Solubility, Concentrations, & Molarity
440 likes | 808 Views
April 26-May 7, 2010. Solubility, Concentrations, & Molarity. Solutions Definitions. Solution - homogeneous mixture consisting of two parts. Solute - substance being dissolved. Solvent - the substance that is dissolving the solute ( present in greater amount ).

Solubility, Concentrations, & Molarity
E N D
Presentation Transcript
April 26-May 7, 2010 Solubility, Concentrations, & Molarity
Solutions Definitions • Solution - homogeneous mixture consisting of two parts • Solute - substance being dissolved • Solvent - the substance that is dissolving the solute (present in greater amount)
Measuring Concentration • Concentration…a measure of solute-to-solvent ratio • Common Units: • Molarity moles solute / L of solution • ppm g solute / 1,000,000 g of solution • % → g solute/ 100 g of solution • concentrated vs. dilute • “lots of solute” “not much solute” • “watery” Add water to dilute a solution; boil water off to concentrate it.
mol mol L M L M = Measuring Concentration“The amount of solute in a solution” A. mass % = mass of solute mass of solution B. parts per million (ppm) also, ppb and ppt – commonly used for minerals or contaminants in water supplies C. molarity (M) = moles of solute L of solution – used most often in this class % by mass – medicated creams % by volume – rubbing alcohol
Mass Percent • Parts of solute in every 100 parts solution. • If a solution is 0.9% by mass, then there are 0.9 grams of solute in every 100 grams of solution. • Or 10 kg solute in every 100 kg solution. • Since masses are additive, the mass of the solution is the sum of the masses of solute and solvent. Tro's Introductory Chemistry, Chapter 13
liter of solution Molarity
QUICK REVIEW OF MOLES!! • The concentration can then be used to convert the moles of solute into the liters of solution, or visa versa. • Since we normally measure the amount of solute in grams, we will need to convert between grams and moles. • How many moles are in 258 g of K2S?
moles of solute liters of solution Molarity = Solution Concentration Molarity • If a sugar solution concentration is 2.0 M , 1 liter of solution contains 2.0 moles of sugar, 2 liters = 4.0 moles sugar, 0.5 liters = 1.0 mole sugar:
mol NaOH L solution Example 13.4—How Many Liters of a 0.114 M NaOH Solution Contains 1.24 mol of NaOH? Given: Find: 1.24 mol NaOH volume, L Solution Map: Relationships: 1.00 L solution = 0.114 mol NaOH Solve: Check: The unit is correct, the magnitude seems reasonable as the moles of NaOH > 10x the amount in 1 L. Tro's Introductory Chemistry, Chapter 13
Molarity • Find the molarity of a solution containing 75 g of MgCl2 in 250 ml of water. 75 g MgCl2 1 mol MgCl2 95.21 g MgCl2 0.25 L water = 3.2M MgCl2
Calculating Molarity What is the molarity of a potassium chloride solution that has a volume of 400 ml and contains 85.0 grams of KCl?
Example 2 Vinegar contains 5.0 grams of acetic acid,CH3COOH, in 100.0 ml of solution. Calculate the molarity of acetic acid in vinegar.
Practice—Determine the Mass of CaCl2 (MM = 110.98) in 1.75 L of 1.50 M Solution, Continued. Given: 1.75 L solution Find: g CaCl2 Equivalence: 1.50 mol CaCl2 1 L solution; 110.98 g = 1 mol CaCl2 Solution Map: L solution mol CaCl2 g CaCl2 Apply Solution Map: Check Answer: Units are correct. Tro's Introductory Chemistry, Chapter 13
g solute / g of solution x 1,000,000= Parts Per Million (ppm) • EXAMPLE • There are 2.2 mg of lead in exactly 500g of water. Convert this to ppm.
Assignment • Page 45 • #1, #2, #3, #4, #5, #6, #8
Answers to Page 45 2. 3.
Answers to assignment Page 45 4. 5. 8.
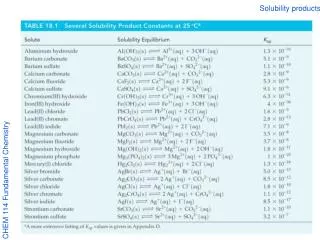
Solubility • When one substance (solute) dissolves in another (solvent) it is said to be soluble. • Salt is soluble in water. • When one substance does not dissolve in another it is said to be insoluble. • Oil is insoluble in water. • There is usually a limit to the solubility of one substance in another. • Gases are alwayssoluble in each other. • Two liquids that are mutually soluble are said to be miscible. • Alcohol and water are miscible. • Oil and water are immiscible. • The maximum amount of solute that can be dissolved in a given amount of solvent is called solubility.
