ELECTROCHEMISTRY
ELECTROCHEMISTRY. References: Engg.Chemistry by Jain and Jain Engg.Chemistry by Dr. R.V.Gadag and Dr. A.Nithyananda Shetty Principles of Physical Chemistry by Puri and Sharma.

ELECTROCHEMISTRY
E N D
Presentation Transcript
References: • Engg.Chemistry by Jain and Jain • Engg.Chemistry by Dr. R.V.Gadag and Dr. A.Nithyananda Shetty • Principles of Physical Chemistry by Puri and Sharma
Electrochemistry is a branch of chemistry which deals with the properties and behavior of electrolytes in solution and inter-conversion of chemical and electrical energies.
An electrochemical cell can be defined as a single arrangement of two electrodes in one or two electrolytes which converts chemical energy into electrical energy or electrical energy into chemical energy. • It can be classified into two types: • Galvanic Cells. • Electrolytic Cells.
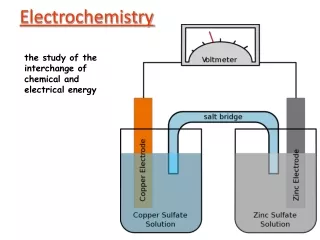
Galvanic Cells: A galvanic cell is an electrochemical cell that produces electricity as a result of the spontaneous reaction occurring inside it. Galvanic cell generally consists of two electrodes dipped in two electrolyte solutions which are separated by a porous diaphragm or connected through a salt bridge. To illustrate a typical galvanic cell, we can take the example of Daniel cell.
At the anode: Zn → Zn 2+ + 2e- At the cathode: Cu 2+ + 2e- → Cu Net reaction: Zn(s)+Cu 2+ (aq)→ Zn 2+ (aq)+ Cu(s)
ELECTROLYTIC CELL An electrolytic cell is an electro –chemical cell in which a non- spontaneous reaction is driven by an external source of current although the cathode is still the site of reduction, it is now the negative electrode whereas the anode, the site of oxidation is positive.
Representation of galvanic cell. • Anode Representation: Zn│Zn2+ or Zn ; Zn2+ Zn │ ZnSO4 (1M) or Zn ; ZnSO4 (1M) • Cathode Representation: Cu2+/Cu or Cu2+ ;Cu Cu2+(1M) ; Cu or CuSO4(1M)/Cu • Cell Representation: Zn │ ZnSO4 (1M)║ CuSO4(1M)/Cu
Liquid Junction Potential. • Difference between the electric potentials developed in the two solutions across their interface . Ej = Ø soln, R - Ø soln,L Eg: *Contact between two different electrolytes (ZnSO4/ CuSO4). *Contact between same electrolyte of different concentrations(0.1M HCl / 1.0 M HCl).
Salt Bridge. • The liquid junction potential can be reduced (to about 1 to 2 mV) by joining the electrolyte compartments through a salt bridge.
Function Of Salt Bridge. • It provides electrolytic contact between the two electrolyte solutions of a cell. • It avoids or at least reduces junction potential in galvanic cells containing two electrolyte solutions in contact.
Emf of a cell. • The difference of potential, which causes a current to flow from the electrode of higher potential to one of lower potential. Ecell = Ecathode- Eanode • The E Cell depends on: • the nature of the electrodes. • temperature. • concentration of the electrolyte solutions.
Standard emf of a cell(Eo cell) is defined as the emf of a cell when the reactants & products of the cell reaction are at unit concentration or unit activity, at 298 K and at 1 atmospheric pressure.
The emf cannot be measured accurately using a voltmeter : • As a part of the cell current is drawn,thereby causing a change in the emf. • As a part of the emf is used to overcome the internal resistance of the cell.
The emf of the cell Ex is proportional to the length AD. Ex α AD • The emf of the standard cell Es is proportional to the length AD1. Es α AD1 Ex ═ AD Es AD1 Ex = AD x Es AD1
Standard Cell. • It is one which is capable of giving constant and reproducible emf. • It has a negligible temperature coefficient of the emf. • The cell reaction should be reversible. • It should have no liquid junction potential. Eg: Weston Cadmium Cell. The emf of the cell is 1.0183 V at 293 K and 1.0181 V at 298 K.
