Volume of Titrant
510 likes | 713 Views
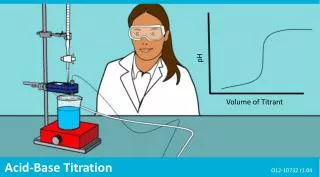
pH. Volume of Titrant. Acid-Base Titration. O12-10732 r1.04. Acid-Base Titration. Introduction. Each page of this lab that contains the symbol

Volume of Titrant
E N D
Presentation Transcript
pH Volume of Titrant Acid-Base Titration O12-10732 r1.04
Acid-Base Titration Introduction Each page of this lab that contains the symbol should be inserted into your journal. After completing a lab page with the snapshot symbol, tap (in the upper right hand corner) to insert the page into your journal. Journals and Snapshots The Snapshot button is used to capture the screen. The Journal is where snapshots are stored and viewed. The Share button is used to export or print your journal to turn in your work. Note: You may want to take a snapshot of the first page of this lab as a cover page for your Journal.
Acid-Base Titration Lab Challenge Many foods and household products contain acids and bases. Knowing if a sample is acidic or basic is only part of the question. How do you determine the actual amount of acid or base dissolved in a solution?
Acid-Base Titration Background Titrant • Titration is a common quantitative laboratory method used to determine the concentration of a reactant. • A reagent of known concentration, called the titrant, is used to react with a measured volume of another reactant, called the analyte. • The point at which the number of moles of the titrant and the analyte are equal is called the equivalence point. On a titration curve, the equivalence point is the point where the slope is the steepest. Analyte Equivalence point
Acid-Base Titration ...Background • When a basic solution is added to an acidic solution of unknown concentration, hydroxide ions (OH-) from the basic solution react with hydronium ions (H3O+) from the acidic solution to form neutral water and a salt. The type of salt formed depends on the acid and base used. • This is called a neutralizationreaction. + Neutral solution Basic solution Acidic solution
Acid-Base Titration Self-Check • Titrations are used to determine the _____________ of a reactant. • density • pH • mass • concentration This image is a reminder that you need to tap to take a snapshot of this page after you have entered your response.
Acid-Base Titration Safety • Follow all common laboratory safety procedures • Sodium hydroxide, hydrochloric acid, and acetic acid are corrosive irritants. Avoid contact with the eyes and wash hands after handling. • When mixing acids with water, always add the acid to the water, not the other way around, as the solution can get hot enough to boil. • Be sure that all acids and bases are neutralized before being disposed of down the drain. BE SAFE Always wash hands to remove residue before leaving
Acid-Base Titration Materials and Equipment Collect all of these materials before beginning the lab. • Drop counter • pH sensor • Magnetic stirrer • Micro stir bar • Beakers (2), 50-mL • Buffer solution pH 4, 25 mL • Buffer solution pH 10, 25 mL • Funnel • Graduated cylinder, 100-mL
Acid-Base Titration ...Materials and Equipment Also collect these additional materials before beginning. • Buret, 50-mL • Buretclamp • Ring stand • Right-angle clamp • Beakers (2), 250-mL • Transfer pipet • Volumetric pipetor graduated cylinder, 10-mL • Wash bottle filled with distilled (deionized) water
Acid-Base Titration ...Materials and Equipment Also collect these additional materials before beginning. • Waste container • deionized (distilled water), 200 mL • Standardized sodium hydroxide solution, 100 mL • Hydrochloric acid solution, 10 mL • Acetic acid solution, 10 mL Distilled Water waste waste
Acid-Base Titration Sequencing Challenge The steps to the left are part of the procedure for this lab activity. They are not in the right order. Determine the correct sequence of the steps, then take a snapshot of this page. A. Setup the titration equipment and calibrate the pH sensor. B. Measure and record the starting volumes of NaOH and HCl. Start collecting data and then release the titrant. C. Thoroughly clean the equipment and then repeat the procedure using acetic acid. D. When the pH is 12, close the stopcock and then stop collecting data. Record the final volume of NaOH.
