIF1, eIF1A, and eIF3, all bind to the ribosome subunit-mRNA complex. • They have been implicated in preventing the large ribosomal subunit from binding the small subunit before it is ready to commence elongation.
eIF4 is a cap-binding protein composed of 3 parts: • eIF4E has cap-binding activity • eIFA has RNA helicase activity and unwinds 5’-leaders of eukaryotic mRNAs. • eIF4G is an adaptor protein that bind to proteins like: eIF3 (the 40S ribosomal subunit binding protein) and Pab1p (a poly (A) tail binding protein) thereby associating 40S subunit with both ends of mRNA and stimulate initiation.
eIF5 encourages association between the 43S complex (comprising the 40S subunit plus Met-tRNAimet) and large ribosomal subunit: • eIF5A is a GTPase-activating protein, which helps the large ribosomal subunit associate with the small subunit. It is required for GTP-hydrolysis by eIF2 and contains the unusual amino acid hypusine. • eIF5B is a GTPase, and is involved in assembly of the full ribosome (which requires GTP hydrolysis). • eIF6 binds to the 60S subunit and blocks its reassociation with 40S subunit.
http://wormbook.sanger.ac.uk/chapters/www_mechregultranslation/RhoadsMRTfig1.jpghttp://wormbook.sanger.ac.uk/chapters/www_mechregultranslation/RhoadsMRTfig1.jpg
Termination in Eukaryotes • Eukaryotes have 2 release factors: • eRF1 that recognizes all three termination codons • eRF3, a ribosome-dependent GTPase that helps eRF1 release the finished polypeptide.
Polysomes • A single mRNA molecule usually has many ribosomes traveling along it, in various stages of synthesizing the polypeptide. This complex is called a polysome
References • http://www.frontiers-in-genetics.org/en/pictures/translation_1.jpg • http://en.wikipedia.org/wiki/Eukaryotic_initiation_factor • Weaver R. F. Molecular Biology. Fourth edition. McGraw Hill Higher Education. • http://www.uic.edu/classes/bios/bios100/summer2002/ribosome01.gif • http://www.agen.ufl.edu/~chyn/age2062/OnLineBiology/OLBB/www.emc.maricopa.edu/faculty/farabee/BIOBK/ribosome.gif • http://en.wikipedia.org/wiki/Ribosome