9.0 Alkaloids
9.0 Alkaloids. RA Macahig FM Dayrit. Introduction.

9.0 Alkaloids
E N D
Presentation Transcript
9.0 Alkaloids RA Macahig FM Dayrit
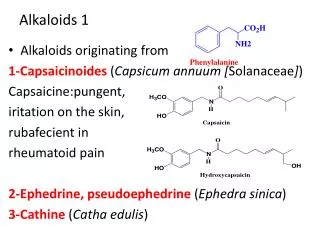
Introduction • Among all the groups of natural products, alkaloids have the most colorful history, having achieved the most fame and notoriety as drugs. Where alkaloids occur, they tend to dominate the biological activity. Despite their relatively limited distribution, the alkaloids probably have the most significant impact in human history particularly in medicine, social issues, economics and politics. • In 1819, Carl Friedrich Meissner, a pioneering German pharmacist, coined the term “alkaloid” which referred to any natural product with the characteristic presence of the basic nitrogen atom, excluding peptides. (Amides, however, are generally included.) 9.0 Alkaloids (Dayrit)
Introduction • Because many alkaloids can be purified from crude extracts by acid-base extraction and recrystallization, these were the first natural products to be purified, characterized and commercialized. • The powerful and immediate effects of alkaloids are thought to be due to the presence of the cationizable N-atom which gives it lipid- and water-soluble characteristics, and enables it to cross membrane barriers more readily. 9.0 Alkaloids (Dayrit)
Introduction • Many of the earliest pure compounds to be used as drugs developed were alkaloids: • Cocaine: anaesthetic, from South American Erythroxylum coca • Quinine: antimalarial, from the bark of the Cinchona tree • Morphine: anaesthetic, from opium (Papaver somniferum) • Emetine: for amoebiasis, from ipecac, the powdered roots (Cephaelis species) • Strychnine: poison, from the seeds of Strychnos nux-vomica • The alkaloids have a relatively limited distribution in nature compared with the other natural product groups. Alkaloids were originally thought to be uniquely plant products until the 1950s when several alkaloids were isolated from bacteria, fungi and algae, insects, and amphibians. • A number of fungi produce toxic alkaloids, notably Claviceps purpurea. 9.0 Alkaloids (Dayrit)
Introduction • In the plant kingdom, the angiosperms produce alkaloids: Apocynaceae, Papaveraceae, Rubiaceae, Ranunculaceae, Solanaceae, and Berberidaceae. Among the monocots, only the Amaryllidaeae and Liliaceae produce alkaloids. • The discovery of many alkaloids are associated with anthropological explorations. Here, a Kamsá youth from a Brazilian tribe holds a blossom of Culebra borrachera which is a toxic psychoactive plant. It contains tropane alkaloids that can induce a frightening state of psychotic delirium, and ultimately stupor and death. (cms.herbalgram.org) 9.0 Alkaloids (Dayrit)
Introduction • Some animals, notably some soft corals and frogs produce highly bioactive alkaloids. In some cases, however, the alkaloids were found to have been ingested in the diet by the organism and then modified for use. Well-known examples are the pyrrolizidine alkaloids in caterpillars and moths. Alkaloids are much less common in mammals. Intermedine (I) and lycopsamine (II) ingested from the leaves of Mikania scandens monocrotaline (III) 9.0 Alkaloids (Dayrit)
Introduction • There are four major groups of nitrogen-containing organic compounds in biological systems: the amino acids (peptides and proteins), the nucleoside bases (DNA and RNA), the porphyrins; and the alkaloids. The first 3 groups are primary metabolites; the alkaloids are secondary metabolites. • Alkaloids are defined simply as nitrogen-containing natural products. In terms of chemical structure, alkaloids can be classified into the following: 2, 3, and 4 alkyl amines; and heterocyclic amines (e.g., pyrrolidine, pyridine, indole, quinoline, and isoquinoline). 9.0 Alkaloids (Dayrit)
Ajmaline, antiarrythmic that functions by inhibition of glucose uptake by heart tissue mitochondria • Atropine (hyoscyamine), anticholinergic, antidote to nerve gas poisoning • Caffeine, widely used central nervous system stimulant • Camptothecin, potent anticancer agent • Cocaine, topical anesthetic, potent central nervous system stimulant, and adrenergic blocking agent, drug of abuse (Kutchan, The Plant Cell, 7, 1059-1070, July 1995)
Codeine, relatively nonaddictive analgesic and antitussive; Coniine, first alkaloid to be synthesized, extremely toxic, causes paralysis of motor nerve endings, used in homeopathy • Emetine, orally active emetic, amoebicide; morphine, powerful narcotic analgesic, addictive drug of abuse; Nicotine, highly toxic, causes respiratory paralysis, horticultural insecticide • Pilocarpine, peripheral stimulant of the parasympathetic system, used to treat glaucoma • Quinine, traditional antimalarial, important in treating Plasmodium falcipafum strains that are resistant to other antimalarials
Sanguinarine, antibacterial showing antiplaque activity, used in toothpastes and oral rinses • Scopolamine, powerful narcotic, used as a sedative for motion sickness • Strychnine, violent tetanic poison, rat poison, used in homeopathy; Taxol, antitumor agent • (+)-Tubocurarine, nondepolarizingmuscle relaxant producing paralysis, adjuvant to anesthesia • Vinblastine, antineoplastic that is used to treat Hodgkin’s disease and other lymphomas.
