Immunization Schedule for Filipino children
400 likes | 1.24k Views
Immunization Schedule for Filipino children. Objectives. To present the 2013 Immunization schedule for Filipino children with focus on Minimum age for vaccination Route of vaccination Recommended schedule for vaccination. Philippine EPI BCG Hepatitis B Measles

Immunization Schedule for Filipino children
E N D
Presentation Transcript
Objectives • To present the 2013 Immunization schedule for Filipino children with focus on • Minimum age for vaccination • Route of vaccination • Recommended schedule for vaccination
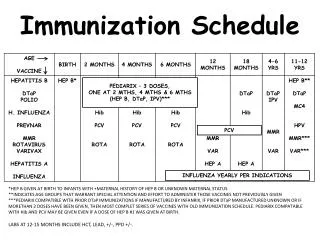
Philippine EPI BCG Hepatitis B Measles OPV DTwP Hib MMR RV PCV • Other recommended vaccines • are not part of the Philippine EPI but because of merit are advocated by the PPS, PIDSP, PFV DTaP IPV MMRV Td/Tdap Hepatitis A Varicella Influenza HPV • Vaccines for Special Groups Typhoid Meningococcal vaccine PCV/PPV Rabies
BCG • Given intradermally (ID) • BCG should be given at the earliest possible age after birth preferably within the first 2 months of life.
In the EPI schedule, hepatitis B is given as monovalent hepatitis B vaccine at birth then subsequent doses are given at 6, 10, 14 weeks of age as combination vaccines containing DTwP/Hep B/Hib.
DOH ADMINISTRATIVE ORDER 2012-0157: Administration of Rotavirus vaccine
PCV (Pneumococcal Conjugate Vaccine)PPV (Pneumococcal Polysaccharide Vaccine) • The minimum age for pneumococcal conjugate vaccine (PCV) is 6 weeks. • Primary vaccination of PCV consists of 3 doses with an interval of at least 4 weeks between doses, plus a booster (at 12-15 mos of age) given 6 months after the 3rd dose. • Healthy children 2 to 5 years old who have no previous PCV vaccination may be given one dose of PCV 13 or 2 doses of PCV 10 at least 8 weeks apart. • Routine use of PCV is not recommended for healthy children aged ≥ 5 years. • For high risk children > 2 years of age, PPV is recommended after completing PCV series (refer to Vaccines for Special Groups). • For healthy children, no additional doses of PPV are needed if PCV series is completed.
Influenza vaccine dosing algorithm for children aged 6 months through 8 yearsAdvisory Committee on Immunization Practices United States, 2012-2013 influenza season
Vaccines for Special Groups • Not part of EPI or recommended vaccines but data support its use in certain conditions or populations
Meningococcal Vaccine • Tetravalent meningococcal (ACYW-135) conjugate vaccine (MCV4) given IM • Tetravalent meningococcal polysaccharide vaccine (MPSV4) given SC • Bivalent meningococcal polysaccharide A and C vaccine given IM/SC • Children aged 9 mos & above at high risk for invasive disease (persistent complement component deficiencies, anatomic/functional asplenia, HIV, travelers to or residents of areas where meningococcal disease is hyperendemic or epidemic, or belonging to a defined risk group during a community or institutional meningococcal outbreak,) should receive 2 doses of MCV4 given 2 months apart. • If MPSV4 or bivalent meningococcal polysaccharide A and C vaccine are used for high-risk individuals as the first dose, a second dose using MCV4 should be given 2 months later. • If the person remains at increased risk, an initial booster dose of MCV4 should be given 3 years after completing the primary series, with continued boosters at 5-year intervals after the initial booster dose. • Additional booster doses of MPSV4 or bivalent meningococcal polysaccharide A and C vaccine are not recommended.
PNEUMOCOCCAL VACCINES (PCV/PPV) • Given intramuscularly (IM) • For children > 2 years w/ the ff underlying medical conditions such as but not limited to anatomic/functional asplenia, chronic heart or lung disease, chronic renal failure or nephrotic syndrome, DM , cochlear implant, cerebral spinal leak, congenital immunodeficiency, HIV infection, & other immunocompromising condition resulting from disease or treatment of disease. • Children aged 24 to 71 months who were previously given 4 doses of PCV, give one dose of PPV at least 8 weeks after last dose of PCV • Children aged 24 to 71 months who were previously given 3 doses of PCV, give one dose of PCV 13 or if previously given less than 3 doses of PCV, give 2 doses of PCV 13 at least 8 weeks apart. This should be followed by one dose of PPV at least 8 weeks after the last dose of PCV. • Children aged 6 to 18 years with no previous doses or incomplete doses of PCV or previously given PPV, give a single dose of PCV 13, followed by one dose of PPV at least 8 weeks after last dose of PCV 13.
PNEUMOCOCCAL VACCINES (PCV/PPV) • A second dose of PPV is recommended 5 years after the first dose of PPV ONLY for those with • anatomic/functional asplenia, congenital immunodeficiency, HIV infection, chronic renal failure or nephrotic syndrome, malignancy, solid organ transplantation, and other diseases associated with immunosuppressive drugs or radiation therapy. No more than 2 PPV doses should be given.
Rabies Vaccine • The Anti-rabies Act of 2007 recommends routine rabies pre-exposure prophylaxis (PrEP) for children ages 5-14 years in areas where there is high incidence of rabies and animal bites (defined as more than twice the national average). • There are 2 recommended regimens for Pre-exposure Prophylaxis: • Intramuscular regimen: PVRV 0.5 ml or PCECV 1 ml given on days 0,7, 21 or 28. • Intradermal regimen: PVRV or PCECV 0.1 ml given on days 0, 7, 21 or 28 . • A repeat dose should be given if the vaccine is inadvertently given subcutaneously. • Rabies vaccine should never be given in the gluteal area since absorption is unpredictable. • Periodic booster doses in the absence of exposure are not recommended for the general population. • In the event of subsequent exposures, those who have completed 3 doses of pre-exposure prophylaxis, regardless of interval between re-exposure and last dose of the vaccine, will require only booster doses on day 0 and 3. Booster doses may be given IM (0.5 ml PVRV or 1 ml PCECV) or ID (0.1 ml PVRV or PCECV). There is no need to give rabies immune globulin