Understanding Periodic Table Trends: Atomic Radius, Ionization Energy, Electron Affinity, and Electronegativity
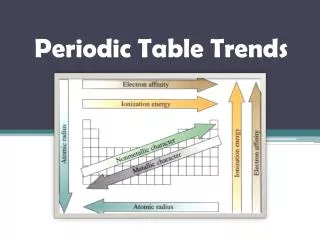
This overview discusses key trends in the periodic table, focusing on atomic radius, first ionization energy, electron affinity, and electronegativity. As you move down a group, atomic radius increases due to additional energy levels. Conversely, moving across a period causes atomic radius to decrease because the increasing nuclear charge pulls electrons closer. First ionization energy decreases down a group but increases across a period. Electron affinity generally decreases down a group and increases across a period, while electronegativity follows a similar trend, decreasing down and increasing across.

Understanding Periodic Table Trends: Atomic Radius, Ionization Energy, Electron Affinity, and Electronegativity
E N D
Presentation Transcript
Atomic Radius • As you move down a group, atomic radius increases • The number of energy levels increases as you move down a group as the number of electrons increases. Each subsequent energy level is further from the nucleus than the last. Therefore, the atomic radius increases as the group and energy levels increase
Atomic Radius • As you move across a period, atomic radius decreases • As you go across a period, electrons are added to the same energy level. At the same time, protons are being added to the nucleus. The concentration of more protons in the nucleus creates a "higher effective nuclear charge." In other words, there is a stronger force of attraction pulling the electrons closer to the nucleus resulting in a smaller atomic radius.
First Ionization Energy • The energy required to remove the outermost (highest energy) electron from a neutral atom in its ground state • As you move down a group, first ionization energy decreases • Electrons are further from the nucleus and thus easier to remove the outermost one. "SHIELDING" - Inner electrons at lower energy levels essentially block the protons' force of attraction toward the nucleus. It therefore becomes easier to remove the outer electron
First Ionization Energy • As you move across a period, first ionization energy increases • As you move across a period, the atomic radius decreases, that is, the atom is smaller. The outer electrons are closer to the nucleus and more strongly attracted to the center. Therefore, it becomes more difficult to remove the outermost electron
Electron Affinity • The electron affinity of an atom or molecule is defined as the amount of energy released when an electron is added to a neutral atom or molecule to form a negative ion. • As you move down a group, electron affinity decreases • This is caused by the increase in atomic radius
Electron Affinity • As you move across a period, electron affinity increases • This is caused by the decrease in atomic radius
Electronegativity • Electronegativity measures an atom's strength to attract and form bonds with electrons • As you move to the right across a period of elements, electronegativity increases • When the valence shell of an atom is less than half full it is easier to lose an electron. Conversely, when the valence shell is more than half full, it is easier to pull an electron into the valence shell than to donate one.
Electronegativity • As you move down a group, electronegativity decreases • The atomic number increases down a group and thus there is an increased distance between the valence electrons and nucleus, or a greater atomic radius