Survey of some well-known alkaloids. 9.0 Alkaloids (Dayrit)
Merck, one of the world’s largest pharmaceutical companies, made its initial fortune from cocaine, morphine, and codeine. 9.0 Alkaloids (Dayrit)
Introduction • Many alkaloids are toxic in the cytoplasm, even in the plants that produce them. Their accumulation, therefore, requires a well-regulated and compartmentalized system. The most common storage organ is the vacuole. • In some plants, alkaloids are synthesized in one part of the plant and then are transported to another part for storage. In tobacco, for example, alkaloids (e.g., nicotine) are synthesized in the roots and are then transported via the xylem to the leaves where they are accumulated. 9.0 Alkaloids (Dayrit)
Introduction Among the natural products groups, the biochemistry of alkaloids is the most complex. • Many alkaloids are mixed metabolites. • Enzymes involved in alkaloid biosynthesis are associated with diverse subcellular compartments including the cytosol, vacuole, tonoplast membrane, endoplasmic reticulum, chloroplast stroma, thylakoid membranes, and perhaps unique biosynthetic or transport vesicles. • Localization studies have shown that sequential alkaloid biosynthetic enzymes can also occur in distinct cell types, suggesting the intercellular transport of pathway intermediates. (PJ Facchini, Ann. Rev. of Plant Physiol. and Plant Mol. Bio., Vol. 52: 29-66) 9.0 Alkaloids (Dayrit)
Overview Despite the wide variety and complex structures observed among the alkaloids, most of the compounds from this group are formed from only a handful of starting materials. The nitrogen and the initial carbon skeleton are derived from the following: A. aliphatic amino acids: lysine, glutamic acid, and ornithine; B. aromatic amino acids: phenylalanine, tyrosine, and tryptophan; and C. others: secologanin: terpene-derived C10 unit. 9.0 Alkaloids (Dayrit)
Overview • The major reactions in alkaloid biosynthesis are common biosynthetic mechanisms: • Oxidation: epoxidation, aromatic hydroxylation, etc. • Oxidation: dehydrogenation (-2[H]); in the case of the alkaloids, this includes the conversion of amine imine. • Reduction: hydrogenation (+2[H]); in the case of the alkaloids, this includes the conversion of imine amine. • Phenolicdimerization by radical coupling. • Decarboxylation, in particular in the conversion of amino acid to amine. 9.0 Alkaloids (Dayrit)
Reactions which are particular to alkaloid biosynthesis. 9.0 Alkaloids (Dayrit)
tryptophan Reactions which are particular to alkaloid biosynthesis. 9.0 Alkaloids (Dayrit)
Aliphatic alkaloids • The aliphatic alkaloids can be classified into three main types, depending on its biogenesis: • the amino acid ornithine • the amino acid lysine • the polyketide pathway with the nitrogen atom being introduced in a late step. 9.0 Alkaloids (Dayrit)
Aliphatic alkaloids from ornithine Glutamic is transformed into ornithine by addition of another CH2 unit. Therefore, in a way, glutamic acid is the original precursor and ornithine is the immediate precursor. Decarboxylation and loss of one nitrogen leads to formation of the pyrrolidine ring. There are three main types: pyrrolidine (monocyclic), tropane (bicyclic) and pyrrolizidine (fused). 9.0 Alkaloids (Dayrit)
Ornithine comes from glutamic acid. 9.0 Alkaloids (Dayrit)
Pyrrolidine alkaloids from ornithine. Labeling studies show that although pyrrolidine itself is symmetric, the biosynthesis is regiospecific. This suggests that once ornithine is held by the enzyme, biosynthesis proceeds without release of any of the intermediates. 9.0 Alkaloids (Dayrit)
Tropane alkaloids from ornithine. Consistent with what has been observed, labeling shows that the biosynthesis is regiospecific. 9.0 Alkaloids (Dayrit)
Pyrrolizidine alkaloids from ornithine. Pyrrolizidine alkaloids are common in the butterflies Senecio and Crotolaria species. 9.0 Alkaloids (Dayrit)
Pyrrolizidine alkaloids are converted into aphrodisiac substances which the male butterflies store in its wing hair pencils. These compounds also protect the plants against feeding by mammals because these compounds are converted in the liver into toxic and carcinogenic compounds. 9.0 Alkaloids (Dayrit)
Polyhydroxylated cycloalkyl alkaloids found in the leaves, flowers and seeds of Ipomoea carnea (Convolvulaceae) cause natural intoxication of livestock by inhibiting key digestive enzymes. Alkaloids 1 and 2 are powerful inhibitors of lysosomal a-mannosidase; 3, 4, and 6 showed potent inhibitory activity toward rat lysosomal b-glucosidase; and alkaloid 5 was a moderate inhibitor of a- and b-mannosidases.(Haraguchi, et al., J. Agric. Food Chem. 2003, 51, 4995-5000.) 9.0 Alkaloids (Dayrit)
Aliphatic alkaloids from lysine Lysine is modified following an analogous pathway to ornithine. There are many similarities between the ornithine-derived and lysine-derived alkaloids. The alkaloids produced are the 6-membered piperidine, homotropane and quinolizidine structures. 9.0 Alkaloids (Dayrit)
Alkaloids from lysine. 1-Piperidine and pellieterine are key intermediates to this group of alkaloids.
