Vancomycin
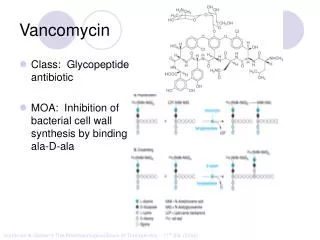
Vancomycin. Class: Glycopeptide antibiotic MOA: Inhibition of bacterial cell wall synthesis by binding D-ala-D-ala. Goodman & Gilman’s The Pharmacological Basis of Therapeutics – 11 th Ed. (2006). “Penicillin binding protein”. Peptidoglycan Synthesis. Vancomycin. IV, PO

Vancomycin
E N D
Presentation Transcript
Vancomycin • Class: Glycopeptide antibiotic • MOA: Inhibition of bacterial cell wall synthesis by binding D-ala-D-ala Goodman & Gilman’s The Pharmacological Basis of Therapeutics – 11th Ed. (2006)
“Penicillin binding protein” Peptidoglycan Synthesis
Vancomycin • IV, PO • Spectrum: Gram (+) • Drug of Choice • MRSA • Indications • IV: Serious methicillin-resistant Staphylococcal infections: pneumonia, endocarditis, osteomyelitis, SSSI • PO: pseudomembranous colitis (metronidazole preferred) • Staphylococcal infections in Penicillin allergic patients • NOTE: Do not use in non-Penicillin allergic patients. Vancomycin does not kill as rapidly as antistaphylococcal β-lactams, and may negatively impact clinical outcome • Unique Qualities • Monitor trough serum concentrations • Poor oral absorption • Adjust dose for renal impairment • ADRs • “Red Man” Syndrome • Ototoxicity • Nephrotoxicity w/ other nephrotoxic agents
Vancomycin • Mechanism of action: Inhibits bacterial cell wall synthesis • Spectrum of action: Gram positive organisms • Including: Listeria, Rhodococcus, Peptostreptococcus • Bacteriostatic against enterococcus • Mechanism of resistance: • Enterococcus: Van A – E • Peptidoglycan precursor has decreased affinity for vancomycin – D-ala-D-ala replaced by D-ala-D-lac • Staphylococcus aureus: • VISA isolates: • Increased amount of precursor with decreased affinity • Thicker cell wall • hVISA: heterogenous bacterial population
Vancomycin • Dose: • Based on total body weight and renal function • 15 – 20 mg/kg • Normal renal function: q 12 dosing • Goal trough concentrations: • 10 – 15 mcg/mL: bacteremia, skin and soft tissue infections • 15 – 20 mcg/mL: osteomyelitis, meningitis, pneumonia
Linezolid • Class: Oxazolidinedione • MOA: Binds P site of 50s ribosomal subunit, preventing translation initiation Goodman & Gilman’s The Pharmacological Basis of Therapeutics – 11th Ed. (2006) http://www.chm.bris.ac.uk/motm/linezolid/linezolid.htm
Linezolid • IV, PO • Gram (+) Indications • VRE (E. faecium) • Nosocomial pneumonia (S. aureus) • Community-acquired pneumonia (S. pneumoniae) • cSSSI (S. aureus) • Unique Qualities • F~100%, IV=PO • Reserve use for treatment of multiple drug resistant strains • No CYP interaction • ADRs • Generally well tolerated w/ minor SE in short term Rx • Myelosuppression: anemia, leukopenia, pancytopenia, thrombocytopenia • Peripheral and optic neuropathy
Linezolid • Penetration: • Plasma • Pulmonary lining • Blister fluid • Dose (IV or PO): 600 mg Q12H • Drug-drug interactions: • Non-selective inhibitor of MAO • Possible serotonergic or adrenergic interaction with anti-depressant medications (incidence < 1%) > MIC90 for Staphylococcus
Daptomycin • Class: Cyclic lipopeptide • MOA: In the presence of Ca2+, binds bacterial membrane resulting in depolarization Goodman & Gilman’s The Pharmacological Basis of Therapeutics – 11th Ed. (2006) http://cubicin.com/am_moa.htm
Daptomycin • Indications: • Treatment of complicated SSTI’s caused by gram positive bacteria • Treatment of Staphylococcus bacteremia and right-sided endocarditis • Not used for treatment of pneumonia due to binding reaction with surfactant inactivates daptomycin • MOA: • Binds membrane Rapid depolarization Cell death
Daptomycin • Pharmacokinetic profile: • Concentration-dependent killing • Post-antibiotic effect • Available for intravenous use only • Penetration: • Good penetration into vascular tissues and plasma • Currently testing penetration into cerebral spinal fluid • Dose: • SSTIs: 4 mg/kg IV daily • Bacteremia: 6 mg/kg IV daily • Adjust for decreased renal function – CrCl < 30, use qod • Can Interact with certain assays for INR testing – results in falsely high INR recommend point of care testing
