Percentage Composition
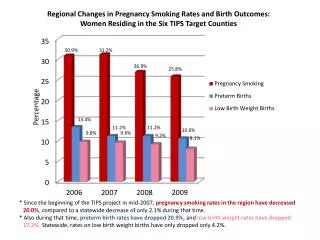
Percentage Composition. Recall that the FORMULA WEIGHT [or molar mass] of a substance is the MASS IN GRAMS of ONE MOLE of particles of that substance. One mole contains 6.022 X 10 23 particles. This number is called AVOGADRO’S NUMBER .

Percentage Composition
E N D
Presentation Transcript
Recall that the FORMULA WEIGHT [or molar mass] of a substance is the MASS IN GRAMS of ONE MOLE of particles of that substance. • One mole contains 6.022 X 1023particles. • This number is called AVOGADRO’S NUMBER.
Within the mass of a mole of any compound, EACH ELEMENT contributes a CERTAIN PERCENTAGE to the OVERALL MASS. • We can calculate these percentages. This is known as PERCENTAGE COMPOSITION.
Calculating Percentage Composition • Calculating Percentage Composition is easy: • 1. Find the FORMULA WEIGHT of the compound. • 2. Calculate the MASS PERCENTAGE of EACH ELEMENT in the compound.
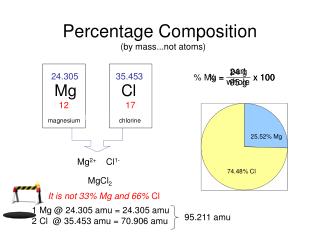
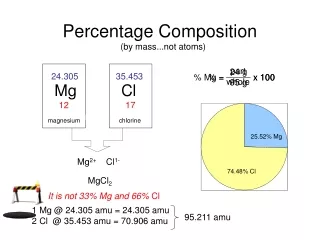
SAMPLE PROBLEM • Calculate the Percentage Composition of Potassium Cyanide, KCN. • 1. Find the FORMULA WEIGHT for KCN: • 1 K X 39.098 g = 39.098 g • 1 C X 12.011 g = 12.011 g • 1 N X 14.007 g = 14.007 g • Add Results 65.116 g/mol
Now we will use the data from EACH ELEMENT to find the Percentage Composition: • 1 K X 39.098 g = 39.098g • 1 C X 12.011 g = 12.011g • 1 N X 14.007 g = 14.007 g • 65.116 g/mol • We will use the TOTAL MASS of EACH ELEMENT and divide it by the FORMULA WEIGHT, then MULTIPLY the results by 100%!
Mass of K • %K = 39.098 g X 100% = 60.04% K • 65.116 g/mol • Formula Weight of KCN • %C = 12.011 g X 100% = 18.45 % C • 65.116 g/mol • % N = 14.007 g X 100% = 21.51 % N • 65.116 g/mol
Finally, CHECK YOUR MATH! Add all the PERCENTAGES together….they should be VERY CLOSE TO 100%! • 60.04 % + 18.45% + 21.51% = 100%
PRACTICE PROBLEM • Calculate the PERCENTAGE COMPOSITION of • Calcium Carbonate, CaCO3. • 1 Ca X 40.08 g = 40.08 g • 1 C X 12.011 g = 12.011 g • 3 O X 15.999 g = 47.997 g • 100.088 g/mol
% Ca 40.08 g X 100% = 40.08% Ca • 100.088 g/mol • % C 12.011 g X 100% = 12.01% C • 100.088 g/mol • % O 47.997 g X 100% = 47.99% O • 100.088 g/mol • 40.08 % + 12.01% + 47.99% = 100.08% = 100%
One Your Own • Find the Percentage Composition of Aluminum Phosphate, AlPO4. • ___ Al x ______ g = ________g • ___ P x ______ g = ________ g • ___ O x ______ g = ________ g • _______ g/mol 1 26.982 26.982 1 30.974 30.974 4 15.999 63.996 121.95
26.982 22.13 121.95 30.974 25.40 121.95 63.996 52.48 121.95 22.13 25.40 52.48 100.01 • % Al ________ x 100% = _______ %Al • % P ________ x 100% = _______ %P • % O ________ x 100% = ________ % O • ____% Al + ____% P + ____ %O = _____%