Lipids
Lipids. What are Lipids?. Class of biomolecules that are all water insoluble No defining characteristic except that all of them share the same trait: non-polar Most common are fatty acids and esters of fatty acids Functions Energy Storage Cell membrane Thermal blanket and cushion

Lipids
E N D
Presentation Transcript
What are Lipids? • Class of biomolecules that are all water insoluble • No defining characteristic except that all of them share the same trait: non-polar • Most common are fatty acids and esters of fatty acids • Functions • Energy Storage • Cell membrane • Thermal blanket and cushion • Precursors of hormones
Fatty Acids • Fatty acids consist of a hydrocarbon chain with a carboxylic acid at one end. • A 16-C fatty acid: CH3(CH2)14-COO- Non-polarpolar • General Structure:
Fatty Acids • Double bonds in fatty acids usually have the cis configuration. • Most naturally occurring fatty acids have an even number of carbon atoms. • IUPAC naming: • COOH carbon as C1 • Farthest carbon is ω • Total carbons/# double bonds/Δdoublebond position
Fatty Acids • Some fatty acids and their common names: • 14:0myristic acid; 16:0 palmitic acid; 18:0 stearic acid; • 18:1 cisD9 oleic acid • 18:2 cisD9,12 linoleic acid • 18:3 cisD9,12,15a-linonenic acid • 20:4 cisD5,8,11,14arachidonic acid • 20:5 cisD5,8,11,14,17eicosapentaenoic acid (an omega-3) • Essential fatty acids • ALA- ω-3 FA precursor for EPA and DHA • LA – ω-6 FA precursor of AA
Fatty Acid Chemistry • Peroxidation • a non-enzymatic reaction catalyzed by oxygen • may occur in tissues or in foods (spoilage) • the hydroperoxide formed is very reactive and leads to the formation of free radicals which oxidize protein and/or DNA (causes aging and cancer) • principle is also used in drying oils (linseed, tung, walnut) to form hard films
Drying Oils: Application of Peroxidation • based on the ease of autoxidation and polymerization of oils (important in paints and varnishes) • the more unsaturation in the oil, the more likely the “drying” process • Non-drying oils: • Castor, olive, peanut, rapeseed oils • Semi-drying oils • Corn, sesame, cottonseed oils • Drying oils • Soybean, sunflower, hemp, linseed, tung, oiticica oils
Fatty Acid Chemistry • Transesterification • Acid-catalyzed exchange betweein R groups of esters • Frequently-used in polyester, methanolysis and biodiesel industries
Fatty Acid Chemistry • Hydrogenation • Reduces double bonds on unsaturated fatty acids • Prevents oils from going rancid
Fatty Acid Chemistry • Saponification • Base hydrolysis of triglycerides to produce fatty acid salts • High saponification number, shorter chain length
Soaps • Types of soaps: • Sodium soap – ordinary hard soap • Potassium soap – soft soap (shaving soaps are potassium soaps of coconut and palm oils) • Castile soap – sodium soap of olive oil • Green soap – mixture of sodium and potassium linseed oil • Transparent soap – contains sucrose • Floating soap – contains air • Calcium and magnesium soaps are very poorly water soluble (hard water contains calcium and magnesium salts –these insolubilize soaps)
Fatty Acid Chemistry • Iodine Number • measures the degree of unsaturation in a given amount of fat or oil • the iodine number is the number of grams of iodine absorbed by 100 grams of fat • Cottonseed oil: 103 –111 • Olive oil: 79 – 88 • Linseed oil: 175 –202 • frequently used to determine adulteration of commercial lots of oils
Fatty Acid Chemistry • Acetyl Number • the number of milligrams of KOH needed to neutralize the acetic acid of 1 g of acetylated fat
Fatty Acid Chemistry • Reichert-Meisl Number • measures the amount of volatile fatty acids (low MW and water soluble Fas) • the R-M number is the number of milliliters of 0.1N alkali required to neutralize the soluble fatty acids distilled from 5 Gm of fat • butter fat has a high R-M number • Indicator of non-fat compounds in edible fats, or volatile fatty acids
Fats and Oils • fats are solids or semi solids • oils are liquids • melting points and boiling points are not usually sharp (most fats/oils are mixtures) • when shaken with water, oils tend to emulsify • pure fats and oils are colorless and odorless (color and odor is always a result of contaminants) – i.e. butter (bacteria give flavor, carotene gives color)
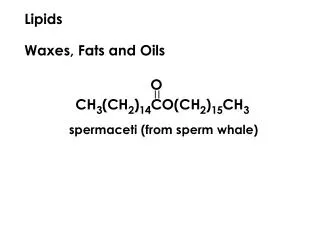
Waxes • simple esters of fatty acids (usually saturated with long chain monohydric alcohols) • Beeswax – also includes some free alcohol and fatty acids • Spermaceti – contains cetylpalmitate (from whale oil) –useful for pharmaceuticals (creams/ointments; tableting and granulation) • Carnauba wax – from a palm tree from brazil – a hard wax used on cars and boats
Spermaceti source Carnauba wax source Bee’s wax
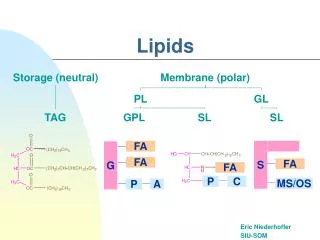
Glycerides • Function • Storage of energy • Cushions
Triglycerides • Esters of fatty acids and glycerol • Constituent of fats and oils
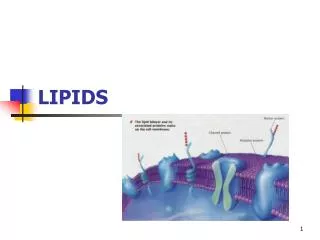
Phosphoglycerides • aka glycerophospholipids • Major constituent of cell membranes • Structure • Glycerol backbone with esterifies fatty acids at C1 and C2 • Fatty acids are usually non-identical • Phosphate group attached to C1 • A polar group is usually esterified to Pi (usually serine, choline, ethanolamine, glycerol, or inositol
