Enthalpy
200 likes | 419 Views
Click on buttons to go forward or back. Question mark buttons go to answers. Enthalpy. By: Veronica Mendez and Quinn Shollenberger. ONLY CLICK ON THE QUESTION MARKS AND/OR ARROW. Lost?.

Enthalpy
E N D
Presentation Transcript
Click on buttons to go forward or back Question mark buttons go to answers Enthalpy By: Veronica Mendez and Quinn Shollenberger ONLY CLICK ON THE QUESTION MARKS AND/OR ARROW
Lost? If at any time you are lost during the tutorial, right click and a menu should come up. Click on Go To Slide and select the slide you want or go back to the first slide.
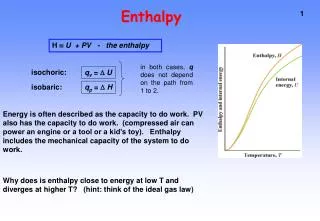
Definition • Enthalpy- the heat content of a system at constant pressure. • Enthalpy- ∆ H
Equation #1 • ∆H = nCp∆T • n is moles • Cp is specific heat capacity (usually a given value) • ∆T is the change in temperature
Equation #2 • ∆H =∑ ∆Hp - ∑ ∆Hr • ∑ ∆Hp is the summation of the products • ∑ ∆Hr is the summation of the reactants
Example problem • Find ΔH of the reaction. • 1CH4 + 2O2 --> 1CO2 + 2H2O • Mole Ratio is 1:2:1:2 because the coefficient in front of CH4 is 1; coefficient in front of O2 is 2; coefficient in front of CO2 is 1; coefficient in front of H2O is 2.
Example problem: Set up • ΔH = [∑(ΔHCO2 + ΔHH2O)] – [∑(ΔHCH4 + ΔHO2)]
Example Problem: Plugging in the numbers • ΔH = [(-393.5KJ/m X 1m) + (-286KJ/m X 2m)] – [(-74.8KJ/m X 1m) + 0][(-393.5 KJ) + (-572 KJ)] –(-74.8 KJ) • ΔH = -890.7 KJ (exothermic)
Example Problem: Further explanations • The numbers from the equation: ΔH = [(-393.5KJ/m X 1m) + (-286KJ/m X 2m)] – [(-74.8KJ/m X 1m) + 0][(-393.5 KJ) + (-572 KJ)] –(-74.8 KJ) can be found on any Chemistry reference source. This includes CP values.
Reminder • Do not forget to cross out your moles • Be careful with positive and negative signs
Quiz question 1 • Calculate ΔH at 25°C for: Na2O (s) + H2O (g) NaOH (s)
Quiz question 2 • If 15.0 g of Gold is heated from 16.1°C to 49.3°C, what is the heat absorbed by the gold?
Quiz question 3 • When 4.4 Kg of NaCl cools from 67.2°C to 25.0°C, how much heat is gained by the surroundings from the NaCl? (Cp NaCl 50.5 J°C-1M-1)
Quiz Question 4 • Determine the standard molar heat of combustion (ΔH ) id Methanol, CH3OH, when it is burned. Use H20(g)
Explanation to Quiz question 1 • Steps: • Balance equation: Na2O+H2O 2NaOH • [(- 427KJ/m)(2m)]-[(242KJ/m)(1m) + (- 415.9KJ/m)(1m)] • ΔH = -196.1KJ
Explanation to Quiz question 2 • ∆H = nCp∆T • 15.0 g(1m/ 197g)= 0.07614m • (0.07614m)(25.4J/m°C)(33.2°C) • ∆H = 64.2 J
Explanation to Quiz Question 3 • ∆H = nCp∆T • Convert kg into g • 4400 g (1m/58.5g) = 75.2m • (75.2m)(50.5J/m°C)(42.2 °C) • ∆H = 1.60 x 10 J
Explanation to Quiz question 4 • Combustion reaction and balance the equation 2CH3OH + 3O2 2CO2 + 4H2O
Explanation for Quiz question 4 cont… • [(-393.5KJ/m)(2m)+(242KJ/m)(4m)] – [0+(-239KJ/m)(2m)] • ∆H = 1277 KJ