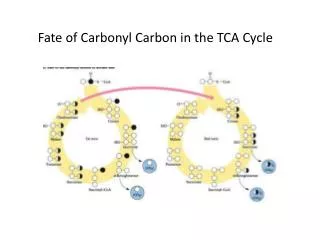
tca cycle
TCA CYCLE

tca cycle
E N D
Presentation Transcript
TCA CYCLE PRESENTED BY :P.ARSULA PETERBIOCHEMISTRY LECTURERemail:arshu4127@gmail.comSt.Xavier’s catholic nursing college Nagercoil
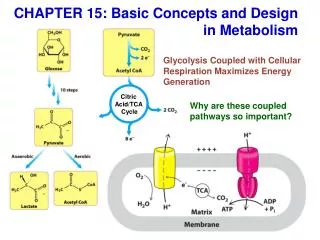
Definition: The citric acid cycle, also known as the tricarboxylic acid (TCA) cycle or the Krebs cycle, represents one part of the three- part aerobic cellular respiration pathway. In cellular respiration, a molecule of glucose enters the pathway and adenosine triphosphate (ATP) ultimately gets produced for use as a source of energy to run cellular processes.
The Krebs Cycle • Occurs in the matrix of the mitochondrion • Aerobic phase (requires oxygen) • 2-carbon acetyl CoA joins with a 4-carbon compound to form a 6- carbon compound called Citric acid
TCA Cycle Summary 1 acetate through the cycle produces 2 CO2, 1 GTP, 3NADH, 1FADH2
Citric acid (6C) is gradually converted back to the 4-carbon compound -ready to start the cycle once more • The carbons removed are released as CO2 -enzymes controlling this process called decarboxylases • The hydrogens, which are removed, join with NAD to form NADH2 -enzymes controlling the release of hydrogen are called dehydrogenases
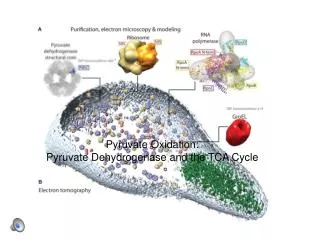
Pyruvate Oxidation Pyruvate + CoA + NAD+ acetylCoA + CO2 + NADH + H+
Pyruvate Dehydrogenaseis a large complex: pyruvate dehydrogenase (E1), dihydrolipoyl transacetylase (E2), dihydrolipoyl dehydrogenase (E3) Requires 5 coenzymes: TPP, Lipoic Acid, Coenzyme A, FAD, NAD+
Pyruvate dehydrogenase Complex (PDC) • It is a multi-enzyme complex containing three enzymes associated together non-covalently: • E-1 : Pyruvate dehydrogenase , uses Thiamine pyrophosphate as cofactor bound to E1 • E-2 : Dihydrolipoyl transacetylase, Lipoic acid bound, CoA as substrate • E-3 : Dihydrolipoyl Dehydrogenase FAD bound, NAD+ as substrate • Advantages of multienzyme complex: • Higher rate of reaction: Because product of one enzyme acts as a substrate of other, and is available for the active site of next enzyme without much diffusion. • Minimum side reaction. • Coordinated control.
Reaction 1: Citrate Synthase A thioester: so a high energy compound Hydrolysis helps drive this reaction forward Allosteric, -ATP,NADH, succinyl-CoA
Reaction 2: Aconitase Dehydration followed by hydration
Reaction 3: Isocitrate Dehydrogenase FirstOxidative decarboxylation Allosteric enzyme -ATP,NADH
Reaction 4: -Ketoglutarate Dehydrogenase SecondOxidative decarboxylation
Reaction 5: Succinyl CoA synthatse:Sccinyl CoA, like Acetyl CoA has a thioester bond with very negative free energy of hydrolysis. In this reaction, the hydrolysis of the thioester bond leads to the formation of phosphoester bond with inorganic phosphate. This phosphate is transferred to Histidine residue of the enzyme and this high energy, unstable phosphate is finally transferred to GDP resulting in the generation of GTP.
Reaction 6: Succinate Dehydrogenase Oxidation 2 e- carrier is FAD
Succinate Dehydrogenase • Part of electron transport chain in the inner membrane of mitochondria. • Removal of H across a C-C bond is not sufficiently exergonic to reduce NAD+,but it does yield enough energy to reduce FAD. • Malonate is a competitive inhibitor
Reaction 7: Fumarase Hydration trans-addition of the elements of water across the double bond, forms L-malate
Reaction 8: Malate Dehydrogenase Oxidation This and the previous two reactions form a reaction triad +
TCA Cycle Summary 1 acetate through the cycle produces 2 CO2, 1 GTP, 3NADH, 1FADH2
Regulation of CAC: Rate controlling enzymes: Citrate synthatase Isocitrate dehydrogenase a-keoglutaratedehydrogenase Regulation of activity by: Substrate availability Product inhibition Allosteric inhibition or activation by other intermediates
Energetics • Energy is conserved in the reduced coenzymes NADH, FADH2 and one GTP • NADH, FADH2 can be oxidized to produce ATP by oxidative phosphorylation 1.5 1.5 7.5 ETS 1.5 7.5 7.5
ATP Energy from Glycolysis(Aerobic) • In the electron transport system NADH = 3 ATP FADH2 = 2 ATP • Glycolysis Glucose 2 pyruvate + 2 ATP + 2 NADH NADH in cytoplasm FADH2 mitochondria Glucose 2 pyruvate + 8 ATP
ATP Energy from Pyruvate 2 pyruvate 2 acetyl CoA + 2 CO2 + 2 NADH 2 pyruvate 2 acetyl CoA + 2 CO2 + 6 ATP
ATP Energy from Citric Acid Cycle One turn of the citric acid cycle 3 NADH x 3 ATP = 9 ATP 1 FADH2 x 2 ATP = 2 ATP 1 GTP x 1 ATP = 1 ATP Total = 12 ATP Glucose provides two acetyl COA molecules for two turns of citric acid cycle 2 acetyl CoA 24 ATP + 4 CO2
ATP from Glucose For 1 glucose molecule undergoing complete oxidation Glycolysis8ATP 2 Pyruvate to 2 Acetyl CoA 6 ATP 2 Acetyl CoA to 4 CO224 ATP Glucose + 6 O2 6 CO2 + 6 H2O + 38 ATP


![Tricarboxylic acid cycle (TCA Cycle) [Kreb’s cycle] [Citric acid cycle]](https://cdn1.slideserve.com/2408379/tricarboxylic-acid-cycle-tca-cycle-kreb-s-cycle-citric-acid-cycle-dt.jpg)