Organic Biomolecules
610 likes | 891 Views
Murray Low & Udhir Chathuri. Organic Biomolecules. Don’t be scared. Testosterone. Adenosine Triphosphate. Overview. Organic Biomolecules. Principles of Organic Chemistry. Quiz A. Proteins. Carbohydrates. Nucleic Acids. Primary Structures. Quiz C. Phosphate. Quiz B.

Organic Biomolecules
E N D
Presentation Transcript
Murray Low & UdhirChathuri Organic Biomolecules Don’t be scared Testosterone Adenosine Triphosphate
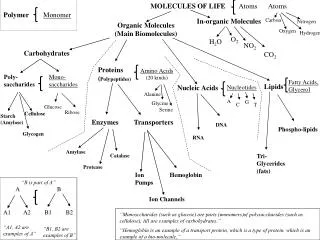
Overview Organic Biomolecules Principles of Organic Chemistry Quiz A Proteins Carbohydrates Nucleic Acids Primary Structures Quiz C Phosphate Quiz B Monosaccharides Quiz D Secondary Structures Tertiary Structures Sugars Dissacharides Polysaccharides DNA Helix Bases Base Pairing
Quiz A- Basic Organic Chemistry Principles • How many bonds do the following atoms form in a covalent compound? a) O b) C c) S d) N e) H f) P 2. Write down the formula of the functional group in each of the following compounds. a) amine b) amide c) alcohol d) carboxylic acid e) ketone f) aldehyde g) ester 3. Give the name for each of the following molecules. a) CHCH e) b) CH3CH2OH f) c) C2H6 g) d)
Quiz A- Basic Organic Chemistry Principles • Which of the following molecules might be unsaturated and which are definitely • unsaturated? a) C3H8 b) C4H8 c) C3H4 d) CH3CHCHCH3 5 a) Name the following molecules. i) CH3CH2CH2CH2CH2CH2CH2CH2OH ii) CH3CH2CH2CH2CH2OH iii) CH3CH2OH b) Identify the 2 types of intermolecular bonding present in each of the above molecules. c) Which of the three molecules do you expect to have the highest boiling point temperature? Explain your answer.
Quiz A- Basic Organic Chemistry Principles 6. Which type of reactions do you expect the following molecules to undergo? a) CH2CH2 b) CHCH c) CH3CH2CH – CH3 Cl 7. Classify the type of reaction occurring in each of the following cases. a) b) 8. Write down all the possible isomers for the compounds which have the following formulae. a) C3H8O a) C4H8
Quiz A- Basic Organic Chemistry Principles 9. Identify the functional groups present in the following molecule.
Proteins Proteins are the most structurally complex organic macromolecules. They have 1o, 2o & 3o structure. The building blocks of all proteins are amino – acids. = side chain Base Acid 20 amino acids in all. The 12 the body can synthesize are termed non-essential amino acids. The 8 that we obtain from our diet are termed essential amino acids.
Proteins Non – essential amino acids L–Alanine L–Asparagine L–Aspartic Acid L–Cysteine L–Glutamic Acid L–Glutamine Glycine L–Proline L–Serine L–Tyrosine L–Arginine L–Histidine
Proteins Essential amino acids(must be in the diet) L–Isoleucine L–Leucine L–Lysine L–Methionine L–Phenylalanine L–Threonine L–Tryptophan L–Valine
Proteins L–Alanine Glycine L–Serine
Proteins – Zwitter Ions All amino acids have the ability to form their corresponding ionic forms as follows. H H O O H H H H N N C C C C H O O H H R R + - O H H The zwitter ion form of the amino acid will predominate in aqueous solution. N C C O H R
Proteins H Peptide Bond O H H H H H N C C O O O O H H H H R H H H N N N C C C C C C O O O H H H R R R Dehydration
Primary structure of Proteins Reactive ends Peptide Bonds Ala •Ser •Gly
Primary structure of Proteins Try• Lys • Ala •Ser •Gly •Val •Pro ………… ………… Polypeptide (Protein) : 50 – 2000 amino acids
Primary/Tertiary structure of Proteins S S S S L–Cysteine R
Secondary structures - -helix Clockwise H O O H 3.6 Amino acids per turn of the helix
Secondary Structures - Zwitter ion formation Some amino acids have extra reactive groups. e.g. Carboxylic acid (COOH) – glutamic acid – aspartic acid Amine (NH2) – lysine Lys + - + + Ionic bond - + - + - - H Asp
Top view Secondary structures - -sheets Hydrogen bonding H H H H N N C C C C O O Side view R R
Tertiary structures • - sheets - helix Has a 3D structure. Contains a combination of secondary structures & primary structures. Structure determined by side – chains. Hydrophobic side chains Hydrophobic Pocket
Quiz B- Proteins 1. Which of the amino-acids have a hydrophobic side-chain? 2. Which of the amino-acids have an acidic side-chain? 3. Which of the amino-acids have a basic side-chain? 4. Which amino acids have a side chain which can become involved in protein hydrogen bonding? 5. Draw the zwitter – ion structure of the amino-acids glycine and serine. • Draw the chemical structure of the tripeptide formed from the following sequence • of amino-acids. •Lys •Tyr Cys Highlight the acidic end of the molecule and the basic end of the molecule.