Quinolizidine alkaloids have the characteristic fused 6,6-bicyclic group and are derived from lysine. Lupinine is a dimeric metabolite while sparteine is trimeric. 9.0 Alkaloids (Dayrit)
Aliphatic alkaloids from polyketides Some aliphatic alkaloids are derived from the polyketide pathway. The biogenesis of these alkaloids can be determined by studies using labeled acetyl CoA. 9.0 Alkaloids (Dayrit)
Polyketide-derived piperidine alkaloids. Some alkaloids having the piperidine-type structure are not derived from lysine. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine and tyrosine The aromatic alkaloids derived from phenylalanine and tyrosine form a diverse and often structurally complex group of metabolites. By tradition, these alkaloids are identified according to plant family, of which the best known are: Papaveraceae, Morphinan, Erythria, Berberidaceae, Amaryllidaceae. Structurally and biosynthetically, there are six main groupings: 1. Phenylethylamines 2. Phenylethylamine + alkyl aldehyde or ketone 3. Phenylethylamine + benzaldehyde (C6-C1) 4. Phenylethylamine + C6-C2 5. Phenylethylamine + C6-C3 6. Phenylethylamine + secologanin 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine/tyrosine. Structurally, there are six main types. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine/tyrosine. Structurally, there are six main types. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine/ tyrosine: Simple phenylethylamines. Biosynthesis of this group involves simply loss of the carboxylic acid carbon. Some important members of this group are the hallucinogenic compound mescaline and the drug epinephrine. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine/ tyrosine: Condensation of phenylethylamine with alkyl aldehydes or ketones. This group of alkaloids is formed via a Pictet-Springler or Bischler-Napieralski condensation. The alkaloids shown here are found in the hallucinogenic peyote cactus plant. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine / tyrosine + Ar-C1. Phenylethylamine couples with Ar-C1 (benzaldehyde) via a Pictet-Springler condensation. This is followed by oxidation of the phenol. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine / tyrosine + Ar-C1. The two oxidized rings can couple via different folding conformations leading to para-para, para-ortho or ortho-para coupling. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine / tyrosine + Ar-C1. (overview) Coupling of the radical intermediates in different folding conformations leads to para-para, para-ortho and ortho-para couplings. This is a theme that is repeated for other alkaloids with similar structural characteristics. The alkaloid families that comprise this group include the Amaryllidaceae and Mesembrine species. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine / tyrosine + Ar-C2. Condensation of phenylethylamine with an Ar-C2 group, such as phenylpyruvic acid, yields the benzyltetrahydro isoquinoline structure. These alkaloids are characteristic of the Papaveraceae. Reticuline is a key intermediate of this group. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine / tyrosine + Ar-C2. The Aprophine alkaloids are produced by oxidation of reticuline. Various isomeric radical intermediates are formed.
Alkaloids from phenylalanine / tyrosine + Ar-C2. Four regiochemical couplings are observed: ortho-ortho, ortho-para, para-ortho, and para-para.
Alkaloids from phenylalanine / tyrosine + Ar-C2. The Aprophine alkaloids are produced by radical coupling of the benzylisoquinoline system of reticuline.
Schematic presentation of the biosynthesis of codeine, laudanine, and (S)-scoulerine from (S)-norcoclaurine in the opium poppy. The cellular localizations of the enzymes indicated have been determined experimentally. (Jorgensen et al., Curr Opinion in Plant Biol 2005, 8:280–291) 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine/tyrosine + Ar-C2. The morphine alkaloids are produced from thebaine. Note that the level of methylation decreases towards the end of the biosynthetic sequence from thebaine codeine morphine. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine / tyrosine + Ar-C2.Coupling using the N-methyl group. 9.0 Alkaloids (Dayrit)
Alkaloids from phenylalanine / tyrosine + Ar-C3.This group is homologous to the benzyltetrahydro-isoquinolines (dopamine + C2). The biosynthetic steps are assumed to be similar. 9.0 Alkaloids (Dayrit)