Titrations 19.4
Titrations 19.4. Calculations involving neutralization reactions. Learning Objectives. Understand the neutralization reaction … strong acid and base reaction Concept of titration, end point, and equivalence point and how they relate to neutralization reaction

Titrations 19.4
E N D
Presentation Transcript
Titrations 19.4 Calculations involving neutralization reactions
Learning Objectives • Understand the neutralization reaction … strong acid and base reaction • Concept of titration, end point, and equivalence point and how they relate to neutralization reaction • How to solve titration problems … how to determine unknown acid or base concentration
Neutralization Reaction • Acid + Base Salt + Water • Called a neutralization reaction • Is a double replacement reaction • Requires use of an indicator to identify neutralization point • Strong acid + Strong base = neutral solution • If # of H+ = # of OH-
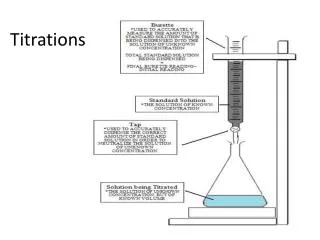
Titration • A titration is a reaction where a solution of known concentration (standard solution) is used to identify a solution of unknown concentration
Practical Examples of Titrations • Determining blood glucose levels. • Determination of medical proportions in a IV drip. • Amount of salt and sugar in food. • Level of acidity in wine and cheese. • Level of acidity in fresh water samples.
End Point • The end point of a titration is when the solution just barely changes color • The equivalence point is the point where the acid and base exactly react with one another • Ideally, the end point should be the same as the equivalence point
Titration Calculation In a titration, 27.4 ml of a 0.0154 M HCl solution is added to 20.0 mL sample of NaOH solution of unknown concentration. What is the molarity of the basic solution? HCl(aq) + NaOH(aq) H2O(l) + NaCl(aq) SA SB Neutralization First we need to figure moles of HCl used to neutralize NaOH in reaction. 0.0274 L x 0.0154 mol/L = 4.22 x 10-4 mol of HCl
Titration Calculation = 4.22 x 10-4 mol of HCl HCl(aq) + NaOH(aq) H2O(l) + NaCl(aq) = 4.22 x 10-4 mol of NaOH M of NaOH = 4.22 x 10-4 mol of NaOH .0200 L M of NaOH = .021 M
Titration Endpoint How can you tell when you have added enough NaOH?
Titration Calculation Shortcut • Objective: to find the M of the unknown acid or base. • Use a balanced chemical equation to determine the mole/mole ratio of acid to base. MaVana=MbVbnb • M = molarity • V = volume • n = # of H3O+ ions in the acid or OH- ions in the base. (if there are no OH- ions in the base, n = 1)
Cool Down • How many moles of sulfuric acid are required to completely neutralize 0.50 mol of sodium hydroxide? • How many moles of potassium hydroxide are needed to completely neutralize 1.56 mol of phosphoric acid?