Carbohydrates Monosaccharides Can be split into two groups : Aldoses & Ketoses. ‘ oses ’ “CH2O” ‘ Ald’ Aldehyde ‘ Ket’ Ketone ‘ C6H12O6’ Ketoses e.g. Fructose Aldoses e.g. Glucose Both Glucose & Fructose are Hex - ‘oses’. Sugars can be 3 to 7 carbons long.
Carbohydrates In aqueous solution sugars form rings e.g. glucose. 1 2 6 3 5 4 4 1 5 2 3 6
Carbohydrates Sugars polymerize via glycosidic 1–4 linkages. H H H H H H H H O O O O H H H H C C C C 5 5 5 5 C C C O O O C O H H H H H H H H H H H H H H H H 1 1 1 4 1 C C C O O O H H H C C C C O H C 4 4 4 O O O C C C C C C O O C C O O 3 3 3 2 2 2 3 2 H H H H H H H H H O O O H O Glycosidic linkage H H H H H 2C6H12O6 C12H22O11 + H2O O Dehydration H
Carbohydrates glucose + glucose maltose glucose + fructose sucrose polysaccharide monosaccharide disaccharide Maltose e.g. Glucose Starch (100s - 1000s of units)
Carbohydrates The resulting polysaccharide of 1–4 linakges of glucose is starch (storage polysaccharide in plants). Amylose Unbranched starch 1–4 linkages Starch Amylopectin Branched Starch 6 • 1–4 linkages & • 1–6 linkages Branched 1–6 linkages occur every 30 linkages
Carbohydrates - 1-6 linkage H 6 O H C H 5 C O H H H H 1 C O H C 4 O C C O 3 2 H H H H O H H 6 O O O H H H C C C H H 6 H H 6 5 5 5 C O C C O O H H H H H H H H H H H H 1 1 1 C O H C C C O O H H C C 4 4 4 O C C O O O C C C C O O 3 2 H 3 3 2 2 H H H H H H O H H O O H H H
Carbohydrates Sugars can polymerize via glycosidic 1–4 linkages as well. H H H H O O H H C C H H H H 5 5 H H C C O O O O O O H H H H C C H H 4 4 H H 1 1 H C C O O H H C C 5 5 C C O O O O H H H O C C C C H H 3 2 3 2 H H H H 1 1 C C O O H H C C H H H O O 4 4 O O C C C C H H H 3 3 2 2 • 1–4 • Glycosidic • linkage H H H H O O H H H The resulting polysaccharide is cellulose (structural polysaccharide in plants). O H
Quiz C- Macromolecules • The most abundant (by mass) class of organic macromolecules in the biological • world is : A Carbohydrates B Proteins C Nucleic Acids (DNA & RNA) D Lipids • D – ribose is a pentose. It can also be classified as an aldose. Write down a possible • structure for D – ribose. • The monosaccharide threosehas 4 Os ! That is it has 4 oxygen atoms. • What is the molar mass of threose?
Quiz C- Macromolecules A comprehension exercise 4. When one draws the structure of D – glucose there are 2 ways to do it : either a Fischer projection or a Haworth projection. 1 The Fischer projection shows glucose as a linear molecule and the numbering of the carbon atoms is shown on the structure. When it cyclizes it is the oxygen atom on carbon 5 which ends up in the ring. 2 3 4 5 6 6 This ring structure is called the Haworth projection and at equilibrium between the two forms the linear form is only 0.02% of the total glucose present. 5 4 1 2 3
Quiz C- Macromolecules The Haworth projection has the atoms on the RHS of the Fischer projection ‘down’ i.e. below the ring in the Haworth projection, and those on the LHS of the Fischer projection are up (above the ring). The –OH group on carbon 1 (the so called anomeric carbon) can either be up or down, (), or up, (). The form is about twice as common as the form. • Draw the Haworth projection of the form of D – idose, given the Fischer projection • below. b) Draw the Fischer projection of D – galactose given the Haworth projection below. - D -galactose
Nucleic Acids Consists of two groups : Deoxyribonucleic acid (DNA) Ribonucleic acid (RNA) There are 3 basic units to the construction of a nucleic acid. Phosphate Sugar Base + +
Nucleic Acids – ThePhosphateBackbone O O O P O R R H O O O O R P O O P H H H O O Phosphomonoester Phosphoric Acid Phosphodiester Phosphotriester H H O O O P Sugar Sugar H O H
Nucleic Acids – The Sugar Residue H H O H O H H H C O 5´ O 5´ H O H C O C C H H 1´ 1´ H C C H H 4´ 4´ H C C 2´ H 2 ´ C C 3´ 3´ O O H O H H H H 2´–deoxy–D–ribose D–ribose DNA RNA
Nucleic Acids – The Bases H H C C There are two chemical types of bases based on the following structures : N H C N C N C H C C C C N H H H N N H Pyrimidine Purine
Nucleic Acids – The Bases Purine based H H O N C Adenine Guanine H C N H N C N C N C C H C C H C C N N N N H N H H H
