Lecture 20. Adsorption Phenomena
E N D
Presentation Transcript
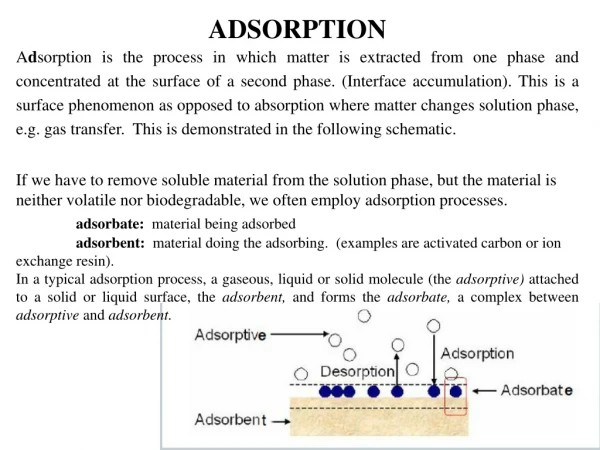
Adsorption is the accumulation of atoms or molecules on the surface of a material. This process creates a film of the adsorbate (the molecules or atoms being accumulated) on the adsorbent's surface. It is different from absorption, in which a substance diffuses into a liquid or solid to form a solution. The term sorption encompasses both processes, while desorption is the reverse process of "adsorption".
Isotherms Adsorption is usually described through isotherms, that is, the amount of adsorbate on the adsorbent as a function of its pressure (if gas) or concentration (if liquid) at constant temperature. some of these isotherms are: • Langmuir • BET
Langmuir Langmuir derived a relationship for q; weight adsorbed per unit wt of adsorbent and C; concentration in fluid based on some quite reasonable assumptions. These are: a uniform surface, a single layer of adsorbed material, and constant temperature. The rate of attachment to the surface should be proportional to a driving force times an area. The driving force is the concentration in the fluid, and the area is the amount of bare surface. If the fraction of covered surface is F , the rate per unit of surface is:
Langmuir rate going on = k1 C ( 1 - ϕ ) The evaporation from the surface is proportional to the amount of surface covered: rate leaving = k2ϕ where k1 and k2 are rate coefficients C = concentration in the fluid ϕ = fraction of the surface covered
Langmuir At equilibrium, the two rates are equal, and we find that: A plot of versus should indicate a straight line of slope and an intercept of . The graph shows data points and lines fitted to both Freundlich and Langmuir equations.
BET Theory BET theory is a rule for the physical adsorption of gas molecules on a solid surface and serves as the basis for an important analysis technique for the measurement of the specific surface area of a material. The concept of the theory is an extension of the Langmuir theory, which is a theory for monolayer molecular adsorption, to multilayer adsorption with the following hypotheses:
BET Theory • gas molecules physically adsorb on a solid in layers infinitely; (b) there is no interaction between each adsorption layer; and (c) the Langmuir theory can be applied to each layer.
BET Theory The resulting BET equation is expressed by: P and P0 are the equilibrium and the saturation pressure of adsorbates at the temperature of adsorption, v is the adsorbed gas quantity (for example, in volume units), and
BET Theory vm is the monolayer adsorbed gas quantity. c is the BET constant. The BET method is widely used in surface science for the calculation of surface areas of solids by physical adsorption of gas molecules.