Phosphoglycerides • Structure=Function • Polar region – glycerol, phosphate group, X • Nonpolar region – fatty acid • Amphiphatic character • Ideal for forming bilayers
Phosphatidyl inositol • Inositol as a polar head group • Has roles in cell signaling as well as a cell membrane component
Ether glycerophospholipids • Possess an ether linkage instead of an acyl group at the C-1 position of glycerol • PAF ( platelet activating factor) - a potent mediator in inflammation, allergic response and in shock (also responsible for asthma-like symptom • The ether linkage is stable in either acid or base • Plasmalogens: cisa,b-unsaturated ethers • The alpha/beta unsaturated ether can be hydrolyzed more easily
Sphingolipids • derivatives of the lipid sphingosine, which has a long hydrocarbon tail, and a polar domain that includes an amino group • Like phosphoglycerides, are also constituents of cell membranes • Can be reversibly phosphorylated and utilized as signaling molecule
Sphingolipid Derivatives • Ceramide- amino group of sphingosineforms an amide bond with a fatty acid carboxyl • Sphingomyelinhas a phosphocholine or phosphethanolamine head group. • common constituent of plasma membranes • similar in size and shape to the glycerophospholipidphosphatidyl choline
Sphingolipid Derivatives • Cerebrosidea sphingolipid (ceramide) with a monosaccharide such as glucose or galactose as polar head group. • Ganglioside– a ceramide with polar head group that is a complex oligosaccharide, including the acidic sugar derivative sialicacid • Both are collectively known as glycosphingolipidsand are commonly found on the outer leaflet of the cell membrane
Ganglioside • Cerebrosides • One sugar molecule • Galactocerebroside – in neuronal membranes • Glucocerebrosides – elsewhere in the body • Sulfatides or sulfogalactocerebrosides • A sulfuric acid ester of galactocerebroside • Globosides: ceramide oligosaccharides • Lactosylceramide • 2 sugars ( eg. lactose) • Gangliosides • Have a more complex oligosaccharide attached • Biological functions: cell-cell recognition; receptors for hormones
Cardiolipids • A polyglycerol phospholipid; makes up 15% of total lipid-phosphorus content of the myocardium – associated with the cell membrane • Cardiolipids are antigenic and as such are used in serologic test for syphilis (Wasserman test)
Sulfolipids • also called sulfatides or cerebroside sulfates • contained in brain lipids • sulfate esters of cerebrosides • present in low levels in liver, lung, kidney, spleen, skeletal muscle and heart • function is not established
Lipid Storage Diease • a group of inherited metabolic disorders in which harmful amounts of fatty materials (lipids) accumulate in various tissues and cells in the body • also known as sphingolipidoses • genetically acquired • due to the deficiency or absence of a catabolic enzyme • Tay Sachs disease • Gaucher’s disease • Niemann-Pick disease • Fabry’s disease
Cholesterol • an important constituent of cell membranes, has a rigid ring system and a short branched hydrocarbon tail. • serves as a component of membranes of cells (increases or moderates membrane fluidity) • precursor to steroid hormones and bile acids • storage and transport –cholesterol esters • Cholesterol is largely hydrophobic. • But it has one polar group, a hydroxyl, making it amphipathic.
Eicosanoids • Includes prostaglandins and related compounds • Derivatives of arachidonic acid • Serves as local hormones and intercellular signaling • Key hormones in inflammation response
Eicosanoids • Types of Eicosanoids • Prostaglandins • Prostacyclins • Thromboxanes • Leukotrienes • epoxyeicosatrienoicacids. • Biological Functions • Inflammation • Fever • regulation of blood pressure • blood clotting • immune system modulation • control of reproductive processes and tissue growth • regulation of the sleep/wake cycle
Terpenes • simple lipids, but lack fatty acid component • formed by the combination of 2 or more molecules of 2-methyl-1,3-butadiene (isoprene) • monoterpene (C-10) – made up of 2 isoprene units • sesquiterpene (C-15) – made up of 3 isoprene units • diterpene (C-20) – made up of 4 isoprene units
Terpenes • tetraterpenes (C-40) are not as common as mono, di, and triterpenes • include the carotenoids such as beta-carotene (precursor of vitamin A) and lycopene found in tomatoes • usually colorful compounds due to highly conjugated system • polyisoprenoids or polyprenols consist of numerous isoprene adducts (8 – 22) • examples include dolichol phosphate, undecaprenyl alcohol (bactoprenol) and the side chains of vitamins K, vitamin E and coenzyme Q
Lipid-linked proteins • Proteins that have lipids covalently bonded to them • Usually found in cell membranes • 3 types are most common: • Prenylated proteins • Farnesylated proteins (C15 isoprene unit) • Geranylgeranylated proteins (C20 isoprene unit) • Fatty acylated proteins • Myristoylated proteins (C14) • Palmitoylated proteins (C16) • glycosylphosphatidylinositol-linked proteins (GPI-linked proteins) • occur in all eukaryotes, but are particularly abundant in parasitic protozoa • located only on the exterior surface of the plasma membrane