Nucleic Acids – The Bases Pyrimidine based H H N O O H H C C C H C H H H C N C N C N H C C C C C C O O O H N H N H N Cytosine Uracil Thymine H H H
Nucleic Acids – The Bases DNA RNA H H N O O H H C C C H C H H H C N C N C N H H H O Adenine Guanine N C C C C C C C O H O O H N C H N N H N H N C N C N H H H C C H C C H C C N N N N H N H H H Cytosine Uracil Thymine
Nucleic Acids – Nucleoside formation + Sugar Base Nucleoside H H Example 1 : N H H C N O C N C H Adenine C C N H N H H N H H H H H O O O H H C C C O O N C H H N H H C C C C H H Adenosine C H C C C C H H C C O O O O N H N H H H H D–ribose H
Nucleic Acids – Nucleoside formation H H Example 2 : N H H H H C N O H C N C Cytosine H C C C N O H N H C C H H H H O O O O H N H H C C O O H H H H C C C C H H H 2´–deoxycytidine C C C C H H H H O O H H 2´ – deoxy–D–ribose
Nucleic Acids – Nucleotide formation + Phosphate Nucleoside Nucleotide H H Example 1 : N H H H H C O N N N C N C C C H N N O O C C C C N N P N H H H N 5´ C C H H H H C C C C N N 1´ H H H H H O O N N O O C C O O H H 4´ O H C O O O 2´ Adenosine P H H H H H H C C C C 3´ H H H H H C C O H C C C C H H H C C H O O O O O O H H H H Adenosine 5´–monophosphate H H
Nucleic Acids – Nucleotide formation H H H H Example 2 : N N C C O H H 5´ C C N N 1´ 2´–deoxycytidine O O C C C C P H O O H 4´ H H H N N H O H H 2´ H C O 3´ H O H H C O H H O O C C H H H H C C O O P H H H C C H H H O C C O H H H O 2´–deoxycytidine 3´–monophosphate H
O O H H C C C H N H C N C N H C H O O O O H H C C C C H O N N H N N N H H H H H H H H O O O O C C C C C H H H H H H H H H H C C C C C C C C C N H H H H deoxyadenosinemonophosphate C C C C C C C C H H H H C C thymidinemonophosphate O H H H H O O O O H N H H H H O O O O O O O O H H H H P P P P O O O O H H H H H H N C N C N C H C C N H N deoxyguanosinemonophosphate deoxycytidinemonophosphate
H O O P O O Nucleic Acids – Chain formation Base H H H O C Chain formation results from the phosphate side groups bridging between two nucleotides. H H C C H C C H O O O H O H H O O O O H H P P H H H H H H O O O O O O O C C C O O P Base Base H H O H H H H H H C C C C C C O Base Base Base H H H Base H H H H H O O C C C C C C C C H H H H H O H H H H C C C C C H H H H H O O O H H C C H H H O O O O O O H H H H C C C C H H P P P H H O O C C H O O O H H O H H H H O O O O P P P O O O O O O Base Base Base H H H H H H H H H O O O C C C H H H H H H C C C C C C H H H C C C C C C H H H H H H O O O H H H
Nucleic Acids – Chain formation 5´ O O O C C N N H H C C N N H H O C C C H H O O H H C C C C C C H H H N N N N N N H H H H O O C C C C H H H H O H H H H C C C C H H H H O O C C C C H H P H H O O O H H H O O O O O O O H H H P P P H H O O O O C H H H H H C C H H H H H N N C C H C C H O 3´ N N C C N N H C C H H C C C C N N H H N N
Nucleic Acids – Chain formation H O H H H H H C O O H H H O C 3´ C H C H H C C C C C C N H H H C H H O H C C C C H H H H C C N H H C C O C C N N H H H N O O H H C H H H C C H C O H H H O O C C N H O O O H H P P O O O O C C H H O O H H O H N H N P H C O O C H H C H O H N H C C N C H C C H O C H O H H 5´ H O O P O O H
Nucleic Acids – Base Pairing Once an individual strand of DNA is encoded, a complementary strand must be created. This is accomplished via Base Pairing. Bases can only be complemented as follows : * Adenine Thymine Guanine Cytosine Information is thus transferred from one strand to another via Base Pairing. Purine Pyrimidine * Uracil replaces Thymine in RNA
Nucleic Acids – Base Pairing H O H H C O C C N H C C N H C C H N H C C H N N N N C H O Adenine Thymine H Hydrogen Bonding H N H C C C H N H H N C N O C N C N C H C C N Guanine Cytosine H N
H O H H C O O C C N H C 5´ C N H H H O H C C C O H H 3´ N H H H H C C C C C C H N H N N N C H C C C C H H H O H O C H O H H O H H O P O H O H N P H O O O H C O C P H H O C H O C O H H N H H H C C C C H H N H C N 3´ H C C C C H H O C H O C N 5´ H O H C N H H C H H O C C O N H P N O O H
Nucleic Acids – Base Pairing 3´ 5´ G C A T T A C G C G A T G C T A 5´ 3´
The DNA Double Helix Richard Wheeler http://en.wikipedia.org/wiki/DNA