Descriptions of Solubility • Saturated solutions have the maximum amount of solute that will dissolve in that solvent at that temperature. • Unsaturated solutions can dissolve more solute. • Supersaturated solutions are holding more solute than they should be able to at that temperature. • Unstable. Tro's Introductory Chemistry, Chapter 13
UNSATURATED SOLUTION more solute dissolves SATURATED SOLUTION no more solute dissolves SUPERSATURATED SOLUTION becomes unstable, crystals form Solubility: forming a “saturated” solution in equilibrium increasing concentration
Changing Temperature = Changing Solubility • When a solution is saturated, it is holding the maximum amount of solute it can at that temperature. • The solubility of the solute in the solvent depends on the temperature. • If the temperature is changed, the solubility of the solute changes. • Higher temperature = Higher solubility of solid in liquid. • Lower temperature = Higher solubility of gas in liquid. Tro's Introductory Chemistry, Chapter 13
Saturation Problems!!Page 44 (#1) • NaCl has a solubility of 35.9 g/100ml of water. Determine whether the following soln are sat., unsat., supersat. • 85 g of NaCl dissolved in 150 mL of water • 25 g of NaCl dissolved in 750 g of water • 135 g of NaCl dissolved in 750 g of water
Saturated, Unsaturated &Super Saturated Problems • Problems on Page 47 • Do one for example • Assign other two
Assignment • Page 45-46 (concentration problems) • #9, #10, #11 • Page 47 (saturation problems) • #2, #3 **DUE on Friday**
Concentration Lab • April 22, 2009
Tasks: Thursday 4-23 • Complete Lab write-up from the Concentration Lab and 7th Chromatography Lab also • Page 46 • #14, #15, #16, #18 10. 3.7 grams 11. 33.6 grams 16. 3.51 grams 18. 4.03 mol/ liter
Page 44 Page 43 189 grams 0.18 L 0.082L 2. a. Unsaturated (0.789) b. Unsaturated (0.975) c. Unsaturated (0.66) 3. a. Supersaturated (5.1 x10-4) b. Supersaturated (0.029) c. Supersaturated (7.08 x 10 -5)
April 24 Physical Properties & Characteristics of Solutions
Physical Properties of Solutions • Conductivity: • Ability to conduct an electric current • Depends on whether it contains charged particles • Do all compounds conduct electricity?
Electrolytes NaCl(aq) Na+ + Cl- HF(aq) H+ + F- Electrolytes - solutions that carry an electric current strong electrolyte weak electrolyte nonelectrolyte Timberlake, Chemistry 7th Edition, page 290
Electrolytes • Electrolytes are substances whose aqueous solution (dissolved ions) is a conductor of electricity. Not all electrolytes conduct same amount of electricity. • In strong electrolytes, all the electrolyte molecules are dissociated into ions. • In nonelectrolytes, none of the molecules are dissociated into ions. No electric current. • In weak electrolytes, a small percentage of the molecules are dissociated into ions.
Healthy Info electrolytes: solutes that dissociate in solution -- conduct electric current because of free-moving ions e.g., acids, bases, most ionic compounds -- are crucial for many cellular processes -- obtained in a healthy diet -- For sustained exercise or a bout of the flu, sports drinks ensure adequate electrolytes. e.g., any type of sugar nonelectrolytes: solutes that DO NOT dissociate -- DO NOT conduct electric current (not enough ions)
Colligative Properties • The properties of the solution are different from the properties of the solvent. • Any property of a solution whose value depends only on the number of dissolved solute particles is called a colligative property. • It does not depend on what the solute particle is. • The freezing point, boiling point, and osmotic pressure of a solution are colligative properties. Tro's Introductory Chemistry, Chapter 13
Ice Cream Lab Data • Average measurements over the entire day: • 6 Tbs. salt= -8*C • 12 Tbs. salt = -10*C • 18 Tbs. salt = -15*C • The more salt that was added the lower the freezing point became!
Surfactants Definition: Types: Detergent- synthetic, works in hard water Emulsion- solution of two immiscible substances (a colloid in which both phases are liquids; "an oil-in-water) Soap- used to surround a molecule making it miscible (ex: surrounds oil-allows it to dissolve in water) • A chemical agent capable of reducing the surface tension of a liquid in which it is dissolved • Occurs at the boundary of immiscible solutions
Cleaning Action of Soap Micelle Timberlake, Chemistry 7th Edition, page 573
Why Do We Do That? • We spread salt on icy roads and walkways to melt the ice. • We add antifreeze to car radiators to prevent the water from boiling or freezing. • Antifreeze is mainly ethylene glycol. • When we add solutes to water, it changes the freezing point and boiling point of the water. Tro's Introductory Chemistry, Chapter 13
Water Molecule Water is a POLAR molecule H+ d+ H2O d+ d- O2- H+ d-
+ H H O - - + - + Polar water molecules hydrates Na+ and Cl- ions Na+ + + hydrated ions - + - Cl- Timberlake, Chemistry 7th Edition, page 287
Na+ ions Water molecules Cl- ions Dissolving of NaCl in Water NaCl(s) + H2O Na+(aq) + Cl-(aq)
Will It Dissolve? • Chemist’s rule of thumb: Like dissolves like • A chemical will dissolve in a solvent if it has a similar structure to the solvent. • When the solvent and solute structures are similar, the solvent molecules will attract the solute particles at least as well as the solute particles to each other. Tro's Introductory Chemistry, Chapter 13
“Oil and Water Don’t Mix” • Oil is nonpolar • Water is polar “Like dissolves like”, nonpolar dissolves nonpolar, nonpolar does not dissolve polar Zumdahl, Zumdahl, DeCoste, World of Chemistry2002, page 470
Will It Dissolve in Water? • Ions are attracted to polar solvents. • Many ionic compounds dissolve in water. • Generally, if the ions total charges < 4. • Polar molecules are attracted to polar solvents. • Table sugar, ethyl alcohol, and glucose all dissolve well in water. • Have either multiple OH groups or little CH. • Nonpolar molecules are attracted to nonpolar solvents. • b-carotene (C40H56) is not water soluble; it dissolves in fatty (nonpolar) tissues. • Many molecules have both polar and nonpolar structures—whether they will dissolve in water depends on the kind, number, and location of polar and nonpolar structural features in the molecule.