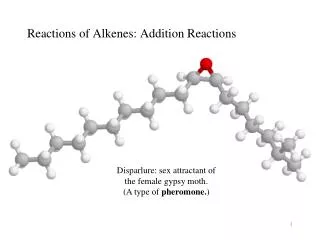
Reactions
Reactions. CH.3 Balancing Reactions Reaction Types. A Chemical Reaction. Reactants Products Types of Chemical reactions: There are many types of reactions but most can be classified into a few simple reactions types. How do you know a chemical reaction has occurred? .

Reactions
E N D
Presentation Transcript
Reactions CH.3 Balancing Reactions Reaction Types
A Chemical Reaction • Reactants Products • Types of Chemical reactions: • There are many types of reactions but • most can be classified into a few simple • reactions types. • How do you know a chemical reaction has occurred?
Chemical equations • Chemist’s shorthand to describe a reaction. • It shows: • All reactants and products • The state of all substances • Any conditions used in the reaction • CaCO3 (s) CaO (s) + CO2 (g) Reactant Products A balanced equation shows the relationship between the quantities of all reactants and products.
Balancing chemical equations • Each side of a chemical equation must have the same number of each type of atom. • CaCO3 (s) CaO (s) + CO2 (g) • Reactants Products • 1 Ca 1 Ca • 1 C 1 C • 3 O 3 O
Balancing chemical equations • Step 1 Count the number of atoms of each element on each side of the equation. • Step 2 Determine which atom numbers are not balanced. • Step 3 Balance one atom at a time by using coefficients in front of one or more substances. • Step 4 Repeat steps 1-3 until everything is balanced.
Chemical Changes Temperature Changes Light Gas formation Precipitation Color changes
Types of Chemical Reactions • Reactions involving electron transfer • synthesis or combination • decomposition • Electrochemical or single replacement • Combustion of hydrocarbons. • Reactions that involve rearrangement but not necessarily involve electron transfer • Metathesis or Double Substitution • Precipitation reactions and • Acid Base Reactions • Polymerization reaction
Properties ofaqueous solutions • There are two general classes of solutes. • Electrolytic • ionic compounds in polar solvents • dissociate in solution to make ions • conduct electricity • may be strong (100% dissociation) or weak (less than 100%) • Nonelectrolytic • do not conduct electricity • solute is dispersed but does not dissociate
Dissolving ionic compounds • When an ionic solid dissolves in water, the solvent removes ions from the crystal. • NaCl + H2O Na+(aq) + Cl- (aq)
Dissolving covalent compounds • Covalent compounds do not dissociate. • C6H12O6 C6H12O6 (aq)
H2O H2O Ionic equations • When ionic substances dissolve in water, they dissociate into ions. • AgNO3 Ag++ NO3- • KClK+ + Cl- • When a reaction occurs, only some of the ions are actually involved in the reaction. • Ag++ NO3- +K+ + Cl- AgCl(s) + K+ + NO3-
Ionic equations • To help make the reaction easier to see, we commonly list only the species actually involved in the reaction. • Molecular equation • KCl + AgNO3AgCl(s) + KNO3 • Full ionic equation • Ag++ NO3- +K+ + Cl- AgCl(s) + K+ + NO3- • Net ionic equation • Ag++Cl-AgCl(s) • NO3- and K+ are referred to as spectator ions.
Some simple solubility rules • All acids are soluble. • All Na+, K+ and NH4+ salts are soluble. • All nitrate and acetate salts are soluble. • All chlorides except AgCl and Hg2Cl2 are soluble. PbCl2 is slightly soluble. • All sulfates are soluble except PbSO4, Hg2SO4, SrSO4 and BaSO4. Ag2SO4 and CaSO4 are slightly soluble. • All sulfides are insoluble except those of the Group IA (1), IIA (2) and ammonium sulfide. • All hydroxides are insoluble except those of the group IA(1) and Ba(OH)2. Sr(OH)2 and Ca(OH)2 are slightly soluble.
Metathesis Reactions • Precipitation reactions • the formation of a solid upon mixing two solutions. • Gas formation • The formation of a gas when two mixtures are reacted • Acid Base titration • The neutralization of acids and bases to form a salt in water.
Precipitation • NaCl + AgNO3 = AgCl (ppt) + NaNO3 • Pb(NO3)2 + K2CrO4 = PbCrO4 (ppt) + KNO3
Gas Formation • Vinegar + Baking soda = Carbon dioxide • CH3COOH + NaHCO3 = NaCH3COO + H2O + CO2
Acid Base Neutralization • Vinegar + Ammonia = Salt + water • CH3COOH + NH4OH = NH4CH3COO + H2O
Common REDOX Reactions • Combustion • Corrosion • Photosynthesis • Kreb’s Cycle • Synthesis and Decomposition • Single Replacement
Oxidation numbers and the periodic table • Some observed trends in compounds. • Metals have positive oxidation numbers. • Transition metals typically have more than one oxidation number. • Nonmetals and semimetals have both positive and negative oxidation numbers. • No element exists in a compound with an oxidation number greater than +8. • The most negative oxidation numbers equals 8 - the group number
Oxidation Numbers • 1. An atom in its elemental state has an oxidation number of 0. • 2. An atom in a monatomic ion has an oxidation number identical to its charge. • 3. An atom in a polyatomic ion or in a molecular compound usually has the same oxidation number it would have it were a monatomic ion. • 4. The sum of the oxidation numbers is 0 for a neutral compound and equal to the net charge for a polyatomic ion.
Identifying oxidation-reduction reactions. • Oxidation-Reduction - REDOX • A chemical reaction where there is a net change in the oxidation number of one or more species. • Both an oxidation and a reduction must occur during the reaction. Mg (s) + Cl2 (g) MgCl2 (s) Here the oxidation number of Mg has changed from zero to +2. Cl has changed from zero to -1.
REDOX reactions • Oxidation • An increase in oxidation number. • Reduction • A decrease in oxidation number. • If the oxidation number of any element changes in the course of a reaction, the reaction is oxidation-reduction. • Example. • 2 Fe(NO3)3 (aq) + Zn(s) 2 Fe(NO3)2 (aq) + Zn(NO3)2 (aq)
Half Reactions • The reactions that show the loss or gain of electrons. • 2 Mg + O2 2 MgO • What are the oxidation states of each of the atoms in the above reaction? • Mg Mg+2 • O2 2 O2-
Oxidation • 2 Mg + O2 2 MgO • Mg Mg+2 • 12 p, 12 e 12 p, 10 e • Mg Mg+2 + 2 e • The loss of electrons
Single replacement reaction • Where one element displaces another in a chemical compound. • H2 + CuO Cu + H2O • In this example, hydrogen replaces copper. • This type of reaction always involves oxidation and reduction (REDOX). • Since one species is replacing another, there are no spectator ions.
Synthesis Reaction • The formation of a new compound from several substances • 2 H2 + O2 = 2 H2O • 2 Na + Cl2 = 2 NaCl • CaO (lime water) + CO2 = CaCO3 (s)
Decomposition Reactions • HgO = Hg + O2 • H2O2= H2O + O2 • with MnO2 catalyst
1774: Antoine Lavoisier solves the question of the burning process and chemical processes
Combustion • The burning of a substance in the presence of oxygen. • Gasoline burns when oxygen is taken into your engine, the products of the reaction are carbon dioxide and water • C8H18 + O2 = CO2 + H2O • Fe + O2 = Fe2O3 (rust)