The Ideal Gas Equation
490 likes | 2.52k Views
The Ideal Gas Equation. pV = nRT. The Ideal Gas Equation. Changing the temperature and pressure of a gas will change its volume. If the volumes of gases are not at stp we need to use the ideal gas equation What is an “ideal gas”?. An Ideal Gas. Identical particles in rapid random motion

The Ideal Gas Equation
E N D
Presentation Transcript
The Ideal Gas Equation pV = nRT
The Ideal Gas Equation • Changing the temperature and pressure of a gas will change its volume. • If the volumes of gases are not at stp we need to use the ideal gas equation • What is an “ideal gas”?
An Ideal Gas • Identical particles in rapid random motion • Particles = hard spheres of negligible size • Particles don’t react when they collide • Collisions between particles are elastic • Kinetic energy before = kinetic energy after • No intermolecular forces
The Effect of Pressure • At constant temperature Increasing pressure Gas compressed into smaller volume Volume decreases as pressure increases V is indirectly proportional to p V 1/p
The Effect of Temperature • At constant pressure Gas increases in volume Increasing temperature Volume increases as temperature increases V is directly proportional to T V T
The Effect of Number of moles • At constant temperature & pressure “n” moles 2n moles Volume increases as number of moles increases V is directly proportional to n V n
V T V 1/p V n • If we combine these three equations V nT p R = gas constant V = RnT p pV = nRT
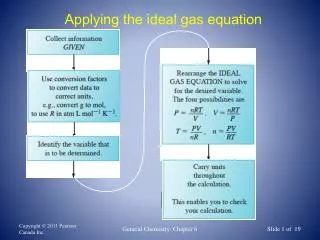
The Ideal Gas Equation • p = pressure (Pa) • V = volume (m3) • n = number of moles • R = the gas constant = 8.31JK-1mol-1 • T = temperature (K) pV = nRT
Converting Units • Temperature • 0oC = 273K • a OC → a + 273K • Pressure • 1kPa = 1000Pa • a kPa = a x 1000Pa
Converting Units • Volume • 1m = 10 dm = 100 cm • 1m3 = 103 dm3 = 1003 cm3 • 1m3 = 1000 dm3 = 1 000 000 cm3 • 1dm3 = 1 1000 m3 = 1 x 10-3 m3 m3 • 1cm3 =1 1000 000 = 1 x 10-6 m3
What volume is occupied by 0.25 mol of a gas at 200kPa and 27oC? 1. Convert units 200kPa = 200 x 1000 Pa = 2 x 105 Pa 27oC = 27 + 273 = 300K 2. Rearrange pV = nRT Equation V = nRT p V = 0.25 x 8.31 x 300 2 x 105 V = 3.12 x 10-3 m3
At 571K a 0.6g sample of He occupies a volume of 7.0 dm3, Calculate pressure. 1. Convert mass into moles n=m/Mr n = 0.6 4 = 0.15 2. Convert units = 7.0 x 10-3 m3 7.0 dm3 = 7 1000 3. Rearrange pV=nRT Equation p = 0.15 x 8.31 x 571 7 x 10-3 p = nRT V p = 1.02 x 105 Pa
0.71g of a gas when contained in a vessel of 0.821dm3 exerted a pressure of 50.65kPa at 227oC. Use these data to calculate Mr of the gas 1. Convert units 0.821dm3 = = 8.21 x 10-4 m3 0.821/1000 m3 227oC = 227 + 273 = 500K 5.065 x 104 Pa 50.65kPa = 50.65 x 1000 Pa = 2. Rearrange pV = nRT Equation n = pV RT n = 5.065 x 104 x 8.21 x 10-4 8.31 x 500 n = 0.01 mol
0.71g of a gas when contained in a vessel of 0.821dm3 exerted a pressure of 50.65kPa at 227oC. Use these data to calculate Mr of the gas 3. Calculate Mr using n = m/Mr = 0.71 0.01 Mr = m n = 70.94 4. Check final answer Gases are small molecules – they rarely have Mr values over 100