Gibbs Free Energy
460 likes | 1.26k Views
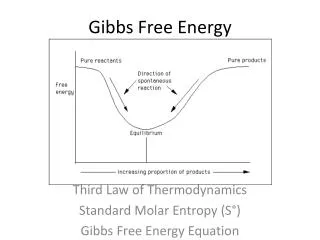
Gibbs Free Energy. Gibbs Free Energy (G) is a measure of enthalpy (heat) taking entropy (randomness) into account ΔG R ° is a measure of the driving force of a reaction G R < 0; forward reaction has excess energy, thus favors forward reaction

Gibbs Free Energy
E N D
Presentation Transcript
Gibbs Free Energy • Gibbs Free Energy (G) is a measure of enthalpy (heat) taking entropy (randomness) into account • ΔGR° is a measure of the driving force of a reaction • GR < 0; forward reaction has excess energy, thus favors forward reaction • GR > 0; forward reaction has deficiency of E, thus favors reverse reaction
Gibbs Free Energy • Two ways to calculate • GR = ΔHR - T ΔSR • GR = niGfi(products) – niGfi(reactants) • GR and Keq are related: • GR = -RT lnKeq • GR = -5.708 log Keq at 25°C
Activity • To apply equilibrium principles to ions and molecules, we need to replace concentrations with activities to account for ionic interactions • ai= iCi • a < C in most situations
Activity • Activity coefficients () are a function of ionic strength • I = ½ mi zi2 • Use Debye-Hückel or extended Debye-Hückelequation (unless saline solution) • log = -Az2I½ • log = - Az2I½ (1 - aBI½)
Aqueous Complexes • Complex: chemical association of 2 or more dissolved species to form a 3rddissolved species • Some examples: • Al3+ + OH-Al(OH)2+ • Al(OH)2+ + OH-Al(OH)2+ • Ca2+ + SO42- CaSO4(aq) • Ca2+ + HCO3- CaHCO3+(aq) • Note: ΣCa in solution = Ca2+ (ion) + CaSO4° + CaHCO3+ + any other Ca-containing complexes • Aqueous complex distinguished from solid of same composition by subscript (aq) or superscript °
Aqueous Complexes • 2 main types • Ion pairs • Coordination compounds
Ion Pairs • Cations and anions form associations in solution because of electrostatic attraction • Associations are weak bonds; form and decompose rapidly in response to changes in solution chemistry • Generally the greater the concentration, the greater the amount of ion pairs
Coordination Compounds • Ions surrounded by sphere of hydration; one or more water molecules displaced by ligands • Ligand = ion (usually anion) or molecule that binds to a central atom (usually a metal) • If a ligand can bind at more than one site, it is called a chelate, and the bond is stronger • Some coordination-type complexes are very stable • e.g., used in cleaning metal waste (EDTA, NTA)
Importance of complexes • Increase mineral solubility by decreasing effective concentrations • Equilibrium calculations are usually made with uncomplexed species • Analytical instruments usually measure total amount (uncomplexed+ complexed) of a species • ΣmCa = mCa2+ + mCaHCO3+ + mCaSO4° + … • ΣmCa = measured amount • mCa2+ used in thermodynamic calculations • We can’t directly measure mCaHCO3+, mCaSO4°: must calculate
Importance of complexes • Many elements exist dominantly as a complex or complexes) • Usually those with low solubilities such as metals • As, Fe, Al, Pb, Hg, Cu, U to name a few • e.g., As5+ usually exists as an oxyanion (H2AsO4-) and As3+ usually exists as an uncharged species (H3AsO3°)
Importance of complexes • Adsorption can be increased or inhibited • Adsorption is usually a weak attraction of charged species to aquifer solids • Charged complexes more likely than uncharged complexes to adsorb and be removed from solution • Bioavailability and toxicity • Some complexes of essential nutrients may pass straight through living organisms • Some complexes of toxic species may pass straight through living organisms • CH3Hg+ most toxic form; elemental Hg much less toxic
Complexes: General Observations • Low solubility elements exist predominantly as complexes (e.g., metals) • Complexation tends to increase with increasing I • More potential ions to complex with • Species are closer together • Mineral solubility also increases with increasing I • Combined effects of complexation and activity; ions in solution less reactive • Increasing charge density (charge per surface area) results in stronger complexes • Charge density increases with increasing valence or decreasing atomic radius • Function of valence and ion size
Complexes and Thermodynamics • The presence and stability of complexes can be predicted using thermodynamics • Ca2+ + SO42-CaSO4(aq) • Kassoc = association constant (also Ka); also called stability constant • The larger the Kassoc , the more stable the complex • Kassochave smaller ranges compared to Keq, probably because bonds are weak • As with Keq, Kassoc values have been determined in the lab and are included in thermodynamic databases of geochemical models
Calculating complexes • Need complete chemical analyses to calculate concentrations of complexes • If important complexingspecies are missing, data interpretation may be in error • Speciation often a function of pH, so must have accurate field pH
Complexation Example • How much is Ca2+ decreased by complexation with SO42-?