Weston Cadmium Cell Sealed wax Cork Soturated solution of CdSO4.8/3H2O CdSO4.8/3H2O crystals Paste of Hg2SO4 Cd-Hg 12-14% Cd Mercury, Hg
Cell representation: Cd-Hg/Cd2+// Hg2SO4/Hg At the anode: Cd (s) → Cd2+ + 2e- At the cathode: Hg2SO4(s) + 2e- → 2 Hg (l)+ SO42-(aq) Cell reaction: Cd + Hg22+ → Cd2+ + 2Hg
Origin of single electrode potential. • Consider Zn(s)/ ZnSO4 Anodic process:Zn(s) → Zn2+(aq) Cathodic process: Zn2+(aq) → Zn(s) • At equilibrium: Zn(s) ↔Zn2+(aq) Metal has net negative charge and solution has equal positive charge leading to the formation of an Helmholtz electrical layer.
Single electrode potential. Electric layer on the metal has a potential Ø (M). Electric layer on the solution has a potential Ø (aq) • Electric potential difference between the electric double layer existing across the electrode /electrolyte interface of a single electrode or half cell.
Helmholtz double layer De-electronation Electronation
MEASUREMENT OF ELECTRODE POTENTIAL. • It is not possible to determine experimentally the potential of a single electrode. • It is only the difference of potentials between two electrodes that we can measure by combining them to give a complete cell. • By arbitrarily fixing the potential of reversible hydrogen electrode as zero it is possible to assign numerical values to potentials of the various other electrodes.
Sign Of Electrode Potential. • The electrode potential of an electrode: Is positive: If the electrode reaction is reduction when coupled with the standard hydrogen electrode Is negative: If the electrode reaction is oxidation when coupled with standard hydrogen electrode. According to latest accepted conventions, all single electrode potential values represent reduction tendency of electrodes.
when copper electrode is combined with SHE, copper electrode acts as cathode and undergoes reduction hydrogen electrode acts as anode. H2(g) → 2H+ +2e- (oxidation) Cu2+ +2e- → Cu (reduction) Hence electrode potential of copper is assigned a positive sign. Its standard electrode potential is 0.34 V.
When zinc is coupled with S.H.E. zinc electrode acts as anode and hydrogen electrode acts as cathode. Zn → Zn2+ +2e- 2H+ + 2e-→ H2. Hence, electrode potential of zinc is negative. The standard electrode potential of zinc electrode is -0.74 V.
Nernst Equation. • It is a quantitative relationship between electrode potential and concentration of the electrolyte species. • Consider a general redox reaction: Mn+(aq) + ne- → M(s) ----(1) We know that, ΔG =-nFE ----- (2) ΔGo=-nFEo-----(3) ΔG =ΔGo +RT ln K
ΔG =ΔGo +RT ln K ΔG =ΔGo +RT ln[M]/[Mn+]-----(4) -nFE= -nFEo + RT ln [M]/[Mn+]----(5) E= Eo – RT/nF ln 1/[Mn+]------(6) E=Eo- 2.303 RT/nF log 1/[Mn+]---(7) At 298K, E= Eo-0.0592/n log 1/[Mn+]-------(8)
problems • 1. A galvanic cell consists of copper plate immersed in 10 M solution of CuSO4 and iron plate immersed in 1M FeSO4 at 298K. If E0cell=0.78 V, write the cell reaction and calculate E.M.F. of the cell.
