Ultraviolet/Visible (UV-Vis) Spectroscopy of Potassium Permanganate
Ultraviolet/Visible (UV-Vis) Spectroscopy of Potassium Permanganate. By Robert Bohman November 4 th 2006 CH EN 4903. Importance to industry. Potassium Permanganate is used to kill bacteria in reclaimed water

Ultraviolet/Visible (UV-Vis) Spectroscopy of Potassium Permanganate
E N D
Presentation Transcript
Ultraviolet/Visible (UV-Vis) Spectroscopy of Potassium Permanganate By Robert Bohman November 4th 2006 CH EN 4903
Importance to industry • Potassium Permanganate is used to kill bacteria in reclaimed water • Use UV-Vis to ensure that the concentration of Potassium Permanganate is at acceptable limit
Overview • Theory • Light Absorption Spectrum • Experimental Procedure • Results • Conclusion • Q & A
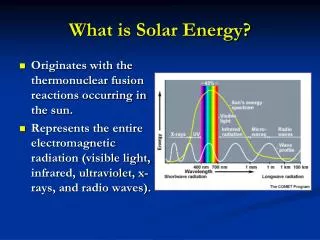
Properties of Light1 • c = λν c = speed of light in vacuum (2.998 x 108 m/s) λ = wavelength (m) v = frequency (Hz) • E = hc/ λ = hcv` h = Planck’s constant (6.626 x 10-34 J•s) v` = wavenumber (m-1)
Understanding Beer’s Law2 • Transmittance T = P/P0 Schematic of Single-Beam Spectrophotometer, P0 is the irradiance entering sample, P is the irradiance leaving sample, and b is pathlength2 P = irradiance (energy per unit area of light beam)
Understanding Beer’s Law3 • Absorbance A = log (P/P0) = -log (T) • Beer’s Law A = εbc ε = molar absorptivity (M-1 cm-1) b = pathlength (cm) c = concentration (M)
Detecting Potassium Permanganate • Potassium permanganate (KMn04) in solution is purple / violet color meaning maximum absorption should be at 500 – 550 nm • Prepared 5 known concentrations of KMnO4: 1ppm, 20ppm, 40ppm, 60ppm, 80ppm
Detecting Potassium Permanganate • Calibration Standards measured first on a Perkins-Elmer Lambda 35 over entire UV-Vis region to determine max absorption • KMnO4 absorbed best at ≈ 520 nm • A Bausch & Lomb Spectronic 21 was used to make all measurements
UV-Vis Absorbance Readings for Potassium Permanganate at 520 nm
Calibration Curve for KMnO4 using UV-Vis Spectroscopy, Absorption vs. Concentration
Determination of Unknown Concentration of KMnO4 • Used cuvette of 1cm length • ε = slope of line = 0.029 ppm-1 cm-1 • Unknown #4 concentration found using c = A/0.029 • 36 ppm = 0.462 %A / 0.029 ppm-1
Error Analysis • Used 10.00 ± 0.05mL volumetric pipette to make all solutions • Measured density of water with: = (999.8392 + 16.945176t – 7.9870401*10-3t2 – 46.170461*10-6t3 + 105.56302*10-9t4 – 280.54253*10-12t5)/(1 + 16.879850*10-3t)5 • H2O = 0.997883 g/mL at 21.5°C • Measured accuracy of scale to be 0.0005g
Error in Unknown • Errors determined graphically from calibration curve • A = ± 0.01% • Concentration = ± 1.00 ppm • Final concentration of Unknown #4 was 36 ± 1.00 ppm
Conclusion • How accurate are results? Can be determined by R2 value for slope of calibration curve. For this example R2 = 0.999
Conclusion • Use Beer’s law to determine concentration of unknown concentration • Find the molar absorptivity through the slope of calibration curve • Determined ε = 0.029 ppm-1 cm-1 • Determined Unknown #4 concentration to be 36 ± 1.00 ppm
Special Thanks To Triston Thorpe and Jason Judkins in helping with the experiment
References • Harris, Daniel C. Sixth Edition Quantitative Chemical Analysis. Pg. 408-409. New York: W.H. Freeman and Company, 2003. • Harris, Daniel C. Sixth Edition Quantitative Chemical Analysis. Pg. 410. New York: W.H. Freeman and Company, 2003. • Harris, Daniel C. Sixth Edition Quantitative Chemical Analysis. Pg. 411-412. New York: W.H. Freeman and Company, 2003. • Harris, Daniel C. Sixth Edition Quantitative Chemical Analysis. Pg. 413. New York: W.H. Freeman and Company, 2003. 5. CRC Handbook of Chemistry and Physics. Pg. F-6. Cleveland, Ohio: The Chemical Rubber Co., 1968.