Lecture 25: VSEPR
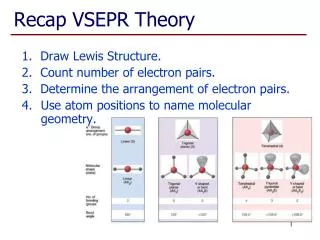
Lecture 25: VSEPR. Reading: Zumdahl 13.13 Outline Concept behind VSEPR Molecular geometries. VSEPR Background. • The Lewis Dot Structure approach provided some insight into molecular structure in terms of bonding, but what about geometry?.

Lecture 25: VSEPR
E N D
Presentation Transcript
Lecture 25: VSEPR • Reading: Zumdahl 13.13 • Outline • Concept behind VSEPR • Molecular geometries
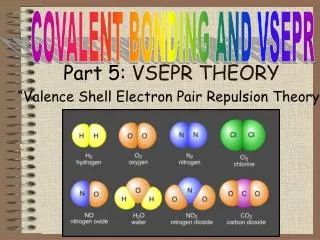
VSEPR Background • The Lewis Dot Structure approach provided some insight into molecular structure in terms of bonding, but what about geometry? • Recall from last lecture that we had two types of electron pairs: bonding and lone. • Valence Shell Electron Pair Repulsion (VSEPR). 3D structure is determined by minimizing repulsion of electron pairs.
VSEPR Background (cont.) • Must consider both bonding and lone pairs in minimizing repulsion. • Example: CH4 Lewis Structure VSEPR Structure
VSEPR Background (cont.) • Example: NH3 (both bonding and lone pairs). Lewis Structure VSEPR Structure
VSEPR Applications • The previous examples illustrate the stratgey for applying VSEPR to predict molecular structure: 1. Construct the Lewis Dot Structure 2. Arranging bonding/lone electron pairs in space such that repulsions are minimized.
VSEPR Applications (cont.) • Shorthand notation: 1. Refer to central atom as “A” 2. Attached atoms are referred to as “X” 3. Lone pairs are referred to as “E” • Examples: H2O: AX2E2 CH4: AX4 BF3: AX3 PCl5: AX5 NH3: AX3E ClF3: AX3E2
VSEPR Applications • Linear Structures (AX2): angle between bonds is 180° • Example: BeF2 180°
VSEPR Applications • Trigonal Planar Structures (AX3): angle between bonds is 120° • Example: BF3 120°
VSEPR Background (cont.) • Pyramidal (AX3E): Bond angles are <120°, and structure is nonplanar due to repulsion lone-pair. • Example: NH3 107°
VSEPR Applications • Tetrahedral (AX4): angle between bonds is ~109.5° • Example: CH4 109.5°
VSEPR Applications • Tetrahedral: angle may vary from 109.5° exactly due to size differences between bonding and lone pair electron densities bonding pair lone pair
VSEPR Applications • Classic example of tetrahedral angle shift from 109.5° is water (AX2E2): “bent”
VSEPR Applications • Comparison of CH4 (AX4), NH3 (AX3E), and H2O (AX2E2):
What is the approximate bond angle in SO2? A. 90° C. 120° D. 109.5° B. 180°
VSEPR Applications • Trigonal Bipyramidal (AX5), 120° in plane, and two orbitals at 90° to plane. • Example, PCl5: 90° 120°
VSEPR Applications • Octahedral (AX6): all angles are 90°. • Example, SF6: 90°
Advanced VSEPR Applications • See-Saw versus Square Planar AX4E: See Saw AX4E2: Square Planar <120° 180° 90°
Advanced VSEPR Applications • Square Planar versus “See Saw” See Saw No dipole moment Square Planar
Advanced VSEPR Applications • Driving force for last structure was to maximize the angular separation of the lone pairs. Same effect occurs in I3- (AX2E3):
What is the orientation of the ClF bonds in ClF3 (28 e-)? AX3E2 A B C
+1 +1 0 0 -1 -1 Advanced VSEPR Applications • VSEPR and resonance structures. Must look at VSEPR structures for all resonance species to predict molecular properties. Example: O3 (AX2E): bent Dipole moment? Yes!
VSEPR Applications • Provide the Lewis dot and VSEPR structures for CF2Cl2. Does it have a dipole moment? 32 e- Tetrahedral
What is the expected shape of ICl2+? AX2E2 A. linear C. tetrahedral D. square planar B. bent