Particle size
260 likes | 477 Views
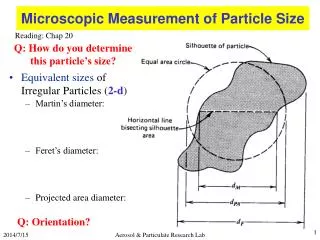
Particle size. Ions molecular clusters nanocrystals colloids bulk minerals Small particles can have a significant % of molecules at their surface Thermodynamics are different (surface free energy) Surface area per mass is huge and charged through interaction with water

Particle size
E N D
Presentation Transcript
Particle size • Ions molecular clusters nanocrystals colloids bulk minerals • Small particles can have a significant % of molecules at their surface • Thermodynamics are different (surface free energy) • Surface area per mass is huge and charged through interaction with water • Sorption of ions to these surfaces can be critical part of contaminant mobility
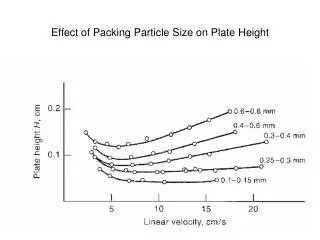
Surface area • Selected mineral groups often occur as colloids / nanoparticles: • FeOOH SA up to 500 m2/g, site density 2-20/nm2 • Al(OH)3 SA up to 150 m2/g, site density 2-12/nm2 • MnOOH SA hundreds m2/g, site density 2-20/nm2 • SiO2 SA 0.1 – 300 m2/g, site density 4-12/nm2 • Clays SA 10-1000 m2/g, site density 1-5/nm2 • Organics SA up 1300 m2/g, site density 2/nm2
DEFINITIONS • Sorption - removal of solutes from solution onto mineral surfaces. • Sorbate - the species removed from solution. • Sorbent - the solid onto which solution species are sorbed. • Three types of sorption: • Adsorption - solutes held at the mineral surface as a hydrated species. • Absorption - solute incorporated into the mineral structure at the surface. • Ion exchange - when an ion becomes sorbed to a surface by changing places with a similarly charged ion previously residing on the sorbent.
Mineral Surfaces • Minerals which are precipitated can also interact with other molecules and ions at the surface • Attraction between a particular mineral surface and an ion or molecule due to: • Electrostatic interaction (unlike charges attract) • Hydrophobic/hydrophilic interactions • Specific bonding reactions at the surface
Inner Sphere and Outer Sphere • Outer Sphere surface complex ion remains bounded to the hydration shell so it does not bind directly to the surface, attraction is purely electrostatic • Inner Sphere surface complex ion bonds to a specific site on the surface, this ignores overall electrostatic interaction with bulk surface (i.e. a cation could bind to a mineral below the mineral pHzpc)
Charged Surfaces • Mineral surface has exposed ions that have an unsatisfied bond in water, they bond to H2O, many of which rearrange and shed a H+ • ≡S- + H2O ≡S—H2O ≡S-OH + H+ OH OH OH2 H+ OH OH OH H+ OH
Surfaces as acid-base reactants • The surface ‘SITE’ acts as an amphoteric substance it can take on an extra H+ or lose the one it has to develop charge • ≡S-O- + H+↔ ≡S-OH ↔ ≡S-OH2+ • The # of sites on a surface that are +, -, or 0 charge is a function of pH • pHzpc is the pH where the + sites = - sites = 0 sites and the surface charge is nil OH OH2+ O- OH O- OH OH2+
pHzpc • Zero Point of Charge, A.k.a: Zero Point of Net Proton Charge (pHZPNPC) or the Isoelectric Point (IEP) • Measured by titration curves (pHzpc similar to pKa…) or electrophoretic mobility (tendency of the solids to migrate towards a positively charged plate) • Below pHzpc more sites are protonated net + charge • Above pHzpc more sites are unprotonated net - charge
POINT OF ZERO CHARGE CAUSED BY BINDING OR DISSOCIATION OF PROTONS
ION EXCHANGE REACTIONS • Ions adsorbed by outer-sphere complexation and diffuse-ion adsorption are readily exchangeable with similar ions in solution. • Cation exchange capacity: The concentration of ions, in meq/100 g soil, that can be displaced from the soil by ions in solution. • Also anion exchange capacity for positively charged surfaces
ION EXCHANGE REACTIONS • Exchange reactions involving common, major cations are treated as equilibrium processes. • The general form of a cation exchange reaction is: nAm+ + mBX mBn+ + nAX • The equilibrium constant for this reaction is given by:
SORPTION ISOTHERMS - I • The capacity for a soil or mineral to adsorb a solute from solution can be determined by an experiment called a batch test. • In a batch test, a known mass of solid (Sm) is mixed and allowed to equilibrate with a known volume of solution (V) containing a known initial concentration of a solute (Ci). The solid and solution are then separated and the concentration (C) of the solute remaining is measured. The difference Ci - C is the concentration of solute adsorbed.
