Understanding Thermodynamics: Laws, Energy Conservation, and Entropy
This chapter delves into the fundamental concepts of thermodynamics, beginning with absolute zero, the lowest limit of temperature where atomic motion ceases. It explores the first law of thermodynamics, emphasizing the conservation of energy as heat transforms into work within systems. The adiabatic processes, where no heat is exchanged, are examined, along with the second and third laws, highlighting the directionality of heat flow and the impossibility of reaching absolute zero. Additionally, it covers heat engines' operation, efficiency, and the concept of entropy as a measure of disorder in energy distribution.

Understanding Thermodynamics: Laws, Energy Conservation, and Entropy
E N D
Presentation Transcript
24.1 – absolute zero • No upper limit of temperature • Since atomic motion determines temp, there is a lower limit – absolute zero • No atomic motion, no further energy could be extracted • -273 °C = 0 kelvin = 0 K • No negative numbers in kelvin scale
24.2 – First law of thermodynamics • In past, heat was considered a fluid – a caloric • James Joule showed it was just energy • First law – heat added to system can 1. increase internal energy or 2. do work or 3. both • System = a collection of atoms, molecules or equipment • ΔU = Q + W • First Law = Conservation of Energy
Work - the system expanding or contracting - can change the internal energy as well • Work done to the system, ↑ internal energy • Work done by system, ↓ internal energy • Some work can generate heat and would have to be taken into account
24.3 – adiabatic process • Gases are compressed or expanded so no heat is exchanged • Achieved in two ways: volume changes very quickly (a car engine) or system is insulated • Work goes entirely into/out of internal energy (↑or↓ temp), none gets leaked away (from system) as heat
Air temperature (internal energy) can be changed by: • Heat exchange – solar/terrestrial radiation, conduction with ground, or phase changes • Pressure changes • ΔT αΔP
In our atmosphere, as air rises, pressure is reduced, causing air to expand and cool • Adiabatic expansion, no heat is exchanged • The air (system) does work on surroundings (↑ volume), losing internal energy (↓ temperature) • About 10°C per 1 km
As air rises, water vapor condenses and it rains • The reverse happens as well – air warms as it descends and becomes dry • Called a chinook – from Rocky Mtns into Great Plains • Why warm and dry east of Sierras
24.4 – second and third laws of thermodynamics • Second law – heat always flows from hot object to cooler one • First law says nothing about direction, as long as energy is conserved • Third Law – no system can reach absolute zero
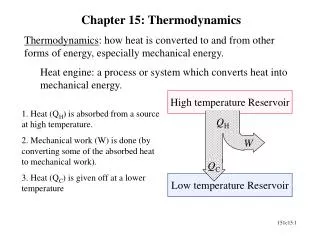
24.5 – heat engines & the 2nd law • Heat engine – a device that converts heat energy into work • Steam, internal combustion • Heat must flow from high temp. source, Thot(reservoir) to a low temp. sink, Tcold • In process, some energy can be extracted and used to do work
Heat energy is absorbed by engine, ↑ internal energy • Some of this is used to do work • The rest must be expelled • No heat engine can convert all heat energy into work – another statement of 2nd law • There is an upper limit to how efficient a heat engine can be • Called Carnot efficiency, only dependent upon high/low temps
In calcs. temps. must be in kelvins • The greater the temp difference, the higher efficiency • Why engines must run so hot
A steam turbine is a heat engine • Hot steam exerts more pressure on the “front” side of blades than the cooled steam on the “back” side does • Therefore, it does work • But, there must be ejected cooler steam • Otherwise, the turbine would just continue to warm until Thot= Tcold, no pressure diff.
24.6 – order tends to disorder • All heat engines produce waste heat from organized fuel • The waste heat has less ability to do work – it degenerates • The energy is less organized, it has more random motion • All natural systems proceed to greater disorder • Nature proceed to what is most likely to happen • Work must be done to reorganize energy
24.7 - entropy • A measure of disorder, > disorder = > entropy • Nature always proceeds to greater entropy • A system allowed to distribute its energy, ↑ entropy, leaving less energy available to do work • It’s possible to decrease entropy for one system, but at the expense of > ↑ in another