Macrolide Mechanism of Action • Bacteriostatic • Inhibits protein synthesis • Bind reversibly to 50s unit of the ribosome • Blocks translocation of peptides from A-site to P-site. Goodman and Gilman’s The Pharmacological Basic of Therapeutics. 11ed. 2006
Macrolides • Achieve higher tissue than plasma concentrations • Penetrate into respiratory, tonsillar, and prostate tissues • Also penetrate into PMN leukocytes • Important for Atypicals like: Chlamydia and Legionella species • PD: Time the bacteria is exposed to therapeutic concentrations above the MIC best predicts efficacy – time dependent killing
Clarithromycin • 14-membered lactone ring • Replace hydroxyl group at C-6 position with methoxyl group • Increase stability under acidic conditions • Partially metabolized via CPYP3A4 converted to active metabolite 14-OH-clarithromycin • Primarily excreted in urine Goodman and Gilman’s The Pharmacological Basic of Therapeutics. 11ed. 2006
Clarithromycin • PO: Biaxin® 250-500 mg q 12 hours; Biaxin XL® 1000 mg qday • Spectrum of Activity: Gram (+) and Gram (-) • Indications: otitis media, CAP, pharyngitis/tonsillitis, sinusitis, uncomplicated skin infections, prevention of MAC, duodenal ulcer disease • S. aureus, S. pyogenes, S. pneumoniae, Mycobacterium avium complex • C. pneumoniae, C. trachomatis, L. pneumoniae • H. influenzae, H.pylori • Drug Interactions: Substrate of CYP 3A4 and Inhibits CPY 3A4(major) CYP 1A2 (weak) • Theophylline, statins, digoxin, warfarin, cyclosporine • Renal Adjustments: • CrCl < 30 ml/min: ½ the normal dose or double the dosing interval • ADR: • Prolongs the QT interval – use with caution in CAD • N/V, diarrhea, headache • Counseling Points: • Take XL formulation with food; do not chew or crush
Azithromycin • 15-membered lactone ring • N-methyl group inserted between C-9 and C-10 • Ketone replaced with –CH2 Goodman and Gilman’s The Pharmacological Basic of Therapeutics. 11ed. 2006
Azithromycin • PO, IV • Azithromyicn: 500 mg x day 1 then 250 mg x day 2-5 • STDs: C. trachomatis: 1 g x 1; N. gonorrheae: 2 g x 1 • Spectrum of Activity: Less Gram (+), increased Gram (-) • Indications: otitis media, pharyngitis/tonsillitis, upper and lower respiratory tract infections, skin and skin structure, CAP, PID, STDs • S. aureus, S. pneumoniae, H. influenzae, Mycobacterium avium complex • C. trachomatis, M. catarrhalis, M. pneumonia, N. gonorrheae, Chlamydia pneumoniae • Drug Interactions: not as significant as other macrolides • Most documented with cyclosporine and tacrolimus • Unique Characteristics: • T ½ 60-70 hours • Caution in patients with CrCl < 10 ml/min • ADRs: • Generally well-tolerated, may cause GI upset
Macrolide Resistance • Decrease of permeation of drug through the cell membrane, or drug efflux pumps • Methylase modifies the ribosomal target • Hydrolysis of macrolides by endogenous esterase
Clindamycin • Class: Lincosamide • Mechanism of Action: Binds exclusively to the 50S subunit of bacterial ribosomes and suppress protein synthesis
Trade names: Cleocin ®, Clindesse®, Clindagel ®, Delivery forms: capsules: 75, 150, 300 mg; granules for oral solution 75mg/5ml; injection 150 mg/ml; vaginal cream 2%; vaginal suppositories 100 mg; Clindamax ®, Evoclin ® topical gel 1%; topical lotion 1%; topical solution 1%; foam 1% Clindamycin
Indications: Serious infections caused by susceptible anaerobic bacteria Off-label indications: CNS toxoplasmosis in AIDS patients in addition to pyrimethamine; chlamydia infections in women; bacterial vaginosis due to Gardnerella vaginalis Dosing: Adults:150-450 mg Q 6 hrs Children:8-20 mg/kg/day divided TID-QID Instructions: Take with full glass of water Warning: Pseudomembranous colitis Clindamycin