Kd • Descriptions of how solutes stick to the surface • What would the ‘real’ behavior be you think?? Kd
SORPTION ISOTHERMS - II • The mass of solute adsorbed per mass of dry solid is given by where S m is the mass of the solid. • The test is repeated at constant temperature but varying values of Ci. A relationship between Cand S can be graphed. Such a graph is known as an isotherm and is usually non-linear. • Two common equations describing isotherms are the Freundlich and Langmuir isotherms.
FREUNDLICH ISOTHERM The Freundlich isotherm is described by where K is the partition coefficient and n 1. When n < 1, the plot is concave with respect to the C axis. When n = 1, the plot is linear. In this case, K is called the distribution coefficient (Kd ).
LANGMUIR ISOTHERM The Langmuir isotherm describes the situation where the number of sorption sites is limited, so a maximum sorptive capacity (S max) is reached. The governing equation for Langmuir isotherms is:
Sorption of organic contaminants • Organic contaminants in water are often sorbed to the solid organic fractions present in soils and sediments • Natural dissolved organics (primarily humic and fulvic acids) are ionic and have a Koc close to zero • Solubility is correlated to Koc for most organics
Measuring organic sorption properties • Kow, the octanol-water partition coefficient is measured in batches with ½ water and ½ octanol – measures proportion of added organic which partitions to the hydrophobic organic material • Empirical relation back to Koc: log Koc = 1.377 + 0.544 log Kow
ADSORPTION OF METAL CATIONS - I • In a natural solution, many metal cations compete for the available sorption sites. • Experiments show some metals have greater adsorption affinities than others. What factors determine this selectivity? • Ionic potential: defined as the charge over the radius (Z/r). • Cations with low Z/r release their waters of hydration more easily and can form inner-sphere surface complexes.
ADSORPTION OF METAL CATIONS - II • Many isovalent series cations exhibit decreasing sorption affinity with decreasing ionic radius: Cs+ > Rb+ > K+ > Na+ > Li+ Ba2+ > Sr2+ > Ca2+ > Mg2+ Hg2+ > Cd2+ > Zn2+ • For transition metals, electron configuration becomes more important than ionic radius: Cu2+ > Ni2+ > Co2+ > Fe2+ > Mn2+
ADSORPTION OF METAL CATIONS - III • For variable-charge sorbents, the fraction of cations sorbed increases with increasing pH. • For each individual ion, the degree of sorption increases rapidly over a narrow pH range (the adsorption edge).
Exchange reaction and site competition • For a reaction: A + BX = B + AX • Plot of log[B]/[A] vs. log[BX]/[AX] yield n and K • When n and K=1 Donnan exchange, exhange only dependent on valence, bonding strictly electrostatic • When n=1 and K≠1 Simple ion exchange, dependent on valence AND size, bonding strictly electrostatic • When n≠1 and K≠1 Power exchange, no physical description (complicated beyond the model) and unbalanced stoichiometry
Electrostatic models • Combining electrostatic interactions and specific complexation using mechanistic and atomic ideas about the surface yield models to describe specific sorption behavior