Acid-Base Titration Setup: HCl Titration • Connect the pH sensor to the data collection system. • Calibrate the pH sensor: • Pour approximately 25 mL of the pH 4 buffer solution in a 50-mL beaker. • Pour approximately 25 mL of the pH 10 buffer solution in a second 50-mL beaker. • Remove the pH sensor from its electrode storage bottle and rinse it with distilled water to thoroughly clean the sensor. • Read the instructions on how to calibrate the pH sensor on the next page. Buffer solutions and pH sensor
Acid-Base Titration To Calibrate the pH Sensor: • Calibration Point 1: • Place the pH probe in a pH 4 buffer solution. • Enter 4.0 as the pH in the Standard Valuebox under Calibration Point 1. • Tap Read From Sensor underCalibration Point 1. • Rinse the pH probe thoroughly using distilled water. • 4. Calibration Point 2: • Repeat the process used in calibration point 1 using a pH 10 buffer solution. • Tap OK to exit the calibration screen and then tap OK again to return to the lab. Note: During the calibration process you will not be able to return to this page. • 1. Open the Calibrate Sensor screens: • Tap • Tap CALIBRATE SENSOR 2.Ensure that the correct measurements are selected: • Sensor: (name of sensor) • Measurement: pH • Calibration Type: 2 point • b. Tap NEXT
Acid-Base Titration Setup: HCl Titration Q1: Using the terms accuracy and precision, explain why it is necessary to calibrate the pH sensor. • Connect the drop counter to the data collection system. • Assemble the titration apparatus. Buretclamp Buret pH sensor Ring stand Right-angle clamp Drop counter Micro stir bar Beaker, 250-mL Magnetic stirrer
Acid-Base Titration Setup: HCl Titration Q2: Why is it necessary to rinse the buretwith standardized NaOH solution? • Use a transfer pipet to rinse the inside of the buret with several milliliters of the standardized sodium hydroxide (NaOH) solution. • Drain the NaOH from the buret into the waste container. • Repeat this process two more times. • Make sure the stopcock on the buret is in the "off" position and then use a funnel to fill the buret with ~50 mL of the standardized NaOH solution (titrant). On position Off position
Acid-Base Titration Setup: HCl Titration Q3: Why is it be necessary to start your titration over again if you accidently allow the titrant to flow out of the stopcock instead of drop? • Practice adjusting the stopcock on the buretso that the titrant goes through the drop counter in distinguishable drops that fall at about 2 to 3 drops per second. • Note:It is important that you have good control of adjusting the stopcock. If you accidently open the stopcock too far and the NaOH flows out (as opposed to drops out), then you will have to start over. • Close the stopcock and then remove the waste container.
Acid-Base Titration Setup: HCl Titration Q4: Record the initial volume of NaOH and the concentration of the standardized NaOH solution in the text box below. • Read the initial volume of the titrant in the buretto 0.01 mL. • Using the 100-mL graduated cylinder measure 100 mLof distilled water and add it to a clean 250-mL beaker. • Using the 10-mL graduated cylinder or volumetric pipetmeasure 10.0 mL of hydrochloric acid (HCl) and add it to the 100 mLof distilled water. Record the exact volume of HCl added in the text box on the next page. • Caution:Always add acid to water.
Acid-Base Titration Setup: HCl Titration Q5:Record the volume of HCladded to the beaker in the text box below. • Add the pH sensor, micro stir bar, and 250-mL beaker containing the acidic solution to the titration apparatus. • Turn on the magnetic stirrer and begin stirring at a slow to medium speed. Note:Make sure the bulb of the pH sensor is fully submerged.
Acid-Base Titration Collect Data: • Tap to start collecting data. • Carefully open the stopcock on the buretso that 2 to 3 drops per second are released. • Continue to collect data until the pH of the solution is 12. Continue to the next page.
Acid-Base Titration Q6: Why is it necessary to stir the solution during a titration? Q7: What substances are being formed in the beaker?
Acid-Base Titration Collect Data: • When the pH of the solution is 12 close the stopcock. • Continue to the next page.
Acid-Base Titration Collect Data: • Tap to stop data collection. • 6. Record the final volume of NaOH in the buret.