pH • pH = -log[H+] • Many reactions involve H+ • Silicate and carbonate weathering • Sulfide weathering (acid mine drainage) • Dissociation of water molecule • Adsorption • Microbial processes; e.g., denitrification • Amphotericoxyhydroxides; Fe(OH)3, Al(OH)3 • Aqueous complexes
pH as Master Variable • pH is key parameter affecting species distribution • Therefore useful to consider activity of other species with respect to pH • pH is a “master variable”
Definitions • Acid is a compound that releases H+ when dissolved in water (proton donor) • Base is a compound that releases OH- when dissolved in water (proton acceptor) • Acids and bases can be liquids or solids • e.g., H2CO3 HCO3- + H+ • H2CO3 donates a proton when it dissociates = Acid • HCO3- + H+ H2CO3 • HCO3- accepts a proton = Base
Definitions • Some species can act as both acid and base, depending on reaction • HCO3-+ H+ H2CO3 • HCO3- accepts a proton = Base • HCO3- CO32- + H+ • HCO3- donates a proton = Acid
Acid/Base Strength • Strength is a measure of the tendency of an acid or base to give or accept protons • Strong acids/bases release all/most available H+/OH- • Strong acids almost completely dissociate in water; large Ka • HCl H+ + Cl- • Sulfuric acid: H2SO4 – acid rain (burning fossil fuels), AMD • Nitric acid: HNO3 – acid rain, nitrification (NH4+ NO3-) • Not usually large natural source of acid • Strong bases: hydroxides of alkali metals (Li, Na, K, Rb, Cs, Fr) and many alkaline earths (Mg, Ca, Sr, Ba, Ra)
Alkali Metals Alkaline Earths
Acid/Base Strength • Weak acids/bases release only a small fraction of available H+/OH- • Acetic acid (CH3COOH), H2CO3, H3PO4, H4SiO4 • Small Ka • Ammonium hydroxide (NH4OH), nickel hydroxide (Ni(OH)2) • In real world geochemistry, we’re mainly interested in weak acids and bases • Strength has nothing to do with concentration • HCl is still a strong acid even if it is greatly diluted • Acetic acid is still weak even if in a concentrated solution • Usually measure using Normality (N)
Important Acids in Groundwater • Carbonic acid: H2CO3 – CO2 • Dominant source of H+ in most groundwater • Silicic acid: H4SiO4 – mineral weathering • Acetic acid: CH3COOH – natural and anthropogenic (landfills); organic acid • Other organic acids (formic, oxalic) • Phosphoric: H3PO4
Dissociation of Silicic Acid • H4SiO4 ↔ H+ + H3SiO4- • 1st dissociation : • Ka1 is small • At pH 7: • H3SiO4-↔ H+ + H2SiO42- • 2nd dissociation: • Ka2 is very small
Dissociation of Silicic Acid (cont.) • H2SiO42-↔ H+ + HSiO43- • 3rd dissociation: • miniscule • HSiO43- ↔ H+ + SiO44- • 4th dissociation: < miniscule
Dissociation Reactions • Dissociation reactions reach equilibrium very quickly • e.g. CH3COOH CH3COO- + H+ • Ka= 1.76 x 10-5 at 25°C, 1 atm • Very small number, most remains undissociated • Knowing Kaand the initial concentration of CH3COOH, we can calculate how much dissociates
Example • Assume 0.1 moles of acetic acid is dissolved in 1 L H2O, determine fraction (x) that dissociates… • Assume γ = 1
Dissociation of Water • H2O H+ + OH-(or H2O + H+ ↔ H3O+) • Kw = [H+] [OH-] = 1 x 10-14 at 25°C • Remember that [H2O] = 1 • Small dissociation constant, but nearly unlimited source of H+ or OH- • For pure H2O at 25°C, [H+] = [OH-] = 10-7mol/L
Dissociation of Water • H2O H+ + OH- • pH = -log [H+] • Useful for reflecting on progress of chemical reactions • Easy to measure • pH = 7 for pure water at 25°C, 1 atm • Usually we consider pH values between 0 and 14 • pH for most natural waters is between 6 and 9 • We can also define pOH = -log [OH-] • Not widely used • At 25°C, pOH = 14 - pH
pH in the Environment • Weak acids/bases do not control the pH of the natural environment, but respond to it • pH is an environmental variable determined by all of the simultaneous equilibria existing in a given environment
Calculating pH of Acids and Bases • What is the pH of 0.1 M acetic acid? • CH3COOH CH3COO- + H+ • Recall we calculated that [H+] = 1.32 x 10-3 mol/L • pH = 2.88 • Note that even though acetic acid is a weak acid, the pH of a fairly concentrated solution of it is quite low
Polyprotic Acids/Bases • A weak acid or base that can yield 2 or more H+ or OH- per molecule of acid/base is polyprotic • H2S(aq) H+ + HS- • HS- H+ + S2- • Note that 1st reaction contributes much more H+ (K1 >> K2) • Other examples: H2CO3, H2SO4, H3PO4
Determining concentrations of species for polyprotic acids/bases • Dissolve 0.1 moles of H2S in pure water at 25°C; what are the concentrations of all the aqueous species? • Again, we’ll assume γ = 1
Sparingly Soluble Bases • Many bases do not readily dissolve in water • e.g., Brucite (Mg(OH)2) solubility • Mg(OH)2(s) Mg(OH)+ + OH-: Keq = 10-8.6 • Let’s calculate the concentrations of species as a function of pH
Brucite solubility • Plot activity of Mg species vs. pH to get an Activity Diagram • log [Mg(OH)+] = 5.4 – pH • log [Mg2+] = 16.8 – 2pH • get straight lines intersecting at pH = 11.4 • pH = 14 – 2.6 = 11.4 • Lines represent equilibrium between the two species/compounds on opposite sides • Mg2+ dominates at pH < 11.4 (most geologic environments)
Dissociation and pH • Dissociation of weak acids/bases controlled by pH • Rewrite mass action equations for H2S • H2S(aq) H+ + HS-
Dissociation and pH • Can do same for HS- vs. S2- • HS- H+ + S2- • Such relationships occur for all weak acids and bases • Knowing the total amount of S and pH, we can calculate activities of all species and generate curves
Total S = 10-4 M pH = 12.9 pH = 7
Total DIC = 10-1 M pH = 10.33 pH = 6.35