Oxidative Phosphorylation
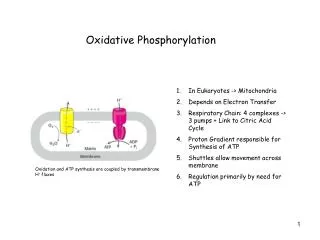
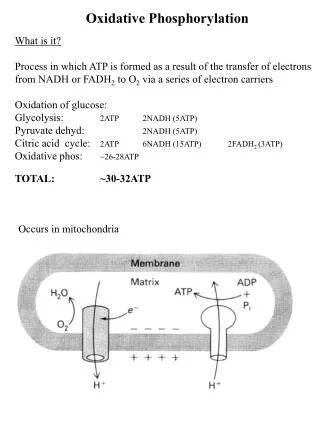
Oxidative Phosphorylation. General Considerations. How do we define oxidative phosphorylation? formation of ATP using the energy released by the transfer of electrons from NADH and FADH 2 through a series of electron carriers What couples the formation of ATP to the transfer of electrons?

Oxidative Phosphorylation
E N D
Presentation Transcript
General Considerations • How do we define oxidative phosphorylation? • formation of ATP using the energy released by the transfer of electrons from NADH and FADH2 through a series of electron carriers • What couples the formation of ATP to the transfer of electrons? • a proton gradient
General Considerations • Where in the cell does oxidative phosphorylation take place? • inner mitocondrial membrane • What do we know about the mitocondrial membranes? • outer membrane – reasonably permeable • contains porins – VDAC • inner membrane – relatively impermeable
Origin of Mitocondria • What is the believed origin of mitocondria? • endosymbiosis • What evidence supports this idea? • mitcondrial DNA • machinery for transcription and translation • similarity of genome to bacteria
Redox Potentials and Free Energy Changes • How does one determine the redox potential of a substance? standard reference half-cell sample half-cell
Redox Potentials and Free Energy Changes • What is the relationship between change in redox potential and change in free energy? • G01 = -nF E10 • n = number of electrons transferred • F =faraday (constant, 23.06 kcal/mole/volt) • Can calculate free energy change from reduction potentials of the reactants • By knowing the electron transfer potential of NADH relative to O2 one can calculate the amount of free energy released when O2 is reduced by NADH.
Redox Potentials and Free Energy Changes • One can also quantify the energy associated with a proton gradient. • G = RTln(c2/c1) + ZF V • c2 = concentration on one side of membrane • c1 = concenetration on other side of membrane • Z = electrical charge of transported material • F = Faraday constant (23.06 kcal/mole/volt)
Electron Transport • What determines the rate of electron transport? • distance between donor and acceptor
Electron Transport • driving force or free energy change
Electron Transport • What are the complexes making up the respiratory chain? • three proton pumps • one link to citric acid cycle
Electron Transport • What is the role of ubiquinone or coenzyme Q?
Electron Transport • What happens to the electrons from NADH? • enter ETS at NADH-Q oxidoreductase
Electron Transport • Initial step is transfer of electrons to FMN a prosthetic group of the enzyme
Electron Transport • Electrons are then transferred to iron-sulfur clusters another prosthetic group
Electron Transport • Electrons from clusters transferred to coenzyme Q • as a result of electron transfer four protons are pumped out of mitocondrial matrix • Reaction summarized: • NADH + Q + 5H+matrix NAD+ + QH2 + 4H+cytosol
Electron Transport • Coenzyme Q also serves as entry point for electrons from FADH2 from oxidation of succinate • succinate-Q reductase complex • inner mitocondrial membrane • FADH2 transfers electrons to iron-sulfur clusters then to Q • no protons are pumped
Electron Transport • Q-cytochrome c oxidoreductase catalyzes the transfer of electrons from Q to cytochrome c • What is a cytochrome? • electron transferring protein with heme prosthetic group • transfers only electrons • iron in heme goes between Fe+2 and Fe+3
Electron Transport • Q-cytochrome c oxidoreductase contains 3 hemes and a iron-sulfur cluster
Electron Transport • What is the Q cycle? • mechanism of coupling of electron transfer from Q to cytochrome c to proton transport
Electron Transport • What is the function of cytochrome c oxidase? • reduction of oxygen to water • What are the major prosthetic groups of this complex? • CuA/CuA • heme a • heme a3-Cub
Cytochrome c Oxidase • Mechanism of action
Cytochrome c Oxidase • cytochrome c oxidase pumps four additional protons from matrix for a total of eight protons removed from matrix
Electron Transport • Toxic derivatives of molecular oxygen may be formed by partial reduction O2 O2_ O2_2 superoxide anion peroxide
Electron Transport • How does the cell protect itself against these reactive oxygen species? • makes use of superoxide dismutase and catalase • 2O2_ + 2H+ O 2 + H2O2 • 2H2O2 O2 + 2H2O
ATP Synthesis • What is the chemiosmotic hypothesis? • ATP synthesis and electron transport are coupled by proton gradient across mitocondrial membrane
ATP Synthesis • What is ATP synthase and what do we know about its structure? • consists of F1 and F0 • F1 has 5 types of polypeptide chains • 3,3,,, • F0 contains proton channel • 10-14 c subunits • a,b2 subunits
ATP Synthesis • How is ATP synthesized?
ATP Synthesis • What is the role of the proton gradient in ATP synthesis? • part of binding-change mechansm • 3 subunits promote ADP & P binding, ATP synthesis, ATP release
ATP Synthesis • How does proton flow through F0 drive the rotation of the subunit? • each c subunits consists of 2 helices with one helix containing an aspartic acid residue • a subunit contains two proton half channels
ATP Synthesis • Proton enters half-channel, neutralizes charge on aspartate • c can rotate clockwise • proton can move into matrix
ATP Synthesis • Since c ring is linked to and subunits, as c turns these subunits rotate • rotation protmotes synthesis of ATP via binding-change mechanism • each 3600 rotation of subunit leads to synthesis of 3 ATP’s • 10 protons generate 3 ATP’s • each ATP requires transport of about 3 protons
Mitocondrial Shuttles • Reoxidation of cytosolic NADH requires shuttle mechanism • glycerol 3-phosphate shuttle • found in muscle
Mitocondrial Shuttles • malate-aspartate shuttle • heart and liver
Mitocondrial Shuttles • What is an ATP-ADP translocase? • transport protein allowing ATP to exit mitocondrion and ADP to enter • result in moving one negative charge out of matrix • decreases proton motive force
Mitocondrial Shuttles • Other mitocondrial transport proteins act as shuttles
Regulation of Respiration • Energy formed from oxidation of glucose • 3 protons = 1 ATP • 1 proton used to move ATP • one pair of electons from NADH = 2.5 molecules of ATP
Regulation of Respiration • What controls rate of electron transport?
Regulation of Respiration • Oxidative phosphorylation can be inhibited by many substances
Regulation of Respiration • What are uncoupling agents? • transport protons across mitocondrial membrane
Regulation of Respiration • Does uncoupling serve any useful purpose? • body heat generation