Solution: Cell reaction: Fe + Cu2+↔ Fe2+ + Cu ECell= E0Cell-0.0592/2 log [Fe2+ ]/[Cu2+] ECell= 0.78 + 0.0296 log 10/1 =0.8096V
Calculate E.M.F. of the zinc – silver cell at 25˚C when [Zn2+] = 1.0 M and [Ag+] = 10 M (E0cell=1.56V at 25˚C). Write the cell representation and cell reaction
Solution: • Cell representation • Zn/ Zn2+((1M)//Ag+(10M) /Ag • Cell reaction: Zn + 2Ag+↔ Zn2+ + 2Ag ECell= E0Cell-0.0592/2 log [Zn2+ ]/[Ag+]2 ECell= 1.56 + 0.0592 log 10/1.0 =1.6192 V
The emf of the cell Mg│ Mg 2+ (0.01M)║ Cu 2+ /Cu is measured to be 2.78 V at 298K. The standard eletrode potential of magnesium electrode is -2.37 V. Calculate the electrode potential of copper electrode
Cell reaction: Mg + Cu2+↔ Mg2+ + Cu E= Eo-0.0592/n log 1/[Mn+] EMg= EoMg-0.0592/2 log 1/[Mg2+] =-2.4291V Ecell=ECu-EMg 2.78 = ECu-[-2.429] ECu=2.78-2.429 =0.3509 V
The emf of the cell Cu│ Cu 2+ (0.02M)║ Ag+ /Ag is measured to be 0.46 V at 298K. The standard eletrode potential of copper electrode is 0.34 V. Calculate the electrode potential of silver. electrode
Energetics of Cell Reactions. • Net electrical work performed by the cell reaction of a galvanic cell: W= QE ------(1) Charge on 1mol electrons is F(96,500)Coulombs. When n electrons are involved in the cell reaction, the charge on n mole of electrons = nF
Q = nF Substituting for Q in eqn (1) W = nFE ----------(2) The cell does net work at the expense of • ΔG accompanying. ΔG = -nFE • ΔG = nFE
From Gibbs – Helmholtz equation. ΔG = ΔH + T [δ(ΔG)/ δT]P ------- (2) -nFE = ΔH – nFT (δ E/ δT)P ΔH = nFT (δ E/ δ T)P – nFE ΔH = nF[T(δ E/ δT)P –E] • We know that, [δ (∆G)/ δT]P = - ΔS ΔS = nF (δE/ δT)P
Problem: Emf of Weston Cadmium cell is 1.0183 V at 293 K and 1.0l81 V at 298 K. Calculate ∆G, ΔH and ΔS of the cell reaction at 298 K. Solution:- ∆G: ∆G = - n FE n = 2 for the cell reaction; F = 96,500 C E= 1.0181 V at 298 K ∆G = -2 x 96,500 x 1.0181 J = -196.5 KJ
∆H: ∆H = nF [ T (δE /δT)P – E] • (δE/δT)p = 1.0181 – 1.0183 / 298-293 = -0.0002 / 5 • = -0.00004VK-1 • T = 298 K • ∆H = 2 x 96,500 { 298 x (-0.00004) – 1.0181) • = -198. 8 KJ • ΔS: ΔS = nF (δE / δT) P • = 2 x 96,500 x (0.00004) = -7.72JK-1
Classification of Electrodes. • Gas electrode ( Hydrogen electrode). • Metal-metal insoluble salt (Calomel electrode). • Ion selective electrode.(Glass electrode).
Gas electrode. • It consists of gas bubbling over an inert metal wire or foil immersed in a solution containing ions of the gas. • Standard hydrogen electrode is the primary reference electrode, whose electrode potential at all temperature is taken as zero arbitrarily.
Representation: Pt,H2(g)/ H+ • Electrode reaction: H+ + e-1/2 H2(g) The electrode reaction is reversible as it can undergo either oxidation or reduction depending on the other half cell. • If the concentration of the H+ ions is 1M, pressure of H2 is 1atm at 298K it is called as standard hydrogen electrode (SHE).
Applications. • To determine electrode potential of other unknown electrodes. • To determine the pH of a solution. E=Eo- 2.303 RT/nF log [H2]1/2/[H+] = 0 -0.0591 log 1/[H+] = -0.0591pH. Cell Scheme: Pt,H2,H+(x)// SHE
The emf of the cell is determined. • E (cell) = E (c) – E(A) = 0 – (- 0.0592 pH) E (cell) = 0.0592 pH pH = E(cell)/ 0.0592
Limitations. • Constuction and working is difficult. • Pt is susceptible for poisoning. • Cannot be used in the presence of oxidising agents.
Metal –metal salt ion electrode. • These electrodes consist of a metal and a sparingly soluble salt of the same metal dipping in a solution of a soluble salt having the same anion. Eg: Calomel electrode. Ag/AgCl electrode.