T c
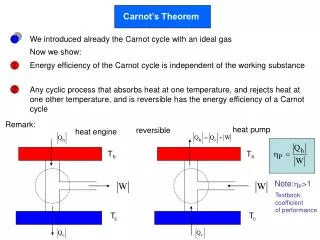
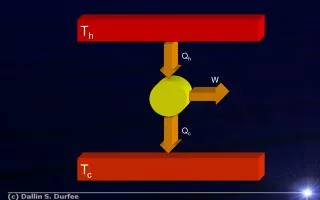
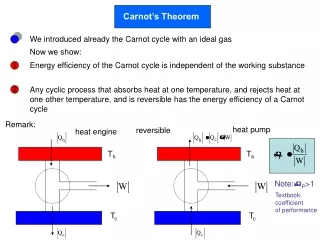
Carnot’s Theorem. heat pump. heat engine. T h. T h. T c. T c. We introduced already the Carnot cycle with an ideal gas. Now we show:. Energy efficiency of the Carnot cycle is independent of the working substance. 1.

T c
E N D
Presentation Transcript
Carnot’s Theorem heat pump heat engine Th Th Tc Tc We introduced already the Carnot cycle with an ideal gas Now we show: Energy efficiency of the Carnot cycle is independent of the working substance 1 Any cyclic process that absorbs heat at one temperature, and rejects heat at one other temperature, and is reversible has the energy efficiency of a Carnot cycle 2 Remark: reversible Note:P>1 Textbook: coefficient of performance
We can design the engine X such that Let’s combine a fictitious heat engine X with with a heat pump realized by a reversed Carnot cycle heat engine X heat pump Th Th X C Tc Tc with Let’s calculate
heat pump heat engine X with > Th Th >0 X C Tc Tc We can design the engine X such that If X would be a Carnot engine it would produce the work However:
False Let X be the heat pump and the Carnot cycle operate like an engine False Any cyclic process that absorbs heat at one temperature, and rejects heat at one other temperature, and is reversible has the energy efficiency of a Carnot cycle. 2 Energy efficiency of the Carnot cycle is independent of the working substance. 1 Why Because: X can be a Carnot engine with arbitrary working substance
heat engine X heat pump Th Th X C Tc Tc Carnot’s theorem: No engine operating between two heat reservoirs is more efficient than a Carnot engine. Again we create a composite device Proof uses similar idea as before: We can design the engine X such that operates the Carnot refrigerator
Rudolf Clausius (2.1.1822 -24.8.1888) Let’s assume that Note: this time engine X can be also work irreversible like a real engine does > My statement holds man Heat transferred from the cooler to the hotter reservoir without doing work on the surrounding Violation of the Clausius statement
Applications of Carnot Cycles We stated: Any cyclic process that absorbs heat at one temperature, and rejects heat at one other temperature, and is reversible has the energy efficiency of a Carnot cycle. - gas turbine - Otto cycle Why did we calculate energy efficiencies for Because: they are not 2-temperature devices, but accept and reject heat at a range of temperatures Energy efficiency not given by the Carnot formula But: It is interesting to compare the maximum possible efficiency of a Carnot cycle with the efficiency of engineering cycles with the same maximum and minimum temperatures
Consider the gas turbine again (Brayton or Joule cycle) Efficiency 2 3 Heating the gas (by burning the fuel) 2 3 Ph cooling Maximum temperature: 4 1 @ : T3 3 adiabates Minimum temperature: 4 : @ T1 Pl 1 1 with
Efficiency of corresponding Carnot Cycle With Unfortunately: Gas turbine useless in the limit Because: 0 Heat taken per cycle Work done per cycle 0
Absolute Temperature A temperature scale is an absolute temperature scale if and only if , where and are the heats exchanged by a Carnot cycle operating between reservoirs at temperatures T1 and T2. T2 T1 We showed: Energy efficiency of the Carnot cycle is independent of the working substance. Definition of temperature independent of any material property Measurement of Temperature ratio
As discussed earlier, unique temperature scale requires fixed point or Tfix =Ttripel=273.16K Kelvin-scale: It turns out: empirical gas temperature proportional to thermodynamic Temperature T Why Because: Calculation of efficiency of Carnot cycle based on yields a=1 With
From definition of thermodynamic temperature If any absolute temperature is positive all other absolute temperatures are positive there is an absolute zero of thermodynamic temperature when the rejected heat 0 however T=0 can never be reached, because this would violate the Kelvin statement