Precautions: Renal impairment/liver disease Elderly Meningitis GI disease Superinfections Pregnancy Category B Drug Interactions: Erythromycin Neuromuscular blocking agents ADRs: Dermatologic, GI, Hypersenstivity Clindamycin
Aminoglycosides • Bactericidal inhibitors of protein synthesis • Concentration dependent bacteria killing • Postantibiotic effect • Major limitation of use is toxicity • Nephrotoxicity • Ototoxicity
Aminoglycosides: Indications • Primarily against aerobic, gram negative bacilli • Activity against gram positive bacteria limited • Synergistic effect against “sensitive” (high-level) streptococci and enterococci when used with a cell wall active agent
Penetrating the Cell Gram Negative Bacteria • Diffuse through porin proteins on the outer membrane of gram negative cell wall • Transport across inner membrane depends on electron transport • Membrane potential drives permeation • Transport can be blocked by reduction in pH and anaerobic environment Adapted from: http://web.indstate.edu/thcme/micro/respiratory/sld006.htm
Mechanism of Action • Bactericidal • Inhibit protein synthesis • Bind to bacterial 30S ribosomal subunit • Blocks initiation of protein synthesis • Cause misreading of mRNA template • Cause premature termination of translocation Goodman and Gilman’s
Aminoglycosides: Resistance Modes of resistance • Decreased permeation of aminoglycosides • Low affinity for bacterial ribosome • Drug inactivation by microbial enzymes • Important clinically • Amikacin is less vulnerable
Structure Goodman and Gilman’s
Aminoglycosides • Resistance: Intrinsic vs. Acquired • Intrinsic: • Anaerobes: lack active electron transport chain to cross membrane • Mutation at 16s rRNA (ie TB) • Acquired: • Efflux: seen in Pseudomonas • Decreased transmembrane potential: seen in Enterococcus
Aminoglycosides • Distribution: • Freely into the vascular space • Interstitial spaces of most tissues • Volume of distribution increases in edematous states and decreases in obese patients (on L/kg basis) • Decreased concentrations: • Bronchial secretions, CSF, biliary tract, synovial fluid, and in the eye • Excreted by the kidneys • Half-life: 1.5 to 3.5 hours
Aminoglycosides • Toxicity: • Nephrotoxicity: • Incidence 5% to 25% • Risk factors: • Ototoxicity (cochlear, vestibular) • Neuromuscular blockade (very rare)
Toxicity • Dependent on: • Total amount of drug AND duration of therapy • Nephrotoxicity • Most often reversible • Accumulation of drug in proximal tubular cells • Mild rise in Scr (0.5-1 mg/dl) • Reduced excretion of drug = increased risk of ototoxicity • Ototoxicity • Largely irreversible if not caught early • Destruction of vestibular and cochlear sensory cells • High-pitched tinnitus is often 1st symptom
Aminoglycosides • Site of infection: determines goal levels and dose • Peak concentrations: • Gram + Synergy: 3 – 5 mcg/mL • UTI: 3 – 4 mcg/mL • Bacteremia: 6 – 8 mcg/mL • Pneumonia: 8 – 10 mcg/mL • Weight based dosing: use IBW or ABW • Interval: once-daily dosing for gram-negative infection (normal renal function, 7 mg/kg/day). Gram + synergy 1mg/kg q 8-12h.
Gentamicin: Once Daily Dosing • 5-7mg/kg/24hrs (ABW) • Target peak 14-20 mcg/ml • Allows low troughs • Avoid in patients with: • Burns, CF, pregnancy, children, endocarditis or CrCl < 20ml/min
Tobramycin • Antimicrobial activity and PK properties very similar to gentamicin • Superior activity against P. aeruginosa • Less active than gentamicin against enterococci • Can be given IV or IM • Dosage and serum levels are same as gentamicin
Amikacin • Broadest spectrum of activity • Resistant to aminoglycoside-inactivating enzymes • Less active against enterococci • Similar dosing interval and monitoring • Peak • Life-threatening infection 25-30 mcg/ml • Serious infection 20-25 mcg/ml • Trough • Life threatening infection 4-8 mcg/ml • Most infections 1-4 mcg/ml