Acid-Base Titration Data Analysis: HCl • * To View a Page in the Journal: • Tap to open the Journal screen. • Tap or to scroll through the thumbnail size pages of the journal. • Tap the thumbnail image of the page to view it. • Determine the total volume of NaOH used in the titration by following steps a-c below. • Enter the final volume of NaOH in the text box on the right. • Record the initial volume of NaOH in the text box on the right • Subtract the initial volume of NaOH from the final volume of NaOH and record the total volume in the text box on the right. Volume NaOH = Final Volume NaOH - Initial Volume of NaOH • Note:Initial and final volumes of NaOH were recorded earlier • in the journal. Refer to the journal to get these numbers.*
Acid-Base Titration Data Analysis: HCl 2. Create a calculation to convert drop count to volume (mL). * CalcVolume1 = [drop count]*(volume of titrant/final drop count) *To Create a Calculation: • Tap to open the Experiment Tools screen. • Tap CALCULATED DATAto open the calculator. • Enter the calculation in the space provided. • Tap Measurements to insert collected data into the calculation. Note: [drop count] = collected data volume of titrant = total volume of NaOH used (it is in your journal) final drop count = the total number of drops added (it is displayed on the right)
Acid-Base Titration 3. Change the x-axis to display CalcVolume1 of NaOH. • Tap then to open the Graph Properties screen. • Tap the Measurement box for the x-axis and tap CalcVolume1. Then tap OK. Tap to scale the axes. 4. Display the volume of NaOH at the equivalence point. The equivalence point is the point on the curve with the greatest slope and is found using the slope tool. • Tap then to see the slope. • Tap or buttons to move to nearby points. • Tap .
Acid-Base Titration Data Analysis: HCl • Calculate the molar concentration of HCl. Follow the steps below: • Determine the number of moles of NaOH added using the volume of NaOH at the equivalence point and the molarity of the standardized NaOH solution. • Convert from moles of NaOH to moles of HCl using the balanced chemical equation. • Use the moles of HCl and the starting volume of HCl to determine the molarity of the acid. • Note: the values need for the calculations above can be found in your journal .
Acid-Base Titration Setup: Acetic Acid Titration Q1: Record the initial volume of NaOH, the concentration of the standardized NaOH solution in the text box below. • Turn off the magnetic stirrer and then remove the beaker. Dispose of its contents according to your teacher's instructions. • Place the waste container under the pH sensor and use the wash bottle to thoroughly clean the micro stir bar and the pH sensor. • Re-fill the buretso that there is ~50 mL of the standardized NaOH solution. • Read the initial volume of the titrant in the buretto 0.01 mL.
Acid-Base Titration Setup: Acetic Acid Titration Q2: Record the volume of the acetic acid solution that was added to the beaker in the text box below. • Using the 100-mL graduated cylinder measure 100 mL of distilled water and add it to a clean 250-mL beaker. • Using a clean 10-mL graduated cylinder or volumetric pipetmeasure 10.0 mL of acetic acid and add it to the 100 mL of distilled water. Caution:Always add acid to water. • Remove the waste container from the titration apparatus and replace it with the 250-mL beaker containing the acetic acid solution. • Reposition the pH sensor and then turn on the magnetic stirrer and begin stirring at a slow to medium speed.
Acid-Base Titration Collect Data: A.A. • Tap to start collecting data. • Carefully open the stopcock on the buret so that 2 to 3 drops per second are released. • Continue to collect data until the pH of the solution is 12. Continue to the next page.
Acid-Base Titration Q3:What substances are being formed in the beaker? Q4: What type of reaction is occurring? HC2H3O2 + NaOH ? Acetic Acid = HC2H3O2
Acid-Base Titration Collect Data: A.A. • When the pH of the solution is 12 close the stopcock. • Continue to the next page.
Acid-Base Titration Collect Data: A.A. • Tap to stop data collection. • Record the final volume of NaOH in the buret.
