Physical Science Week 6
Physical Science Week 6. Unit 1 – Nature of Matter Chemical Properties & Changes. Standard S8P1a. Distinguish between atoms and molecules. Standard S8P1.g. Identify and demonstrate the law of conservation of matter. Standard S8P1.d.

Physical Science Week 6
E N D
Presentation Transcript
Physical Science Week 6 Unit 1 – Nature of Matter Chemical Properties & Changes
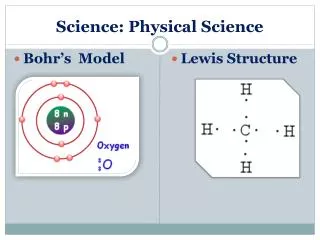
Standard S8P1a • Distinguish between atoms and molecules.
Standard S8P1.g • Identify and demonstrate the law of conservation of matter.
Standard S8P1.d • Distinguish between physical and chemical properties of matter.
Monday Warm Up (Wk 6) • If you have 100 grams of a substance and the density is 4 g/cm3; what is the volume? • Unit Test Friday – Study everything!!!
Cornell Notes Chemical Properties and Changes • Chemical Properties describe matter based on its ability to change into new matter that has different properties.
Flammability • The ability to burn
Reactivity • Ability of two or more substances to combine and form one or more new substances.
Comparing Physical & Chemical Properties • Physical properties can be observed without changing the substance. • Chemical properties, not so easy. • If you burn it to test for flammability, it is burnt.
Characteristic Properties • Most useful in identifying a substance • Always the same no matter the size of the sample • Density, solubility, flammability, reactivity
Chemical Change • A change that occurs when one or more substances change into entirely new substances with different properties.
Signs of chemical change • Gas produced (bubbles) • Odor • Color change • Production of heat • Sound or light produced
Reversing Chemical Change Cake batter to cake is a chemical change. How would you undo the change? Burn a log – how do you undo the change?
Law of Conservation of Mass • Mass cannot be created or destroyed in ordinary chemical and physical changes. • What goes into a reaction must come out – atom by atom.
Electrolysis of water • Water is separated into hydrogen and oxygen by electricity. • H2O -> H2 + O2 ___ H2O -> ___H2 + ___ O2
Tuesday Warm Up (Wk 6) • What evidence should you look for to decide if a chemical change has occurred? • Unit test Friday: Study everything
Physical and chemical Change Lab • Question: How do you tell if a change is physical or chemical? • Hypothesis: • Procedure: • Copy data table
Analyze and Conclude: 1. How can you determine if a chemical or physical change is happening? What is the most important question to ask? 2. Is one piece of evidence sufficient information to determine if it is a chemical or physical change? Give an example. 3. The Stature of Liberty was originally a copper color. After being exposed to air, she turned a greenish color. Explain what happened.
Physical Chemical Physical and Chemical Changes
Wednesday Warm Up (Wk 6) Compare and contrast chemical and physical changes with a Venn Diagram. Unit test Friday: Study everything
CRCT Practice • P. 60-61 #1-9 • P. 84-85 #1-9 • P. 110 – 111 #1-8 • P. 330-331 #1-11 • P. 356 – 357 #1-9
Thursday Warm Up (Wk 6) • How many atoms are in one molecule of C6H12O6? • What are the atoms and how many of each? • Unit test Friday: Study everything
Friday Warm Up • Study silently for unit test 5 minutes.