Carbonyl Compounds
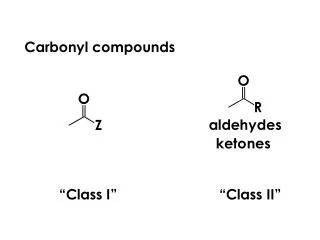
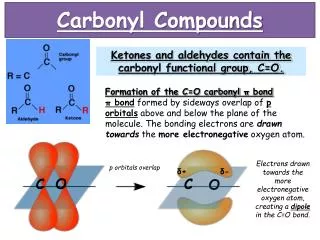
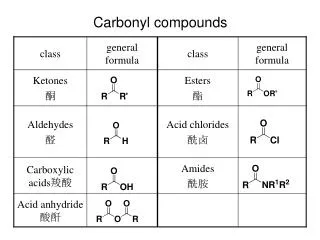
Carbonyl Compounds. Carbonyl group sp 2 hybridized carbon Coplanar bonds, 120 o bond angle p-p overlap bond. C. C. C. O. O. O. R’. H. R. R. Two types of compounds Aldehyde Ketone. Carbonyl compounds. -bond -ve inductive effect

Carbonyl Compounds
E N D
Presentation Transcript
Carbonyl group sp2 hybridized carbon Coplanar bonds, 120o bond angle p-p overlap bond C C C O O O R’ H R R Two types of compounds Aldehyde Ketone Carbonyl compounds
-bond -ve inductive effect • -bond readily polarizable (mesomeric effect) C+ C+ O- O- C+ O:- Polarity of carbonyl bond Dipole moment propan-1-ol 1.69D propanone 2.88 ethanal 2.72
C O H C OH C+ O :Br- H-Br Br Chemical reaction No electrophilic additions (why?)
Energetic Factor Big +ve, unstable product C OH H Br (Unstable) Chemical reaction No electrophilic additions (why?)
Big +ve Ea , unstable Transition state Kinetic Factor C+ OH Ea (Unstable) Chemical reaction No electrophilic additions (why?)
C O No nucleophilic substitution C O- Nu Nu-: Chemical reaction
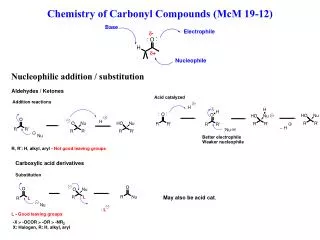
C O H/R’ H/R’ R C O- Nu -: R Nu H/R’ E+ R C E CN Nucleophilic Addition • Reactivity: • Electrophilicity of C atom • of the C=O group. • Strength of Nu:- • Steric effect at the carbonyl • group
H/R’ H/R’ C O R + H-CN C OH R CN Addition of HCN Reactivity: HCHO > CH3CHO > ArCHO > CH3COCH3 > CH3COR > RCOR > ArCOAr
C O Nucleophilic Addition Addition of HCN (using KCN+H+) H/R’ H/R’ slow R C O- • Note: • HCN is NOT • used. • Because • Toxic • Weak acid • little CN- CN-: R CN fast H/R’ HCN R C OH Cyanohydrin (2-hydroxynitrile) CN
H/R’ H/R’ H2O,H+ R R C OH C OH reflux COOH CN c.H2SO4,heat -C=C-COOH (,-unsaturated acid) Usefulness in organic synthesis • +1 carbon (longer carbon • chain) • 2 functional group
C-S bond is formed as S is more nucleophilic than O C O R’ R’ Room temp. Na+ R C O- .. Na+ HSO3- R SO3H R’ Sodium hydrogensulphate(IV) adduct, isolated as colourless crystals R C OH SO3- Na+ Addition of sodium hydrogensulphate(IV)
CH3 CH3 C2H5 C=O C=O C=O C=O C=O C=O C=O =O H CH3 89% CH3 35% 56% 36% (CH3)2CH (CH3)3C C2H5 Ph CH3 C2H5 CH3 CH3 1% 2% 12% 6% Addition of sodium hydrogensulphate(IV) • Limit to aliphatic aldehydes and sterically • unhindered ketones (steric effect) % product from 1mol NaHSO3 in 1 hour:
Addition of sodium hydrogensulphate(IV) • Reversible (can be reversed by aq. Alkali or • acid by shifting eqm. position to LHS by • HSO3- + H+=> SO2 , HSO3- + OH- => SO32-) • Use to purify liquid or gaseous carbonyl • compounds which are difficult to purify • by direct recrystallization.
H R’ C O R’ HO N+ C O- H H R HO N: R H (Hydroxylamine) H R’ R’ -H2O HO N C OH HO N C R R (Oxime) Addition-elimination (condensation)
R’ C O R’ .. .. .. .. NO2 NO2 NH-NH2 NH-N=C R R NO2 NO2 2,4-dinitrophenylhydrazine (Brady’s reagent) 2,4-dinitrophenylhydrazone (yellow or light orange crystals) Addition-elimination
Phenylhydrazone • Products have sharp and characteristic • melting point. • Used as the identification of the original • aldehyde and ketone • Note: • NH3 does not react • Predict the product obtained by • adding H2N-NH2 to propanal.
Oxidation • KMnO4/H+ , K2Cr2O7/H+ (Strong oxidizing agent) • RCHO => RCOOH • RCH2COCH2R’ => RCOOH + R’ CH2COOH + • RCH2COOH + R’COOH • C6H5CHO => C6H5COOH requiring reflux for hours
Oxidation • Tollen’s reagent (silver mirror test) • Reagent: 2Ag+ + 2OH- => Ag2O + H2O • Ag2O + 4NH3 + H2O => 2Ag(NH3)2OH • 2[Ag(NH3)2]+ + RCHO + 3OH- • => RCOO- +2H2O + 4NH3 + 2Ag (mirror) No reaction with ketone (Tollen’s reagent is a mild O.A.)
Oxidation • Fehling’s reagent • Reagent: alkaline solution of copper(II) tartrate • RCHO + 2Cu2+ + 5OH- => RCOO- + 3H2O + Cu2O • (Fehling) (brick-red) • Note: No reaction with Ketones and Aromatic Aldehydes
R O- R R OH H+ C C O C H/R H/R H H/R H H- Reduction Reducing agent: LiAlH4 Lithium Tetrahydridoaluminate NaBH4 Sodium Tetrahydridoborate Both equivalent to a source of hydride ion, H-.
R Reducing agent: H2/Ni, similar to alkene C O H/R H2/Ni RCH2OH Reduction LiAlH4 must be kept dry i.e. in solution of dry ether LiBH4 is less powerful, can be used in aqueous solution.
OH- X2 -H2O -X- CX3 X2 X2 OH- HO C O- -X- -X- CH3 -CH2 CH2X CX3 H/R C C C C O O O O H/R H/R H/R H/R CX3- + R/HCOOH CHX3 + RCOO- Tri-iodomethane reaction (RCOCH3 + I2/OH- => RCOO- + CHI3 ,yellow ppt.)