Acid-Base Titration Data Analysis: Acetic Acid • * To View a Page in the Journal: • Tap to open the Journal screen. • Tap or to scroll through the thumbnail size pages of the journal. • Tap the thumbnail image of the page to view it. • Determine the total volume of NaOH used in the titration. Follow steps a-c below. • Enter the final volume of NaOH in the text box on the right. • Record the initial volume of NaOH in the text box on the right • Subtract the initial volume of NaOH from the final volume of NaOH and record the total volume in the text box on the right. Volume NaOH = Final Volume NaOH - Initial Volume of NaOH • Note:Initial and final volumes of NaOH were recorded earlier • in the journal. Refer to the journal to get these numbers.*
Acid-Base Titration Data Analysis: Acetic Acid 2. Create a calculation to convert drop count to volume (mL). * CalcVolume2 = [drop count]*(volume of titrant/final drop count) *To Create a Calculation: • Tap to open the Experiment Tools screen. • Tap CALCULATED DATAto open the calculator. • Enter the calculation in the space provided. • Tap Measurements to insert collected data into the calculation. Note: [drop count] = the collected data volume of titrant = total volume of NaOH used (it is in your journal) final drop count = the total number of drops added (it is displayed on the right)
Acid-Base Titration 3. Change the x-axis to display CalcVolume2 of NaOH. • Tap then to open the Graph Properties screen. • Tap the Measurement box for the x-axis and tap CalcVolume1. Then tap OK. Tap to scale the axes. 4. Display the volume of NaOH at the equivalence point. The equivalence point is the point on the curve with the greatest slope and is found using the slope tool. • Tap then to see the slope. • Tap or buttons to move to nearby points. • Tap .
Acid-Base Titration Data Analysis: Acetic Acid • Calculate the molar concentration of acetic acid. Follow the steps below: • Determine the number of moles of NaOH added using the volume of NaOH at the equivalence point and the molarity of the standardized the NaOH solution. • Convert from moles of NaOH to moles of acetic acid using the balanced chemical equation. • Use the moles of acetic acid and the starting volume of acetic acid to determine the molarity of the acid. • Note: the values need for the calculations above can be found in your journal .
Acid-Base Titration Analysis • What is the significance of the point on the titration curve where the slope is the steepest?
Acid-Base Titration Analysis 2. What trend did you notice in the slope of the titration curve between the start of the titration and the equivalence point?
Acid-Base Titration Analysis • What is the likelihood that the concentration of acid and base is exactly equal at the experimentally determined equivalence point? Explain your reasoning.
Acid-Base Titration Analysis 4. What difference do you notice between the start of the titration and the equivalence point in the appearance of the titration curve for the two different acids?
Acid-Base Titration Analysis 5. What is the difference between pH at the equivalence points between the two titration curves for the different acids? Explain.
Acid-Base Titration Synthesis • How could you determine the concentration of an unknown sodium hydroxide solution?
Acid-Base Titration Synthesis 2. Use the Prediction Buttonto sketch the titration curve for an acid that is weaker than acetic acid. *To Draw a Prediction: • Tap to open the tool palette. • Tap then use your finger to draw your prediction. • Tap when finished. • If you make a mistake, tap to clear your prediction.
Acid-Base Titration Synthesis • Explain the difference between the strength and the concentration of an acid solution.
Acid-Base Titration Multiple Choice Base • For the titration of an acid with a base, the pH will start _________ and finish ________. • low; low • high; low • low; high • high; high Acid Hint:the acid is the analyte in the beaker and the base is the titrant in the buret.
Acid-Base Titration Multiple Choice • On a titration curve, the equivalence point is ____________________________. • the point with the smallest slope. • the point with the greatest slope. • the point with a slope of zero. • when the pH is equal to zero. Titration Curve
Acid-Base Titration Multiple Choice • An ________ in the hydronium ion concentration in an aqueous solution causes the hydroxide ion concentration to ________ and the pH of the solution to _________. • Increase; increase; increase • Decrease; decrease; decrease • Increase; decrease; decrease • Increase; decrease; increase Acidic solution
Acid-Base Titration Multiple Choice • A reaction between an acid and a base is called a(n) ______________. • pH reaction • titration reaction • s-shaped curve • neutralization reaction
Acid-Base Titration Multiple Choice • Titrations are used to __________________. • determine the concentration of known solutions • determine the pH of known solutions • determine the type of molecules in a solution • differentiate an acid from a base
Acid-Base Titration Congratulations! You have completed the lab. Please remember to follow your teacher's instructions for cleaning-up and submitting your lab.